Unique Pharma,
Pharmaceutical Importer · Congo Dr · Antimalarial & Antiparasitic Focus · $9.2M Total Trade · DGFT Verified
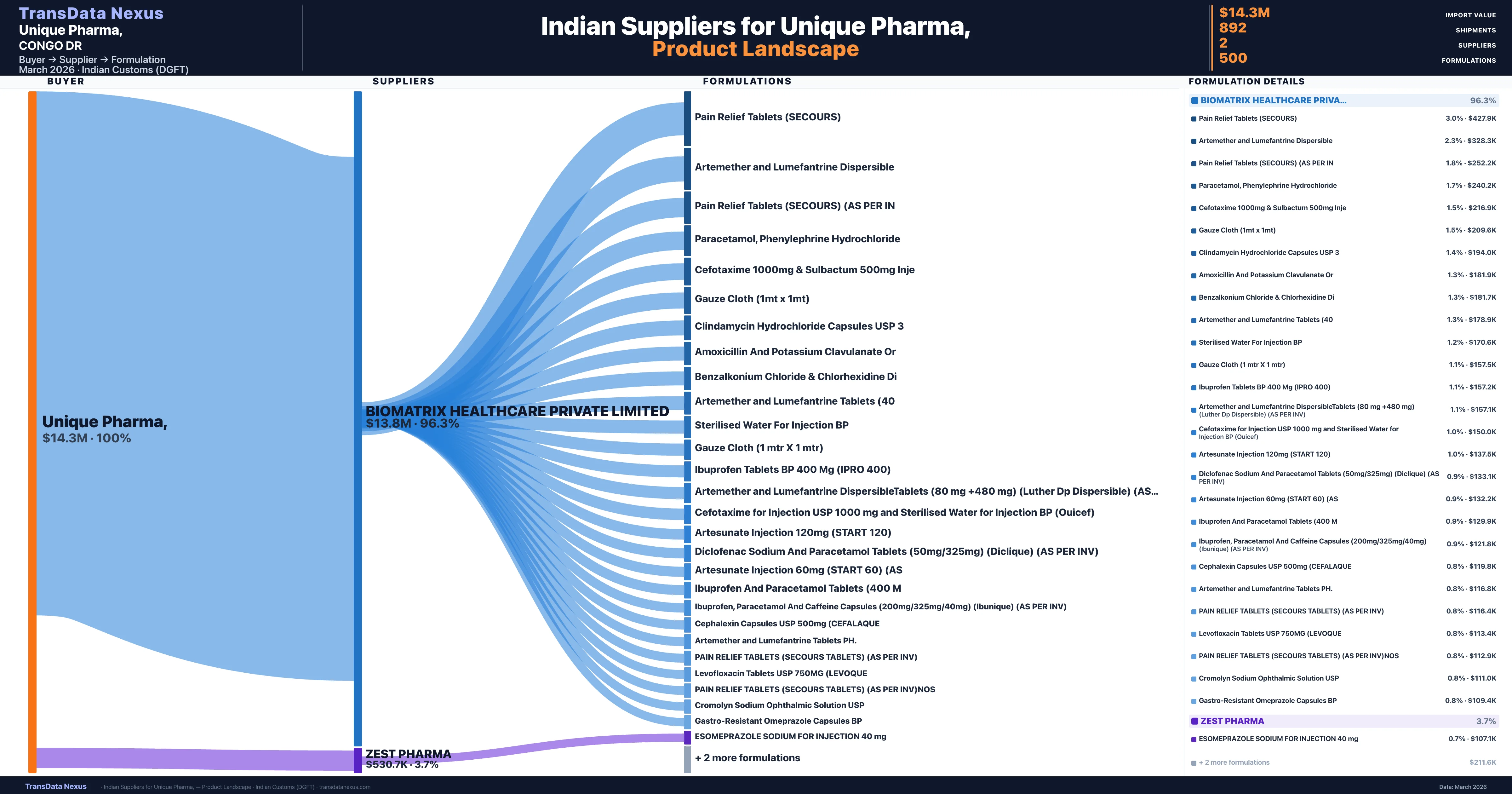
Unique Pharma, is a pharmaceutical importer based in Congo Dr with a total trade value of $9.2M across 10 products in 7 therapeutic categories. Based on 304 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Unique Pharma, sources from 2 verified Indian suppliers, with Biomatrix Healthcare Private Limited accounting for 96.3% of imports.
