Loading...
Loading...
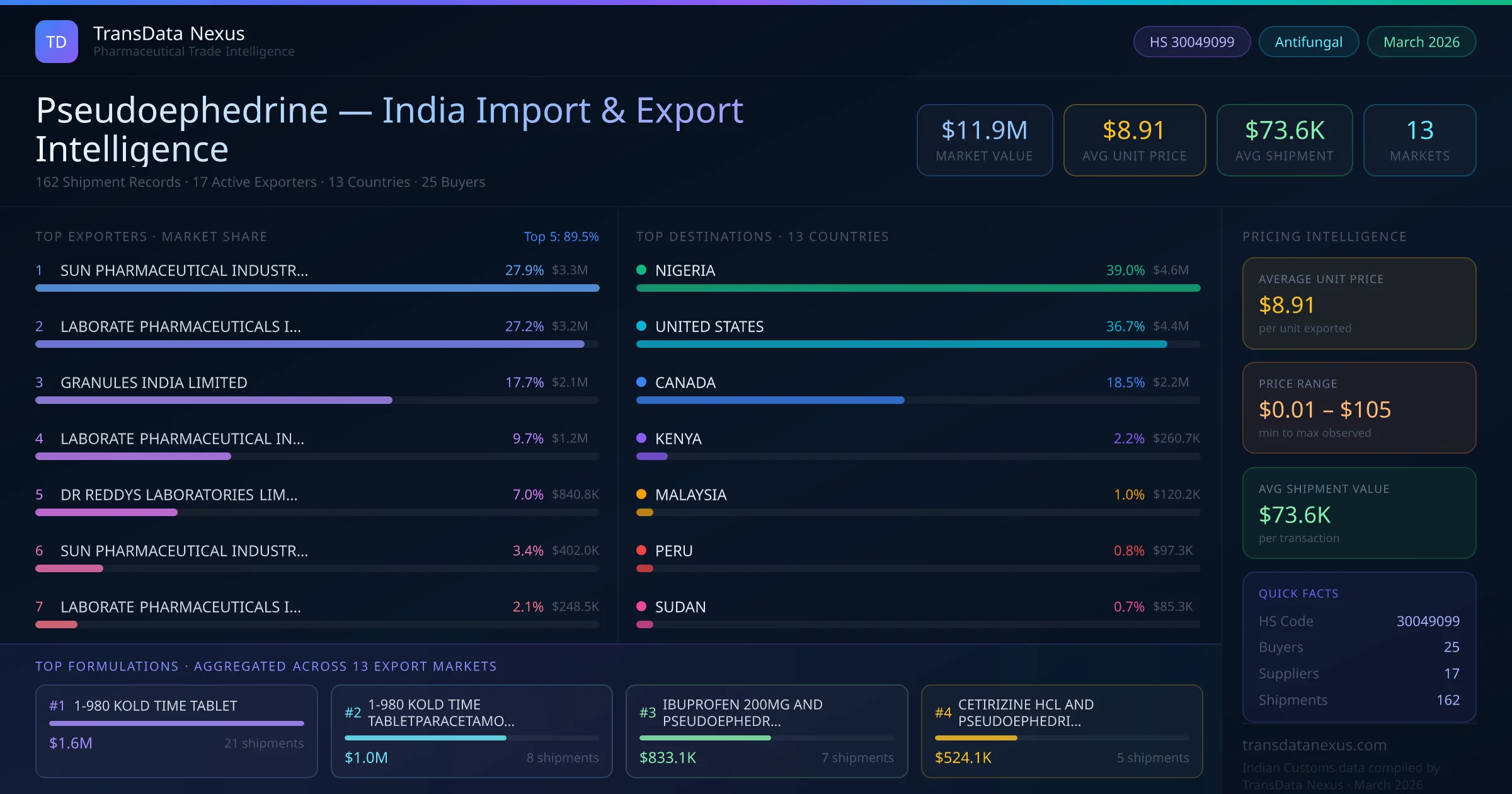
Between 2022 and 2026, India exported $11.9M worth of pseudoephedrine across 162 verified shipments to 13 countries — covering 7% of world markets in the Respiratory & OTC segment. The largest destination is NIGERIA (39.0%). SUN PHARMACEUTICAL INDUSTRIES LIMITED leads with a 27.9% share. All figures are drawn from Indian Customs (DGFT) shipping bill records spanning four years of trade activity.

