Lupin Limited
Indian Pharmaceutical Exporter · Tuberculosis Medications Specialist · $271.4M Total Trade · DGFT Verified
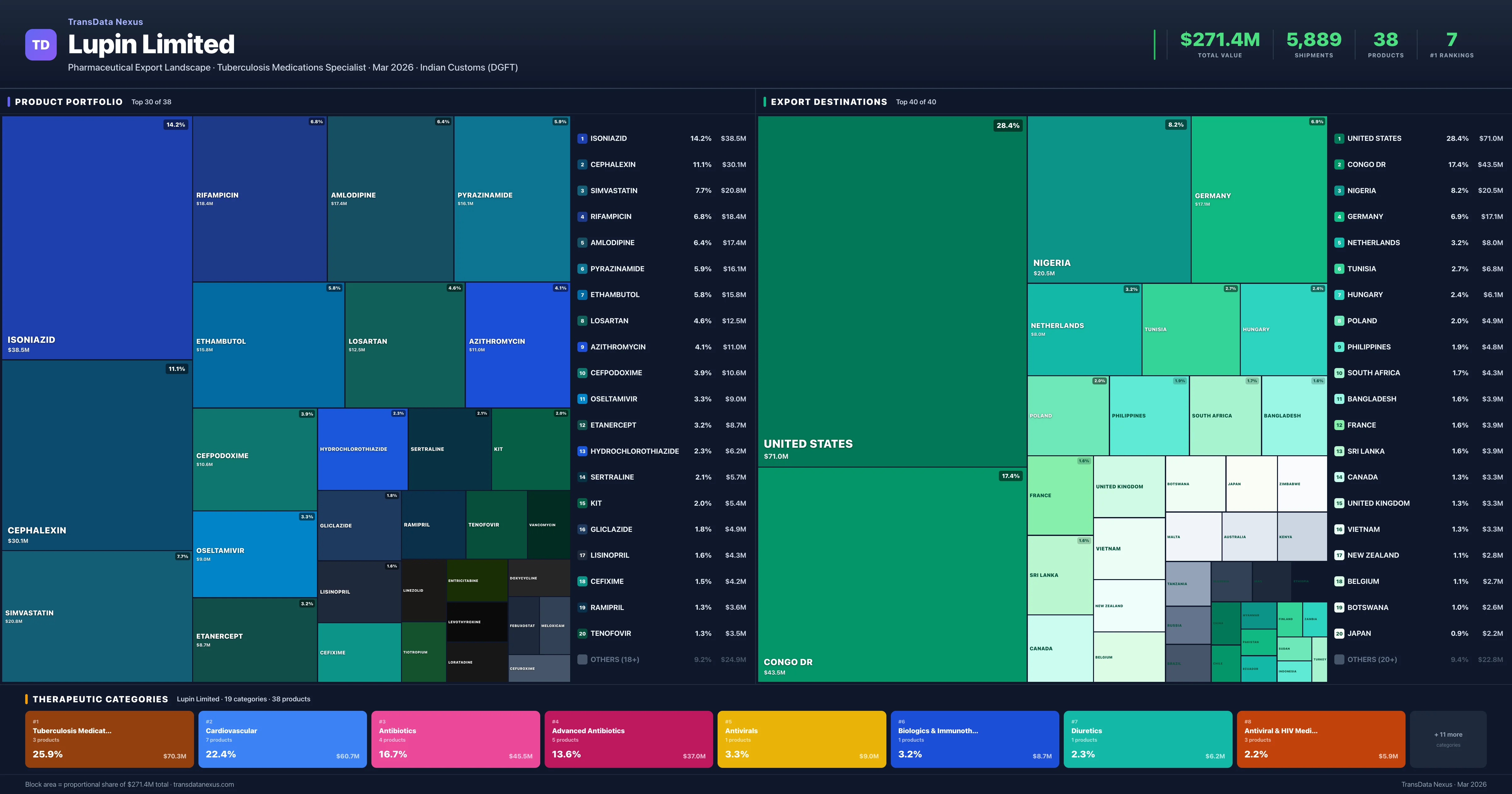
Lupin Limited is an Indian pharmaceutical exporter with a total trade value of $271.4M across 38 products in 19 therapeutic categories. Based on 5,889 verified export shipments from Indian Customs (DGFT) records, Lupin Limited is the #1 Indian exporter in 7 products including Isoniazid, Rifampicin, Pyrazinamide. Top exports include Isoniazid ($38.5M), Cephalexin ($30.1M), Simvastatin ($20.8M).
