In November 2025, the WHO released new Target Product Profiles (TPPs) for pediatric formulations of azithromycin, aiming to accelerate access to better medicines for children. (who.int) This initiative is expected to drive demand for age-appropriate azithromycin formulations, presenting opportunities for Indian exporters specializing in pediatric medicines.
In September 2025, the EMA concluded a referral procedure for azithromycin-containing medicinal products for systemic use, resulting in a legally binding decision across EU Member States. (ema.europa.eu) This decision underscores the importance of monitoring azithromycin's safety profile and ensuring its appropriate use to mitigate antimicrobial resistance risks.
In June 2025, the WHO published a fact sheet highlighting the increasing resistance of Neisseria gonorrhoeae to azithromycin, emphasizing the need for prudent antibiotic use and the development of new treatment guidelines. (who.int) This development may influence prescribing practices and demand for azithromycin in certain markets.
In March 2025, the WHO reported on the use of azithromycin in mass drug administration campaigns for yaws eradication, noting significant progress in countries like Papua New Guinea and the Solomon Islands. (who.int) These efforts highlight the continued global reliance on azithromycin for public health initiatives.
In January 2025, the WHO emphasized the inclusion of azithromycin in its Model List of Essential Medicines for Children, reflecting its critical role in treating pediatric infections and guiding procurement decisions worldwide. (who.int)
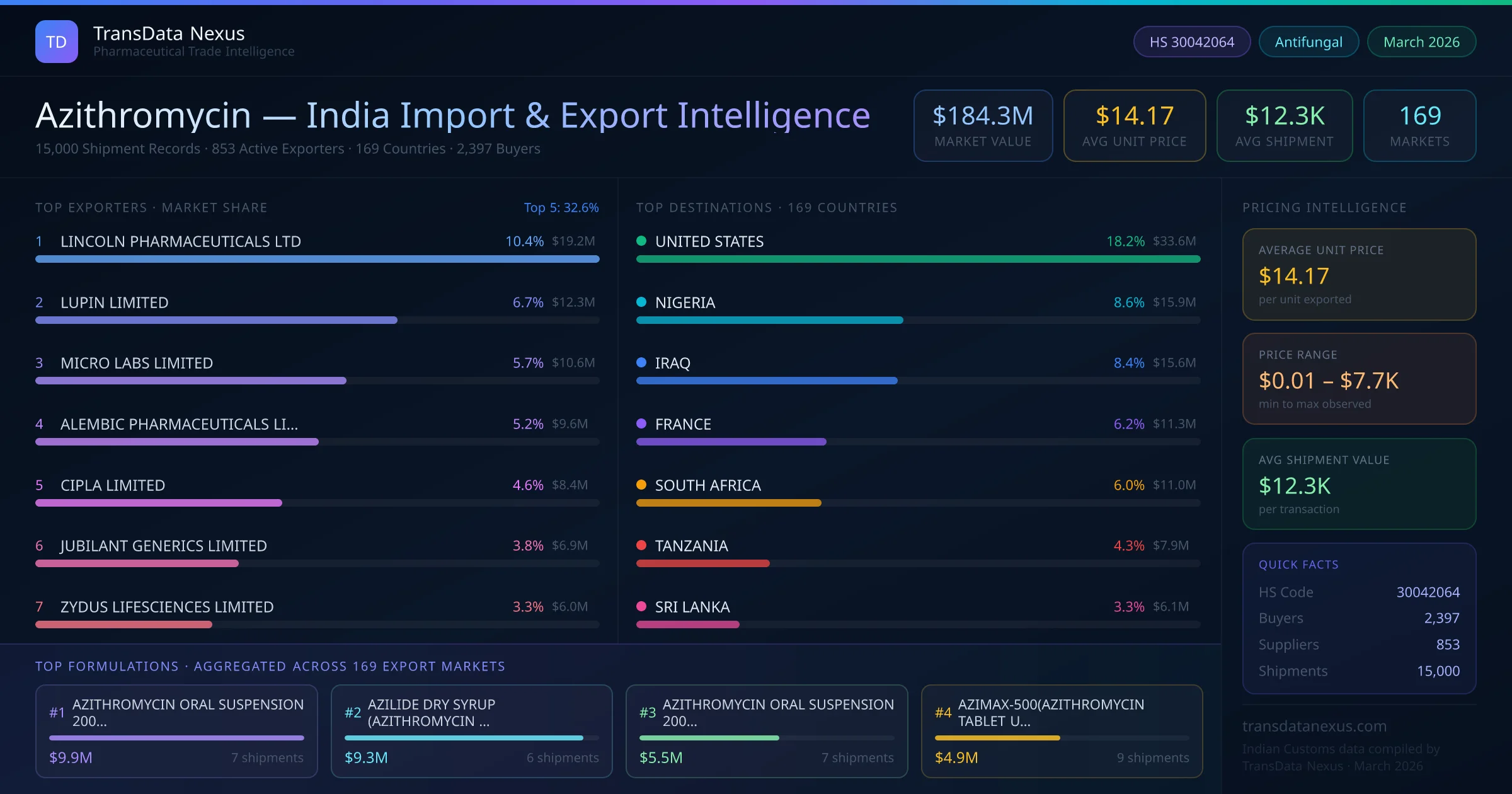
These developments underscore the dynamic regulatory and market landscape for azithromycin, necessitating continuous monitoring by Indian exporters to maintain compliance and capitalize on emerging opportunities.