17 active exporters · Ranked by export value
| # | Supplier Name | Export Value (USD) | Market Share |
|---|---|---|---|
| 1 | SUN PHARMACEUTICAL INDUSTRIES LIMITED | $3.3M | 27.9% |
| 2 | LABORATE PHARMACEUTICALS INDIA LTD | $3.2M | 27.2% |
| 3 | GRANULES INDIA LIMITED | $2.1M | 17.7% |
| 4 | LABORATE PHARMACEUTICAL INDIA LIMITED | $1.2M | 9.7% |
| 5 | DR REDDYS LABORATORIES LIMITED | $840.8K | 7.0% |
| 6 | SUN PHARMACEUTICAL INDUSTRIES LTD | $402.0K | 3.4% |
| 7 | HIGLANCE LABORATORIES PRIVATE LIMITED | $97.3K | 0.8% |
| 8 | APL HEALTHCARE LIMITED | $93.2K | 0.8% |
| 9 | IPCA LABORATORIES LIMITED | $87.8K | 0.7% |
| 10 | ALPS LIFE SCIENCES PRIVATE LIMITED | $85.3K | 0.7% |
Based on customs records from 2022 through early 2026, India's pseudoephedrine export market is led by SUN PHARMACEUTICAL INDUSTRIES LIMITED, which holds a 27.9% share of all pseudoephedrine exports — the largest of any single manufacturer over this period. The top 5 suppliers together account for 89.5% of total export value, reflecting a concentrated supplier landscape among the 17 active exporters. Each supplier handles an average of 10 shipments, indicating selective, specialised distribution patterns.
13 destination markets · Ranked by import value
| # | Country | Import Value (USD) | Market Share |
|---|---|---|---|
| 1 | NIGERIA | $4.6M | 39.0% |
| 2 | UNITED STATES | $4.4M | 36.7% |
| 3 | CANADA | $2.2M | 18.5% |
| 4 | KENYA |
Product-specific regulatory status across FDA, EMA, WHO, and CDSCO · As of March 2026
In the United States, pseudoephedrine is regulated by the Food and Drug Administration (FDA) and is available both over-the-counter (OTC) and by prescription, depending on the formulation. As of February 2026, the FDA approved Abbreviated New Drug Application (ANDA) 218854 for Pseudoephedrine Hydrochloride Extended-Release Tablets USP, 240 mg (OTC), submitted by Aurobindo Pharma USA, Inc., a subsidiary of Aurobindo Pharma Limited. This approval underscores the FDA's commitment to ensuring the availability of safe and effective generic medications.
The Combat Methamphetamine Epidemic Act of 2005 imposes strict regulations on the sale of pseudoephedrine-containing products in the U.S., including purchase limits and identification requirements, to prevent their misuse in methamphetamine production. These regulations necessitate that Indian exporters comply with stringent documentation and quality standards to access the U.S. market. The presence of 17 active Indian exporters indicates a robust engagement with these regulatory requirements, reflecting the industry's adaptability and commitment to compliance.
