In February 2026, the EMA's Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion recommending the approval of Fubelv, a biosimilar etanercept developed by Biosimilar Collaborations Ireland Limited, for multiple indications including rheumatoid arthritis and plaque psoriasis. (ema.europa.eu)
In January 2026, the NPPA conducted a review of biologic drug pricing, including etanercept, to assess market trends and ensure affordability. While no immediate price controls were implemented, the review indicates potential future regulatory actions.
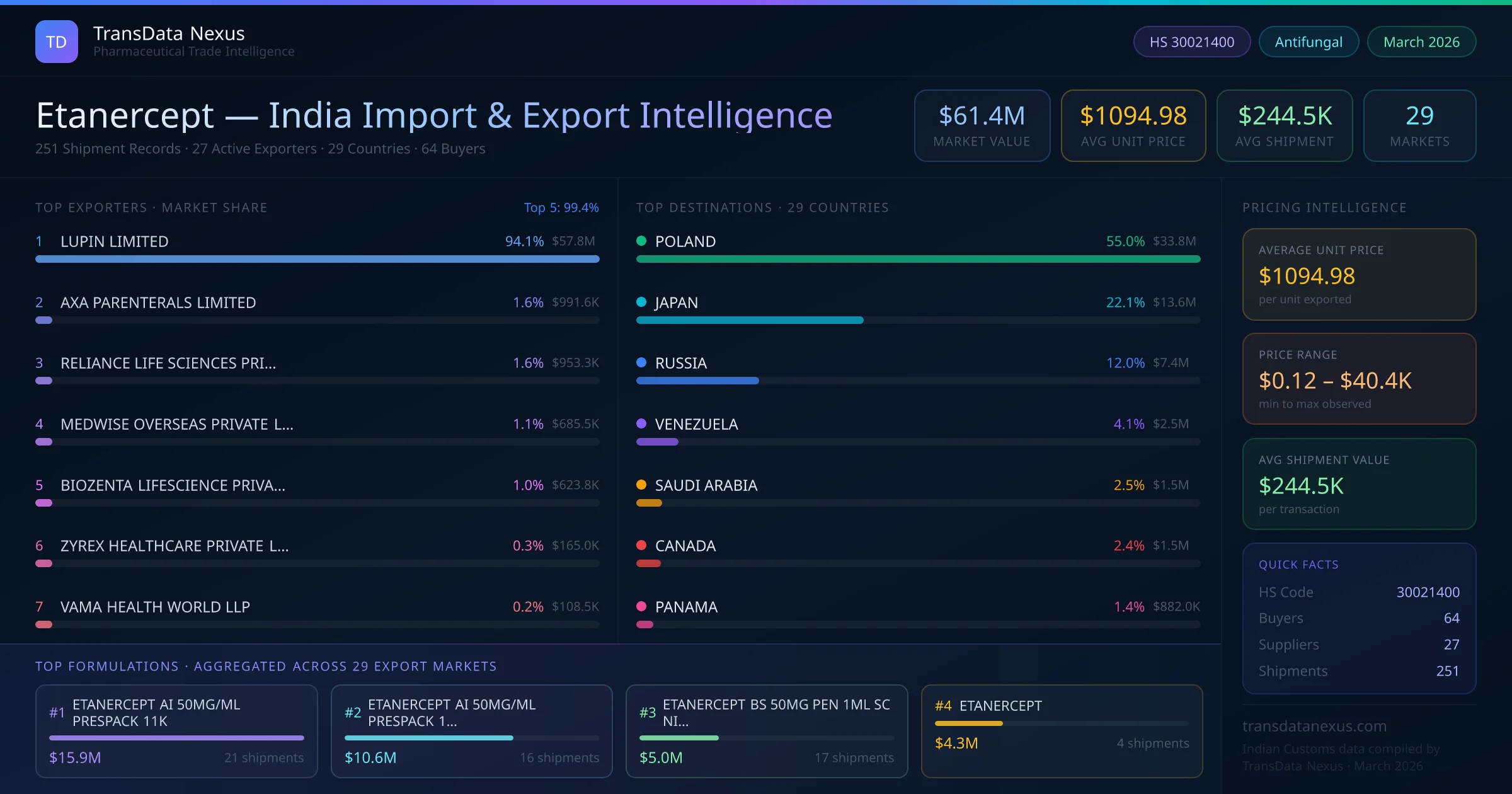
In December 2025, the CDSCO issued revised guidelines for the export of biologics, emphasizing stringent quality control measures and documentation requirements to align with international standards.
In November 2025, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of etanercept for the treatment of autoimmune diseases, highlighting its global therapeutic significance.
In October 2025, the EMA approved a new manufacturing site for Erelzi, a biosimilar etanercept, enhancing production capacity to meet increasing demand in the European market.
These developments reflect the dynamic regulatory and market environment surrounding etanercept, with implications for Indian exporters in terms of compliance, competition, and market access strategies.