Lupin Limited — Export Portfolio & Destination Treemap
Who is Lupin Limited? — Company Overview & Market Position
Lupin Limited, established in 1968 by Dr. Desh Bandhu Gupta, is a leading Indian multinational pharmaceutical company headquartered in Mumbai, Maharashtra. The company specializes in the development, manufacturing, and marketing of a diverse range of pharmaceutical products, including branded and generic formulations, biosimilars, over-the-counter drugs, and active pharmaceutical ingredients (APIs). Lupin's therapeutic portfolio spans areas such as anti-tuberculosis, cardiology, diabetology, respiratory, and central nervous system disorders.
As of March 2026, Lupin reported a total export value of $271.4 million USD, with 5,889 shipments across 38 products in 19 therapeutic categories. The company's top five exported products include Isoniazid ($38.5 million, 1.7% market share), Cephalexin ($30.1 million, 8.6% market share), Simvastatin ($20.8 million, 16.5% market share), Rifampicin ($18.4 million, 1.4% market share), and Amlodipine ($17.4 million, 0.2% market share). The primary therapeutic categories for Lupin's exports are Tuberculosis Medications (25.9%), Cardiovascular (22.4%), and Antibiotics (16.7%). Notably, Lupin holds the number one market position in Isoniazid, Rifampicin, Pyrazinamide, Ethambutol, Etanercept, Tiotropium, and related combination products. The company's portfolio concentration is significant, with the top five products accounting for 46.1% of total exports.
What Does Lupin Limited Export? — Product Portfolio Analysis
Lupin Limited Therapeutic Categories — 19 Specializations
Lupin Limited operates across 19 therapeutic categories, with Tuberculosis Medications (25.9%), Cardiovascular (22.4%), Antibiotics (16.7%) representing the largest segments by export value. The portfolio is well-diversified with the top 5 products representing 46% of exports.
Tuberculosis Medications
3 products · 25.9% · $70.3M
Cardiovascular
7 products · 22.4% · $60.7M
Antibiotics
4 products · 16.7% · $45.5M
Advanced Antibiotics
5 products · 13.6% · $37.0M
Antivirals
1 products · 3.3% · $9.0M
Biologics & Immunotherapy
1 products · 3.2% · $8.7M
Diuretics
1 products · 2.3% · $6.2M
Antiviral & HIV Medications
3 products · 2.2% · $5.9M
CNS & Psychiatric
1 products · 2.1% · $5.7M
Product Portfolio — Top 30 by Export Value
Lupin Limited exports 38 pharmaceutical products across 19 therapeutic categories. Market leader (#1 exporter) in 7 products.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Isoniazid | Tuberculosis Medications | $38.5M | 769 | 1.7% | 1 |
| 2 | Cephalexin | Antibiotics | $30.1M | 601 | 8.6% | 2 |
| 3 | Simvastatin | Cardiovascular | $20.8M | 416 | 16.5% | 2 |
| 4 | Rifampicin | Advanced Antibiotics | $18.4M | 369 | 1.4% | 1 |
| 5 | Amlodipine | Cardiovascular | $17.4M | 360 | 0.2% | 7 |
| 6 | Pyrazinamide | Tuberculosis Medications | $16.1M | 321 | 1.7% | 1 |
| 7 | Ethambutol | Tuberculosis Medications | $15.8M | 317 | 1.4% | 1 |
| 8 | Losartan | Cardiovascular | $12.5M | 360 | 4.2% | 6 |
| 9 | Azithromycin | Antibiotics | $11.0M | 220 | 6.0% | 2 |
| 10 | Cefpodoxime | Advanced Antibiotics | $10.6M | 297 | 13.3% | 3 |
| 11 | Oseltamivir | Antivirals | $9.0M | 184 | 7.3% | 5 |
