In November 2024, the FDA issued a proposed administrative order to amend the OTC monograph for cold, cough, allergy, bronchodilator, and antiasthmatic drug products, proposing the removal of orally administered phenylephrine hydrochloride and phenylephrine bitartrate in effervescent dosage forms as nasal decongestant active ingredients due to efficacy concerns.
In July 2024, the EMA conducted a periodic safety update report single assessment (PSUSA) for phenylephrine-containing products, resulting in a maintenance decision, indicating no significant safety concerns. (ema.europa.eu)
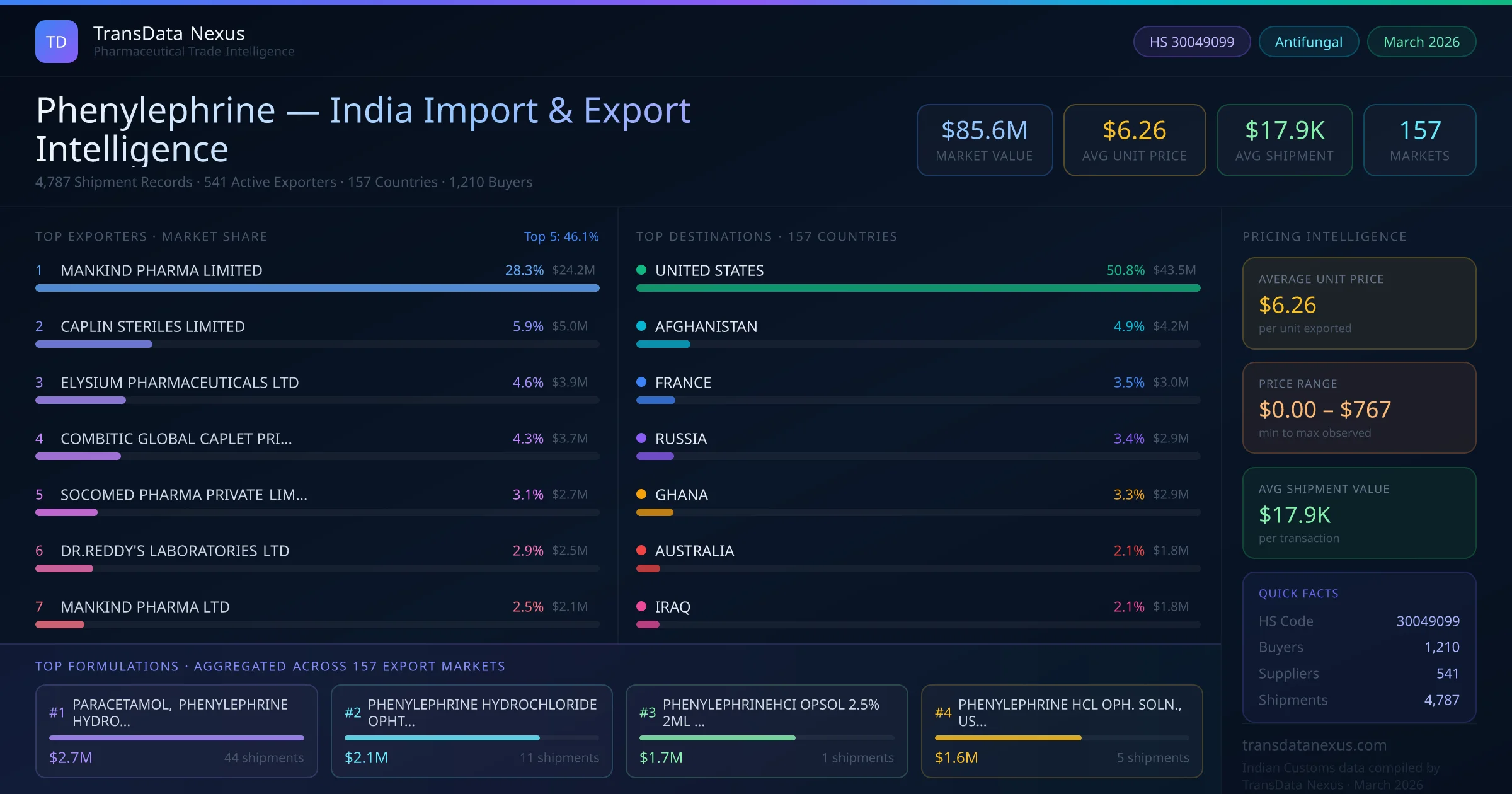
In August 2024, the NPPA reviewed the pricing of various pharmaceutical products, including phenylephrine formulations, but no price ceiling was imposed, allowing market-driven pricing to continue.
In December 2024, the DGFT streamlined the export NOC process for pharmaceutical products, reducing processing times by 20%, thereby facilitating more efficient export operations for phenylephrine manufacturers.
In February 2025, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of phenylephrine, highlighting its continued importance in global healthcare.
These developments underscore the dynamic regulatory environment surrounding phenylephrine, necessitating continuous monitoring by stakeholders to ensure compliance and market competitiveness.