Unique Pharma, — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Unique Pharma,?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Biomatrix Healthcare Private Limited | $13.8M | 863 | 96.3% |
| Zest Pharma | $530.7K | 29 | 3.7% |
Unique Pharma, sources from 2 verified Indian suppliers across 500 distinct formulations. The sourcing is highly concentrated — Biomatrix Healthcare Private Limited accounts for 96.3% of total imports, indicating a strategic single-source relationship.
What Formulations Does Unique Pharma, Import?
| Formulation | Value | Ships |
|---|---|---|
| Pain relief tablets (secours) | $427.9K | 14 |
| Artemether and lumefantrine | $328.3K | 14 |
| Pain relief tablets (secours) (as per | $252.2K | 9 |
| Paracetamol, phenylephrine | $240.2K | 16 |
| Cefotaxime 1000MG & sulbactum 500MG | $216.9K | 7 |
| Gauze cloth (1mt x 1mt) | $209.6K | 11 |
| Clindamycin hydrochloride capsules USP | $194.0K | 6 |
| Amoxicillin and potassium clavulanate | $181.9K | 4 |
| Benzalkonium chloride & chlorhexidine | $181.7K | 6 |
| Artemether and lumefantrine tablets | $178.9K | 5 |
| Sterilised water for injection | $170.6K | 8 |
| Gauze cloth (1 mtr x 1 mtr) | $157.5K | 6 |
| Ibuprofen tablets BP 400 MG (ipro 400) | $157.2K | 5 |
| Artemether and lumefantrine dispersibletablets (80 MG +480 MG) (luther dp dispersible) (as per inv) | $157.1K | 4 |
| Cefotaxime for injection USP 1000 MG and sterilised water for injection BP (ouicef) | $150.0K | 3 |
Unique Pharma, imports 500 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Unique Pharma, Import?
Unique Pharma, Therapeutic Categories — 7 Specializations
Unique Pharma, imports across 7 therapeutic categories, with Antimalarial & Antiparasitic (46.0%), Antibiotics (27.0%), Nutritional Supplements (10.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 91% of total imports.
Antimalarial & Antiparasitic
1 products · 46.0% · $4.3M
Antibiotics
3 products · 27.0% · $2.5M
Nutritional Supplements
2 products · 10.8% · $1.0M
Respiratory & OTC
1 products · 10.3% · $950.0K
Cardiovascular
1 products · 2.5% · $232.1K
Advanced Antibiotics
1 products · 2.1% · $192.9K
Combination Drugs
1 products · 1.2% · $112.5K
Import Portfolio — Top 10 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Artesunate | Antimalarial & Antiparasitic | $4.3M | 177 | 1.6% | 5 |
| 2 | Clindamycin | Antibiotics | $1.3M | 26 | 1.4% | 15 |
| 3 | Amoxicillin | Antibiotics | $1.1M | 21 | 0.2% | 14 |
| 4 | Phenylephrine | Respiratory & OTC | $950.0K | 19 | 1.1% | 12 |
| 5 | Potassium | Nutritional Supplements | $850.0K | 17 | 0.1% | 18 |
| 6 | Captopril | Cardiovascular | $232.1K | 5 | 1.9% | 5 |
| 7 | Ornidazole | Advanced Antibiotics | $192.9K | 17 | 1.1% | 11 |
| 8 | Protein | Nutritional Supplements | $153.0K | 13 | 1.0% | 12 |
| 9 | Cephalexin | Antibiotics | $150.0K | 3 | 0.0% | 9 |
| 10 | Ibuprofen Paracetamol | Combination Drugs | $112.5K | 6 | 5.8% | 2 |
Unique Pharma, imports 10 pharmaceutical products across 7 categories into Congo Dr totaling $9.2M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Unique Pharma,.
Request DemoUnique Pharma, — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Unique Pharma is a prominent pharmaceutical importer and distributor based in the Democratic Republic of Congo (DRC). Established with the mission to provide accessible and high-quality healthcare solutions across Africa, the company has expanded its presence to major African countries, earning the trust of regulators, healthcare professionals, and institutions.