| 12 | Etanercept | Biologics & Immunotherapy | $8.7M | 174 | 14.2% | 1 |
| 13 | Hydrochlorothiazide | Diuretics | $6.2M | 198 | 1.5% | 6 |
| 14 | Sertraline | CNS & Psychiatric | $5.7M | 113 | 1.0% | 4 |
| 15 | Kit | Medical Devices & Diagnostics | $5.4M | 108 | 1.2% | 1 |
| 16 | Gliclazide | Advanced Diabetes Medications | $4.9M | 145 | 1.6% | 7 |
| 17 | Lisinopril | Cardiovascular | $4.3M | 86 | 7.1% | 2 |
| 18 | Cefixime | Advanced Antibiotics | $4.2M | 83 | 2.4% | 2 |
| 19 | Ramipril | Cardiovascular | $3.6M | 155 | 4.0% | 10 |
| 20 | Tenofovir | Antiviral & HIV Medications | $3.5M | 70 | 0.1% | 12 |
| 21 | Vancomycin | Antibiotics | $2.5M | 49 | 2.7% | 8 |
| 22 | Linezolid | Advanced Antibiotics | $2.3M | 46 | 6.1% | 3 |
| 23 | Tiotropium | Respiratory | $2.3M | 45 | 3.3% | 1 |
| 24 | Emtricitabine | Antiviral & HIV Medications | $2.1M | 43 | 1.8% | 4 |
| 25 | Levothyroxine | Diabetes & Endocrine | $2.0M | 41 | 2.6% | 10 |
| 26 | Loratadine | Antihistamines & Allergy | $2.0M | 41 | 1.2% | 7 |
| 27 | Doxycycline | Antibiotics | $1.9M | 39 | 1.6% | 8 |
| 28 | Febuxostat | Lipid & Metabolism | $1.5M | 30 | 3.1% | 4 |
| 29 | Meloxicam | Analgesics & Antipyretics | $1.5M | 30 | 0.8% | 4 |
| 30 | Cefuroxime | Advanced Antibiotics | $1.5M | 40 | 1.1% | 12 |
Lupin Limited exports 38 pharmaceutical products across 19 therapeutic categories with a total export value of $271.4M. The company is the #1 Indian exporter in 7 products: Isoniazid, Rifampicin, Pyrazinamide, Ethambutol, Etanercept and 2 more. The top category is Tuberculosis Medications (25.9% of portfolio), followed by Cardiovascular (22.4%), reflecting a diversified portfolio where the top 5 products account for only 46.1% of total value.
Showing top 30 of 38 products. Contact us for complete data.
All 38 Products
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Lupin Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Lupin Limited, established in 1968 by Dr. Desh Bandhu Gupta, is a leading Indian multinational pharmaceutical company headquartered in Mumbai, Maharashtra. The company specializes in the development, manufacturing, and marketing of a diverse range of pharmaceutical products, including branded and generic formulations, biosimilars, over-the-counter drugs, and active pharmaceutical ingredients (APIs). Lupin's therapeutic portfolio spans areas such as anti-tuberculosis, cardiology, diabetology, respiratory, and central nervous system disorders.
As of March 2026, Lupin reported a total export value of $271.4 million USD, with 5,889 shipments across 38 products in 19 therapeutic categories. The company's top five exported products include Isoniazid ($38.5 million, 1.7% market share), Cephalexin ($30.1 million, 8.6% market share), Simvastatin ($20.8 million, 16.5% market share), Rifampicin ($18.4 million, 1.4% market share), and Amlodipine ($17.4 million, 0.2% market share). The primary therapeutic categories for Lupin's exports are Tuberculosis Medications (25.9%), Cardiovascular (22.4%), and Antibiotics (16.7%). Notably, Lupin holds the number one market position in Isoniazid, Rifampicin, Pyrazinamide, Ethambutol, Etanercept, Tiotropium, and related combination products. The company's portfolio concentration is significant, with the top five products accounting for 46.1% of total exports.