In the European Union (EU) and the United Kingdom (UK), pseudoephedrine is subject to comprehensive regulatory oversight. The European Medicines Agency (EMA) has approved products like Aerinaze, which combines desloratadine and pseudoephedrine, for the symptomatic treatment of seasonal allergic rhinitis with nasal congestion. This approval, granted in July 2007, highlights the established therapeutic use of pseudoephedrine in the EU. (ema.europa.eu)
| $260.7K |
| 2.2% |
| 5 | MALAYSIA | $120.2K | 1.0% |
| 6 | PERU | $97.3K | 0.8% |
| 7 | SUDAN | $85.3K | 0.7% |
| 8 | VENEZUELA | $60.6K | 0.5% |
| 9 | UNITED KINGDOM | $40.4K | 0.3% |
| 10 | ZAMBIA | $20.0K | 0.2% |
NIGERIA is India's largest pseudoephedrine export destination, absorbing 39.0% of total exports worth $4.6M. The top 5 importing countries — NIGERIA, UNITED STATES, CANADA, KENYA, MALAYSIA — together account for 97.4% of India's total pseudoephedrine export value. The remaining 8 destination countries collectively receive the other 2.6%, indicating a focused distribution strategy targeting key markets.
All products in Respiratory & OTC category • Over-the-counter respiratory and cold medications
In February 2023, the EMA's Pharmacovigilance Risk Assessment Committee (PRAC) initiated a safety review of pseudoephedrine-containing medicines due to concerns about rare cases of posterior reversible encephalopathy syndrome (PRES) and reversible cerebral vasoconstriction syndrome (RCVS). This ongoing review may lead to changes in marketing authorizations, emphasizing the need for exporters to stay informed about evolving regulatory landscapes. (ema.europa.eu)
Compliance with EU Good Manufacturing Practice (GMP) standards is mandatory for exporting pharmaceuticals to the EU and UK. These standards ensure product quality and safety, requiring Indian manufacturers to maintain rigorous quality control systems and undergo regular inspections.
As of the 23rd edition of the WHO Model List of Essential Medicines, published in July 2023, pseudoephedrine is not included. This exclusion indicates that while pseudoephedrine is widely used, it is not considered essential for basic health care needs by WHO standards. (who.int)
Pseudoephedrine formulations must comply with international pharmacopoeia standards, including those set by the United States Pharmacopeia (USP), British Pharmacopoeia (BP), European Pharmacopoeia (EP), and Indian Pharmacopoeia (IP). Adherence to these standards ensures the quality, efficacy, and safety of pharmaceutical products in global markets.
In India, pseudoephedrine is classified as a Schedule H drug under the Drugs and Cosmetics Act, requiring a prescription for sale. This classification aims to control its distribution and prevent misuse. The Drug Price Control Order (DPCO), enforced by the National Pharmaceutical Pricing Authority (NPPA), regulates the pricing of essential medicines; however, pseudoephedrine is not currently listed under price control, allowing market-driven pricing.
For export purposes, the Directorate General of Foreign Trade (DGFT) mandates a No Objection Certificate (NOC) for pseudoephedrine shipments, ensuring that exports comply with national regulations and international obligations.
Pseudoephedrine is a well-established pharmaceutical ingredient with no active patents restricting its manufacture or sale. This lack of patent protection has led to a competitive market with multiple generic manufacturers, both in India and globally, contributing to its widespread availability and affordability.
In April 2024, New Zealand's Parliament passed the Misuse of Drugs (Pseudoephedrine) Amendment Act 2024, reclassifying pseudoephedrine as a Class C controlled drug. This legislative change allows cold and flu medicines containing pseudoephedrine to be sold without prescriptions in pharmacies, reversing previous restrictions imposed in 2011.
In February 2023, the EMA's PRAC initiated a safety review of pseudoephedrine-containing medicines due to concerns about rare cases of PRES and RCVS. This review underscores the importance of ongoing pharmacovigilance and may lead to regulatory changes affecting the availability and use of these medicines in the EU. (ema.europa.eu)
These developments highlight the dynamic nature of the regulatory environment surrounding pseudoephedrine, necessitating continuous monitoring and compliance by exporters to maintain market access and ensure public safety.
