In November 2025, the FDA updated Import Alert 55-05 to include additional finished dosage forms and APIs due to concerns over microbiological contamination. This emphasizes the need for stringent quality control measures among exporters.
In October 2024, the FDA issued Import Alert 66-66, focusing on misbranded APIs that fail to meet labeling requirements. This alert serves as a reminder for exporters to ensure compliance with labeling standards to avoid market entry barriers.
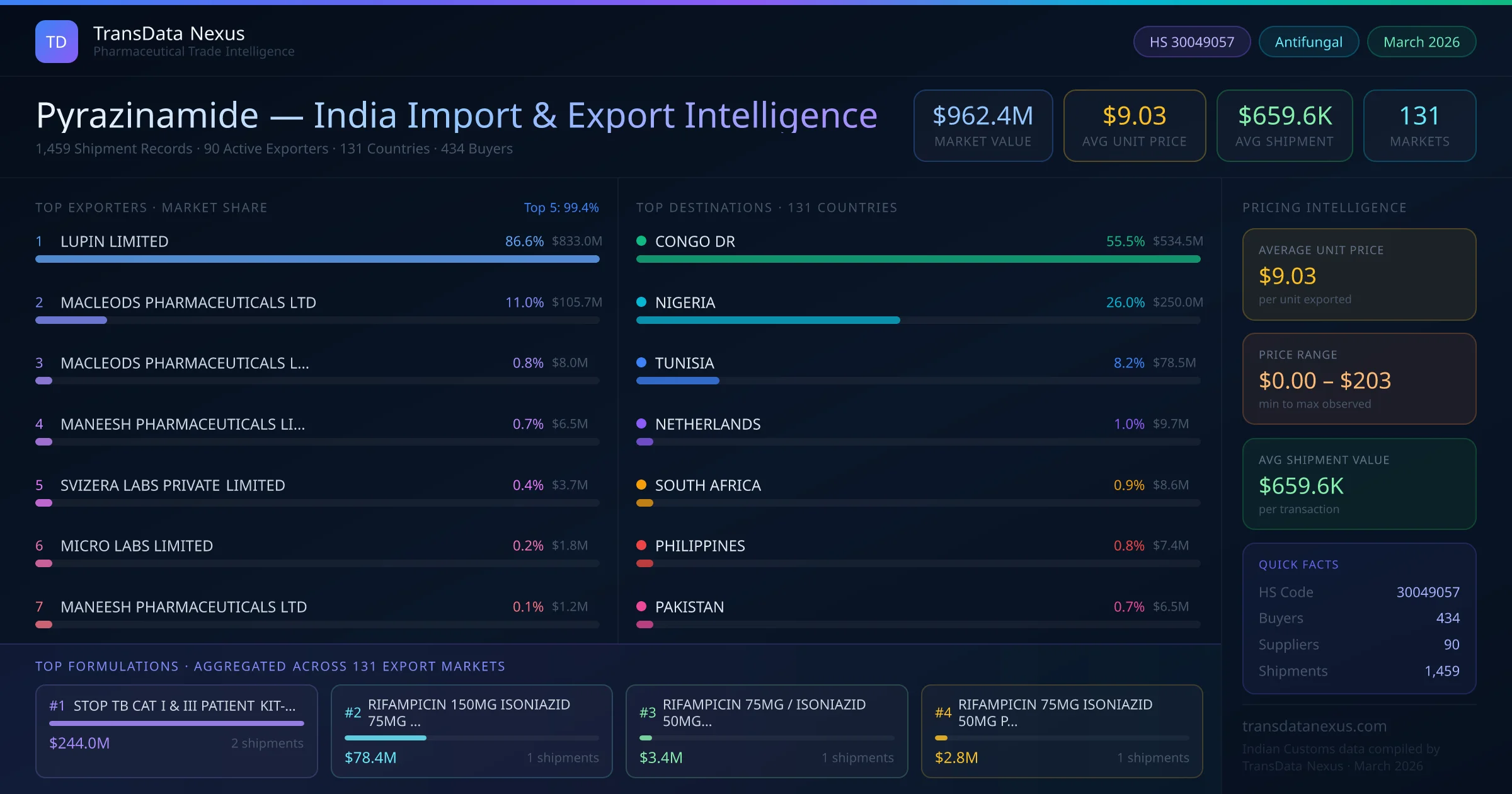
In March 2026, the NPPA revised the ceiling price for Pyrazinamide, reflecting changes in production costs and market dynamics. This adjustment impacts pricing strategies for both domestic sales and exports.
In February 2026, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of Pyrazinamide. This underscores the drug's continued importance in global tuberculosis treatment protocols.
In January 2026, the EMA issued new guidelines on GMP compliance for pharmaceutical manufacturers, affecting the production standards for Pyrazinamide intended for the EU market. Adherence to these guidelines is crucial for market access.
These developments highlight the dynamic regulatory environment affecting Pyrazinamide exports from India. Staying informed and compliant with international standards is essential for maintaining market access and ensuring public health.