2Manufacturing Facilities
Lupin operates a global network of state-of-the-art manufacturing facilities, including 15 sites and 7 research centers worldwide. In India, the company has 12 manufacturing plants and 2 research centers located in Jammu, Mandideep, Indore, Ankaleswar, Dabasa, Tarapur, Aurangabad, Nagpur, Goa, Visakhapatnam, and Sikkim. These facilities are equipped to produce a wide range of pharmaceutical formulations, including tablets, capsules, syrups, and injections. Lupin's manufacturing plants adhere to stringent quality standards and are compliant with Good Manufacturing Practices (GMP) to ensure the production of high-quality pharmaceutical products.
3Key Leadership
Lupin Limited is led by a team of experienced executives committed to driving the company's growth and innovation. Vinita D. Gupta serves as the Chief Executive Officer and Executive Director, overseeing the company's strategic direction and operations. Ramesh Swaminathan holds the position of Executive Director, Global Chief Financial Officer, Chief Risk Officer, and Head of API Plus SBU and IT, responsible for financial management and strategic initiatives. Nilesh Deshbandhu Gupta serves as the Managing Director and Executive Director, contributing to the company's leadership and strategic planning. Other key executives include Christoph Funke, Chief Technical Operations Officer; Dr. Shahin Fesharaki, Chief Scientific Officer; Ravi Agrawal, Senior Vice President of Investor Relations and M&A; Amit Kumar Gupta, Company Secretary and Compliance Officer; Rajalakshmi Azariah, Vice President and Global Head of Corporate Communications; Dr. Fabrice Egros, President of Corporate Development; and Yashwant S. Mahadik, President of Global Human Resources.
Where Does Lupin Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Lupin Limited has established a strong presence in several regulated markets, including the United States, European Union, United Kingdom, Australia, and Japan. In the United States, Lupin operates through its subsidiary, Lupin Pharmaceuticals Inc., which has manufacturing, research and development, and commercial divisions for generics, complex generics, biosimilars, and branded pharmaceuticals. The company achieved a significant milestone in 2017-18 when U.S. revenues crossed $1 billion.
In the European Union, Lupin has expanded its footprint by acquiring a branded product portfolio from Shionogi & Co., Ltd. in Japan, effective December 1, 2016. This acquisition has enhanced Lupin's presence in the European market.
In the United Kingdom, Lupin Healthcare, a subsidiary of Lupin, offers a portfolio of high-quality generics with a focus on anti-retroviral and oral contraceptives. The company has also launched medicines in the neuromuscular space for the UK market, transitioning from a standard generics business to a more specialist organization that partners with the NHS to deliver value.
In Australia, Lupin entered the market through its subsidiary, Generic Health Pte. Ltd., and acquired the worldwide marketing rights to the over-100-year-old Australian brand Goanna, used for pain management.
In Japan, Lupin's subsidiary Kyowa Pharmaceutical Industry Co., Ltd. has been active, and in 2014, Lupin entered into a strategic joint venture agreement with Yoshindo Inc. to create YL Biologics for the clinical development of biosimilars.
2Emerging Markets
Lupin Limited has made strategic inroads into emerging markets, including Africa, Latin America, and Southeast Asia. In Mexico, the company acquired a 100% equity stake in Laboratories Grin, S.A. De C.V. in 2014, marking its entry into the Latin American pharmaceuticals market. In Brazil, Lupin expanded its presence by acquiring a 100% stake in Medquímica Indústria Farmacêutica S.A. in May 2015.
Lupin's commitment to global health is further demonstrated through its partnership with the TB Alliance for the development and commercialization of the investigational drug Telacebec, aimed at treating multiple mycobacterial diseases, including tuberculosis, leprosy, and Buruli ulcer.