Headquartered in Kinshasa, DRC, Unique Pharma operates with a robust distribution network, supplying a diverse range of pharmaceutical products, including branded formulations and generic medicines. The company's product portfolio spans various therapeutic areas such as gynecology, pediatrics, anti-infectives, anti-malarials, cardio-diabetics, and surgical products.
Unique Pharma's commitment to quality is evident in its stringent testing protocols and zero tolerance for process deviations. This dedication has led to the development of leading pharmaceutical brands like Sudrox, Hifer, and Triokit, which are widely accepted in the African market.
2Distribution Network
Unique Pharma's distribution network is strategically positioned to serve the vast expanse of the DRC. The company maintains warehouses in key cities, including Kinshasa, Lubumbashi, and Goma, ensuring efficient coverage of the country's diverse regions.
The logistics capabilities of Unique Pharma are designed to handle the complexities of the DRC's infrastructure, facilitating timely delivery of pharmaceutical products to hospitals, pharmacies, and healthcare centers. While the company's primary focus is on the DRC, its established network and reputation have enabled it to extend its reach to other African countries, contributing to the broader healthcare landscape.
3Industry Role
In the pharmaceutical supply chain of the DRC, Unique Pharma functions as a primary wholesaler and distributor. The company imports a wide array of finished pharmaceutical formulations from international suppliers, particularly from India, and supplies these products to local hospitals, pharmacies, and healthcare institutions.
Unique Pharma's role is pivotal in bridging the gap between international pharmaceutical manufacturers and the Congolese healthcare system, ensuring the availability of essential medicines across the country. The company's extensive distribution network and commitment to quality have solidified its position as a key player in the DRC's pharmaceutical sector.
Supplier Relationship Intelligence — Unique Pharma,
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Unique Pharma's sourcing strategy exhibits a high degree of concentration, with a significant majority of its pharmaceutical imports originating from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations totaling $9.2 million USD across 304 shipments, with 90.9% of this value attributed to the top five products.
The primary supplier, BIOMATRIX HEALTHCARE PRIVATE LIMITED, accounted for 96.3% of the total import value, indicating a strong reliance on this single source. This concentration suggests a strategic choice to maintain consistent product quality and supply chain efficiency. However, such dependency also poses potential risks, including supply disruptions due to geopolitical issues, regulatory changes, or operational challenges faced by the supplier.
The stability of the relationship with BIOMATRIX HEALTHCARE PRIVATE LIMITED appears robust, given the volume and frequency of shipments. Nonetheless, Unique Pharma may consider diversifying its supplier base to mitigate risks associated with over-reliance on a single source.
2Supply Chain Resilience
Unique Pharma's supply chain resilience is closely tied to its sourcing strategy from India, particularly its dependence on BIOMATRIX HEALTHCARE PRIVATE LIMITED. While the company imports a diverse range of 500 unique pharmaceutical formulations, the heavy reliance on a single supplier for the majority of its imports could pose challenges in the event of supply chain disruptions.
The company has not publicly disclosed information regarding backup suppliers or alternative sourcing strategies. This lack of diversification may expose Unique Pharma to risks such as supply interruptions, price fluctuations, and potential regulatory changes affecting its primary supplier.
To enhance supply chain resilience, Unique Pharma might consider establishing relationships with additional suppliers, both within India and from other countries, to ensure a more robust and flexible supply chain capable of adapting to unforeseen challenges.
3Strategic Implications
Unique Pharma's concentrated sourcing strategy from India, particularly its reliance on BIOMATRIX HEALTHCARE PRIVATE LIMITED, has significant strategic implications. This approach has enabled the company to maintain consistent product quality and supply chain efficiency, reinforcing its competitive position in the DRC's pharmaceutical market.
For Indian exporters, the established relationship with Unique Pharma presents an opportunity to strengthen and expand their market presence in the DRC. However, the concentration of imports from a single supplier indicates that Unique Pharma may be open to exploring additional partnerships to diversify its sourcing and mitigate potential risks.
Indian exporters seeking to become alternative suppliers should focus on demonstrating product quality, reliability, and compliance with international standards to meet Unique Pharma's stringent requirements.
Importing Pharmaceuticals into Congo Dr — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Congo Dr
1Regulatory Authority & Framework
The Congolese Pharmaceutical Regulatory Authority (ACOREP) is responsible for overseeing the importation, registration, and distribution of pharmaceutical products in the DRC. ACOREP ensures that all imported medicines meet the country's health and safety standards, safeguarding public health.
