In February 2026, the EMA concluded a PSUSA for simvastatin, resulting in a variation to the marketing authorization to enhance safety information. (ema.europa.eu) This emphasizes the importance of ongoing pharmacovigilance and compliance with updated regulatory requirements.
In December 2025, the EMA updated the product information for Cholib, a combination product containing simvastatin, reflecting new safety data. (ema.europa.eu) This highlights the dynamic nature of regulatory oversight and the necessity for manufacturers to stay informed about changes affecting combination therapies.
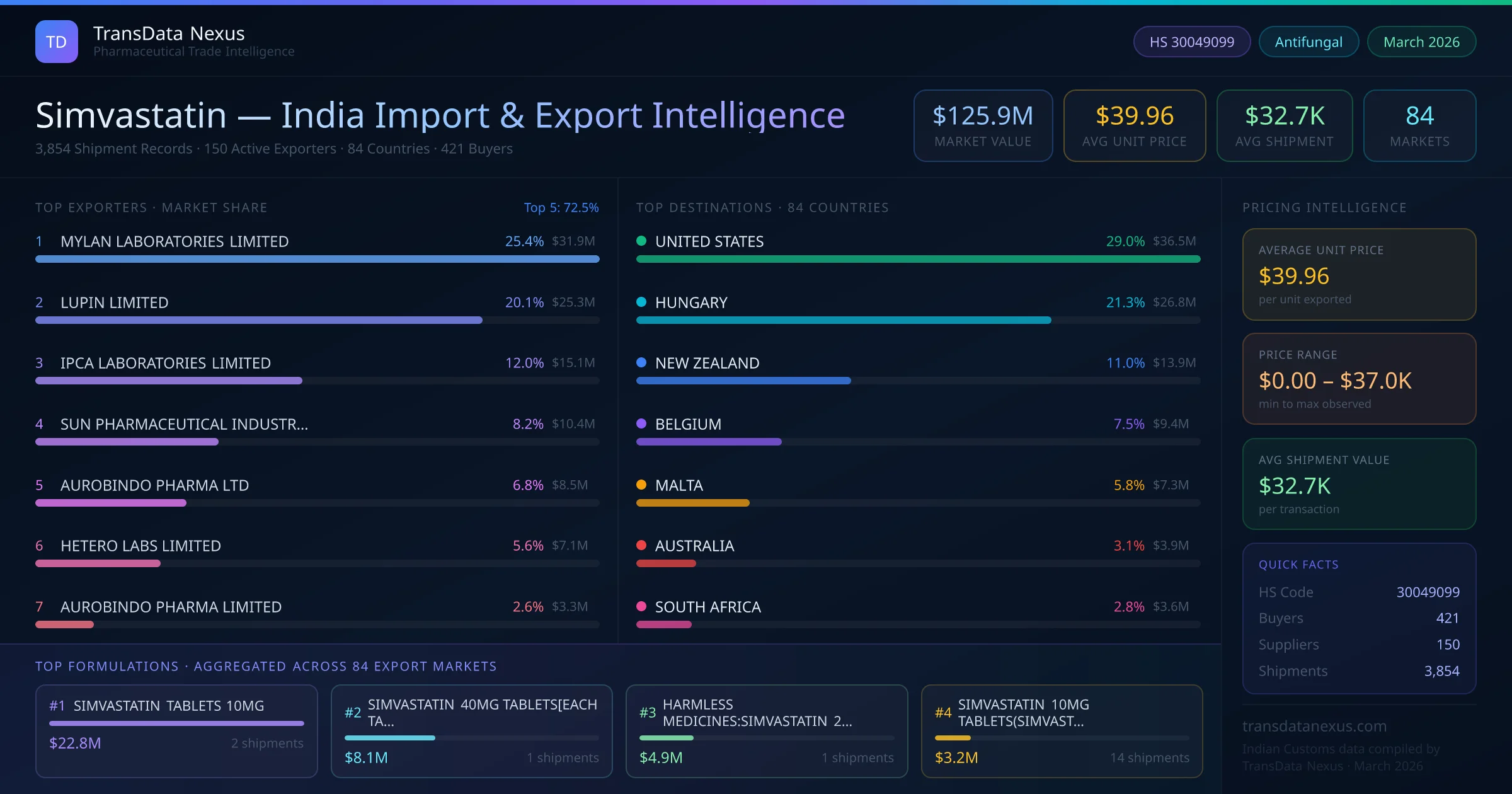
In January 2026, the NPPA revised the ceiling price for simvastatin under the DPCO, impacting pricing strategies for domestic and export markets. Manufacturers must adjust their pricing models to remain competitive while adhering to regulatory mandates.
In March 2026, the FDA issued updated guidance on bioequivalence requirements for generic simvastatin products, affecting the approval process for new ANDAs. Indian exporters must ensure their products meet these revised standards to maintain access to the U.S. market.
In April 2026, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of simvastatin and underscoring its global importance in managing hypercholesterolemia. This reinforces the need for consistent quality and availability of simvastatin in international markets.
These developments highlight the dynamic regulatory environment surrounding simvastatin and the necessity for Indian exporters to remain vigilant and adaptable to maintain and expand their market presence.