3Geographic Strategy
Lupin Limited's geographic strategy is characterized by diversification across developed and emerging markets, mitigating concentration risk and enhancing global reach. The company's strategic acquisitions, such as the purchase of Laboratories Grin in Mexico and Medquímica in Brazil, have bolstered its presence in Latin America. In the United States, Lupin's establishment of manufacturing and R&D facilities, along with its significant revenue milestones, underscores its commitment to the North American market. The company's focus on emerging markets, including Africa and Southeast Asia, aligns with its objective to expand access to essential medicines globally.
Lupin Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Lupin Limited's U.S. subsidiary, Lupin Pharmaceuticals Inc., has achieved significant milestones in the U.S. market. The company operates manufacturing, research and development, and commercial divisions for generics, complex generics, biosimilars, and branded pharmaceuticals. In 2015, Lupin established the Center of Excellence for Inhalation Research in Coral Springs, Florida, and subsequently acquired Novel Laboratories, Inc. to create Lupin Somerset, its first manufacturing facility in the U.S. In 2017-18, Lupin's U.S. revenues crossed $1 billion, reflecting its strong position in the U.S. pharmaceutical market.
2WHO & EU GMP
Lupin Limited's manufacturing facilities adhere to stringent quality standards, including compliance with Good Manufacturing Practices (GMP) as recognized by the World Health Organization (WHO) and the European Union (EU). The company's commitment to quality is evident in its state-of-the-art manufacturing sites and research centers, which are equipped to produce a wide range of pharmaceutical formulations, including tablets, capsules, syrups, and injections.
3CDSCO & Indian Regulatory
In India, Lupin Limited operates under the regulatory oversight of the Central Drugs Standard Control Organisation (CDSCO). The company's manufacturing facilities in India are licensed by the CDSCO, ensuring compliance with national pharmaceutical regulations. Lupin's commitment to quality and regulatory compliance is further demonstrated through its adherence to Good Manufacturing Practices (GMP) and other relevant standards.
4Recent Regulatory Actions
As of March 2026, there are no publicly available records of recent regulatory actions, such as Form 483 observations, warning letters, or import alerts, against Lupin Limited. The company maintains a strong commitment to regulatory compliance and quality standards across its global operations.
Lupin Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Lupin Limited operates in a highly competitive pharmaceutical industry, facing competition from both multinational and regional companies across various therapeutic segments. In the United States, Lupin competes with other generic pharmaceutical companies, leveraging its manufacturing capabilities and product portfolio to capture market share. In Europe, the company's acquisition of a branded product portfolio from Shionogi & Co., Ltd. in Japan has enhanced its
Frequently Asked Questions — Lupin Limited
How many pharmaceutical products does Lupin Limited export from India?
Lupin Limited exports 38 pharmaceutical products across 19 therapeutic categories. The top exports are Isoniazid ($38.5M), Cephalexin ($30.1M), Simvastatin ($20.8M), Rifampicin ($18.4M), Amlodipine ($17.4M). Total export value is $271.4M.
What is Lupin Limited's total pharmaceutical export value?
Lupin Limited's total pharmaceutical export value is $271.4M, based on 5,889 verified shipments recorded in Indian Customs (DGFT) data.
In which products is Lupin Limited the #1 Indian exporter?
Lupin Limited is the #1 Indian exporter in 7 products: Isoniazid (1.7% market share), Rifampicin (1.4% market share), Pyrazinamide (1.7% market share), Ethambutol (1.4% market share), Etanercept (14.2% market share), Kit (1.2% market share), Tiotropium (3.3% market share).
What therapeutic categories does Lupin Limited cover?
Lupin Limited exports across 19 therapeutic categories. The largest are Tuberculosis Medications (25.9%, 3 products), Cardiovascular (22.4%, 7 products), Antibiotics (16.7%, 4 products).
Get Full Lupin Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Lupin Limited identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Lupin Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 5,889 individual customs records matching Lupin Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
38 Products Tracked
19 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.