In August 2023, the WHO updated its Model List of Essential Medicines, incorporating new formulations and treatment regimens for tuberculosis, including those involving isoniazid. These updates aim to enhance treatment efficacy and patient adherence. (who.int)
In December 2025, the EMA's Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion recommending changes to the marketing authorization for Dovprela (pretomanid), a drug used in combination with isoniazid for treating multidrug-resistant tuberculosis. This development reflects ongoing advancements in TB treatment protocols. (ema.europa.eu)
In January 2024, Anuh Pharma Ltd., an Indian manufacturer, received WHO Prequalification for its isoniazid active pharmaceutical ingredient (API), indicating compliance with international quality standards and enhancing its export potential. (extranet.who.int)
In November 2021, the WHO released updated treatment guidelines for isoniazid-resistant tuberculosis, providing new recommendations to improve treatment outcomes for patients with drug-resistant TB strains. (who.int)
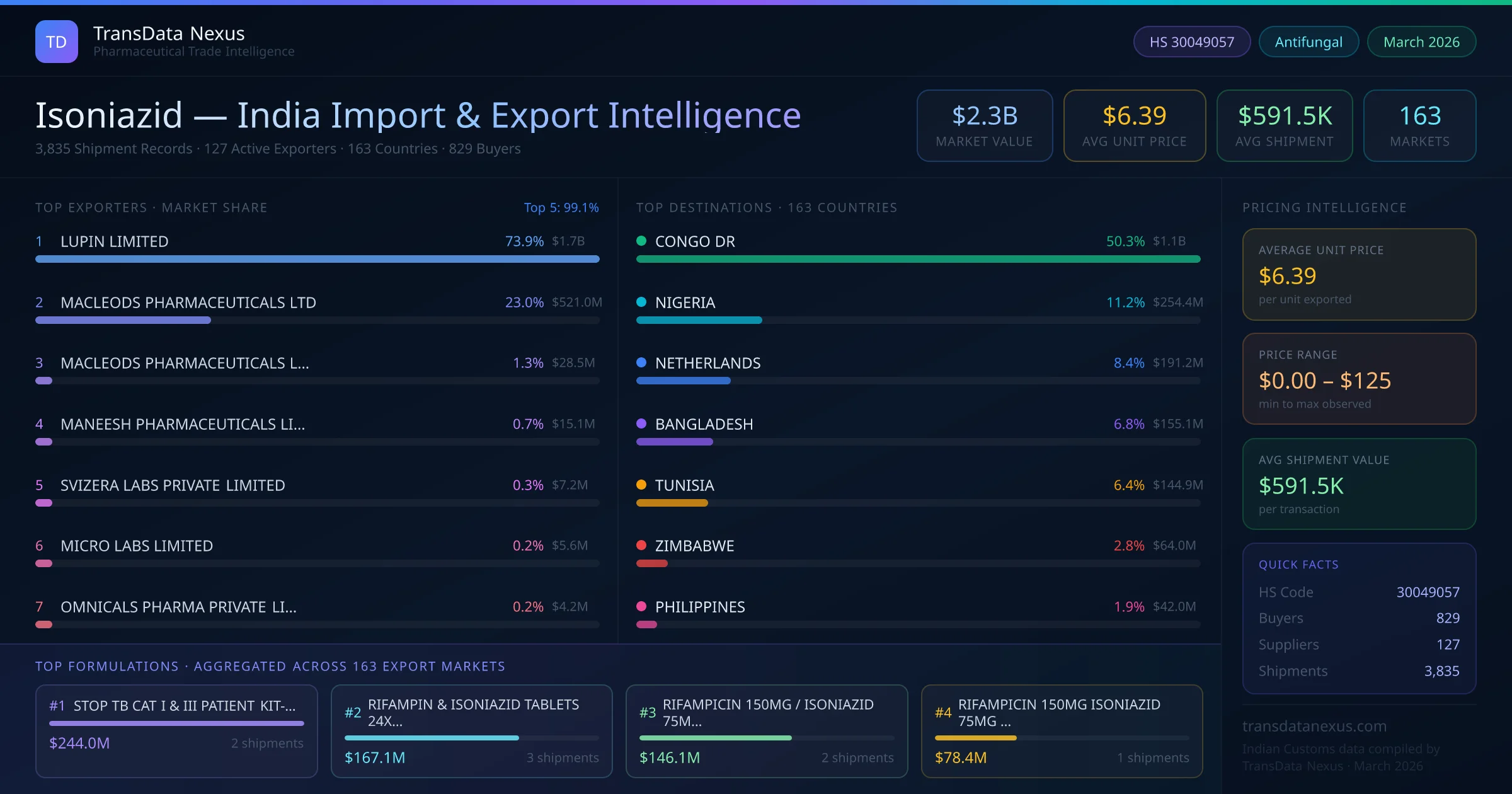
In March 2026, the NPPA revised the ceiling price for isoniazid under the DPCO, reflecting changes in production costs and market dynamics. Manufacturers and exporters must adjust their pricing strategies accordingly to remain compliant.
These developments underscore the dynamic nature of the regulatory and market landscape for isoniazid, necessitating continuous monitoring and compliance by stakeholders.