Key legislation governing pharmaceutical imports includes the Pharmaceutical Law, which outlines the requirements for drug registration, import licensing, and quality control. The law mandates that all imported medicines undergo a pre-shipment inspection and be registered with ACOREP before they can be marketed in the DRC.
The marketing authorization pathway for Indian generics involves obtaining a Certificate of Pharmaceutical Product (CPP) from the exporting country's regulatory authority, followed by registration with ACOREP. This process ensures that imported generics are of acceptable quality and efficacy, aligning with the DRC's health standards.
2Import Licensing & GMP
Import licensing in the DRC requires importers to secure an import license from an authorized commercial bank. Additionally, shipments valued over a specified amount must undergo a pre-shipment inspection by Bureau Veritas to verify compliance with Congolese regulations.
While Good Manufacturing Practice (GMP) certification is officially required for imported medicines, enforcement is often inconsistent. A Certificate of Pharmaceutical Product (CPP) is a critical document in the import process, serving as evidence of the product's quality and compliance with international standards.
Importers must also obtain wholesale distribution authorization from ACOREP, ensuring that they are qualified to distribute pharmaceutical products within the DRC. This authorization is essential for maintaining the integrity and safety of the pharmaceutical supply chain.
3Quality & Labeling
Imported pharmaceutical products are subject to batch testing and stability requirements to ensure their safety and efficacy. Initial screening is conducted locally using methods like the GPHF Minilab, with more comprehensive analysis performed at WHO-prequalified laboratories abroad.
Labeling requirements mandate that all documentation, including product labels and accompanying materials, be in French, the official language of the DRC. This ensures that healthcare professionals and patients can accurately interpret product information. Additionally, serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, the DRC has implemented several policy changes affecting pharmaceutical imports. Notably, the introduction of stricter import licensing requirements and enhanced pre-shipment inspection protocols by Bureau Veritas have been enforced to bolster the quality control of imported medicines.
Additionally, there has been an increased emphasis on GMP certification for imported products, with ACOREP intensifying its oversight to ensure compliance with international manufacturing standards. These regulatory changes aim to improve the safety and efficacy of pharmaceutical products available in the DRC market.
Unique Pharma, — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Unique Pharma's product strategy focuses on therapeutic areas with high demand in the DRC, including anti-malarial and anti-infective treatments, as well as cardio-diabetic and surgical products. The company's import data indicates a significant emphasis on anti-malarial and anti-infective medications, reflecting the prevalence of these health concerns in the region.
The market demand for these products is driven by the high incidence of infectious diseases such as malaria and the growing burden of non-communicable diseases like diabetes and cardiovascular conditions. Unique Pharma's strategic focus on these areas aligns with the healthcare needs of the Congolese population, positioning the company as a key supplier of essential medicines.
2Sourcing Profile
Unique Pharma's sourcing strategy is predominantly centered on importing finished pharmaceutical formulations from India, particularly from suppliers like BIOMATRIX HEALTHCARE PRIVATE LIMITED.
Frequently Asked Questions — Unique Pharma,
What products does Unique Pharma, import from India?
Unique Pharma, imports 10 pharmaceutical products across 7 categories. Top imports: Artesunate ($4.3M), Clindamycin ($1.3M), Amoxicillin ($1.1M), Phenylephrine ($950.0K), Potassium ($850.0K).
Who supplies pharmaceuticals to Unique Pharma, from India?
Unique Pharma, sources from 2 verified Indian suppliers. The primary supplier is Biomatrix Healthcare Private Limited (96.3% of imports, $13.8M).
What is Unique Pharma,'s total pharmaceutical import value?
Unique Pharma,'s total pharmaceutical import value from India is $9.2M, based on 304 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Unique Pharma, focus on?
Unique Pharma, imports across 7 categories. The largest: Antimalarial & Antiparasitic (46.0%), Antibiotics (27.0%), Nutritional Supplements (10.8%).
Get Full Unique Pharma, Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Unique Pharma, identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Unique Pharma,'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 304 individual customs records matching Unique Pharma,.
- 5.Supplier Verification: Unique Pharma, sources from 2 verified Indian suppliers across 500 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
10 Products Tracked
7 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.