In February 2026, the EMA's Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion recommending a change to the terms of the marketing authorization for Stelara, expanding its indication to include the treatment of moderately to severely active Crohn’s disease in pediatric patients from the age of 2 years and older. This expansion may increase the demand for ustekinumab in the European market.
In September 2025, the EMA granted marketing authorization for Usrenty, a biosimilar of ustekinumab, for the treatment of plaque psoriasis, psoriatic arthritis, and Crohn's disease. This approval introduces additional competition in the European market, potentially affecting market share and pricing for existing products.
In November 2025, the EMA approved Usgena, another biosimilar of ustekinumab, further intensifying competition in the European market. The entry of multiple biosimilars may lead to price reductions and increased accessibility for patients.
In August 2025, the European Commission withdrew the marketing authorization for Eksunbi, a ustekinumab product, at the request of the marketing authorization holder, Samsung Bioepis NL B.V., due to commercial reasons. This withdrawal may impact the availability of certain ustekinumab products in the EU market.
In December 2025, the European Commission withdrew the marketing authorization for Absimky, another ustekinumab product, at the request of the marketing authorization holder, Accord Healthcare S.L.U., also due to commercial reasons. Such withdrawals may influence market dynamics and present opportunities for other manufacturers.
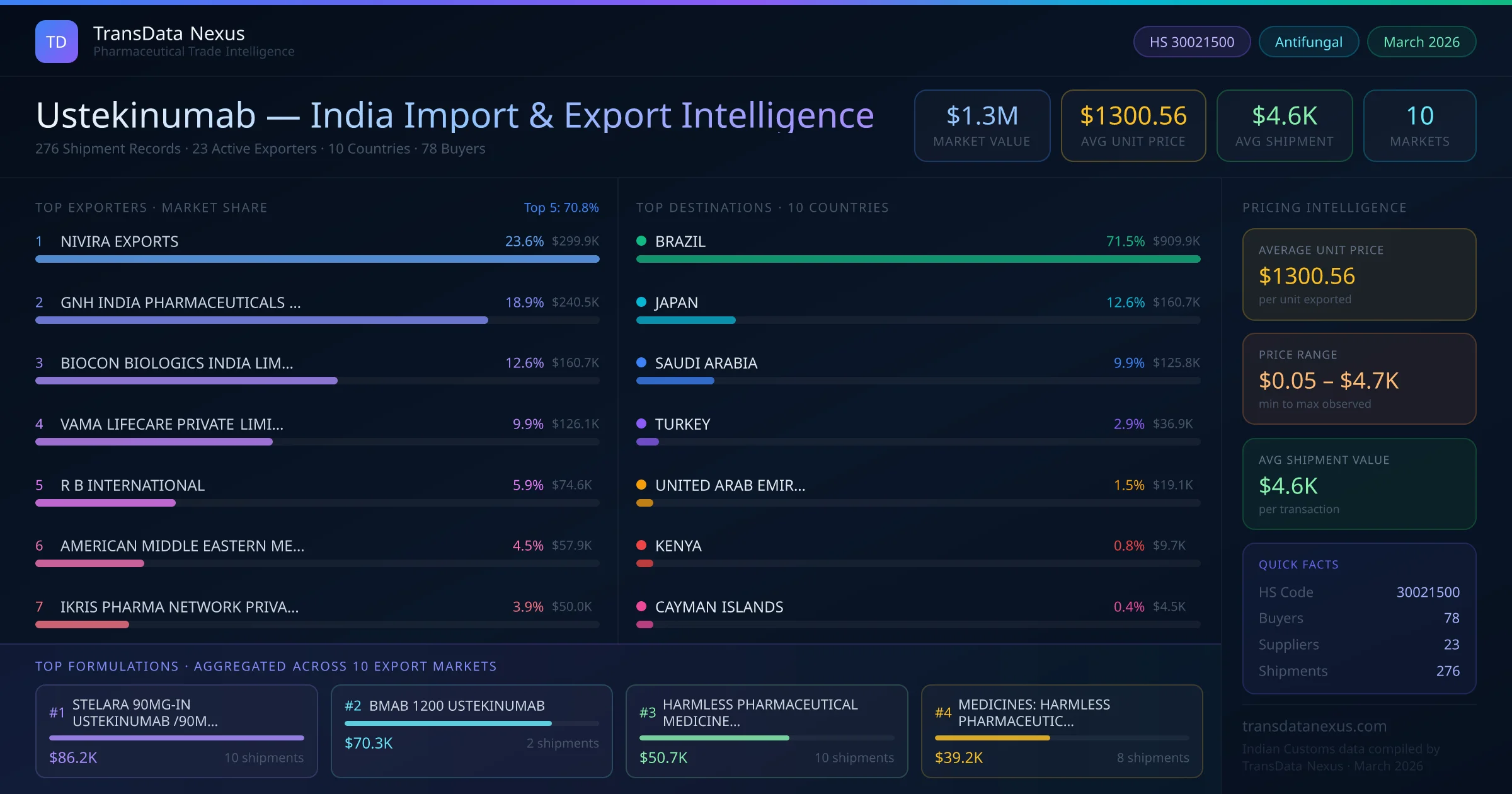
These developments underscore the evolving regulatory and competitive landscape for ustekinumab, highlighting the importance for Indian exporters to stay informed and adapt to changing market conditions.