API sourcing, concentration risk, storage requirements, and current alerts
India's pharmaceutical industry, including the production of pseudoephedrine, heavily relies on Key Starting Materials (KSMs) imported from China. This dependency is significant, with approximately 60–70% of KSMs for various APIs sourced from China. The concentration of KSM production in China poses a strategic risk, as any disruption in Chinese supply chains can directly impact India's API manufacturing capabilities.
Recent events have highlighted this vulnerability. In September 2025, China implemented aggressive pricing strategies, reducing the prices of 41 APIs and KSMs by 40–50%. This move aimed to undercut Indian API manufacturers, making domestic production less competitive and increasing reliance on Chinese imports. Such market dynamics underscore the fragility of India's API supply chain, particularly for critical components like pseudoephedrine.
Our proprietary trade data indicates a high supplier concentration in India's pseudoephedrine exports. The top five exporters account for 89.5% of the total export value, with SUN PHARMACEUTICAL INDUSTRIES LIMITED alone contributing 27.9%. This concentration poses a significant single-source risk; any operational or regulatory issues affecting these key suppliers could disrupt the entire supply chain.
To mitigate such risks, the Indian government introduced the Production Linked Incentive (PLI) scheme in November 2024, aiming to boost domestic API and KSM production. This initiative led to the inauguration of two greenfield plants producing essential molecules like Penicillin G and Clavulanic Acid. While these efforts are steps toward reducing import dependence, the impact on pseudoephedrine production remains to be seen.
Geopolitical tensions have further exacerbated supply chain vulnerabilities. In March 2026, the Strait of Hormuz crisis led to significant disruptions in global shipping routes, affecting approximately 20% of the world's daily oil supply. This crisis caused a sharp decline in maritime transit, with tanker traffic dropping by about 70% and over 150 ships anchoring outside the strait to avoid risks. Such disruptions have cascading effects on global supply chains, including the transportation of pharmaceutical ingredients.
Additionally, the U.S. Food and Drug Administration (FDA) has issued warning letters to API manufacturers in China and India for significant deviations from current Good Manufacturing Practice (cGMP). In February 2025, Indian API manufacturer Global Calcium received such a warning, highlighting ongoing quality concerns and the potential for supply disruptions due to regulatory actions.
RISK_LEVEL: HIGH
Shipment-level records, verified supplier contacts, buyer histories, and pricing analytics for all 162 transactions across 13 markets.
Data-backed answers sourced from Indian Customs shipping bill records
The leading pseudoephedrine exporters from India are SUN PHARMACEUTICAL INDUSTRIES LIMITED, LABORATE PHARMACEUTICALS INDIA LTD, GRANULES INDIA LIMITED, and 7 others. SUN PHARMACEUTICAL INDUSTRIES LIMITED leads with 27.9% market share ($3.3M). The top 5 suppliers together control 89.5% of total export value.
The total export value of pseudoephedrine from India is $11.9M, recorded across 162 shipments from 17 active exporters to 13 countries. The average shipment value is $73.6K.
India exports pseudoephedrine to 13 countries. The top importing countries are NIGERIA (39.0%), UNITED STATES (36.7%), CANADA (18.5%), KENYA (2.2%), MALAYSIA (1.0%), which together account for 97.4% of total export value.
The primary HS code for pseudoephedrine exports from India is 30049099. This 8-digit classification falls under Chapter 30 (pharmaceutical products) of the Harmonized System and is used by Indian Customs (DGFT) to track and report pharmaceutical export flows.
The average unit price for pseudoephedrine exports from India is $8.91 per unit, with prices ranging from $0.01 to $105.29 depending on formulation and order volume.
The primary export ports for pseudoephedrine from India are PATLI ICD (17.3%), SAHAR AIR (13.0%), SAHAR AIR CARGO ACC (INBOM4) (8.6%), HYDERABAD ACC (INHYD4) (8.0%). These ports handle pharmaceutical exports under temperature-controlled and GDP (Good Distribution Practice) compliant conditions.
India is a leading pseudoephedrine exporter due to its large base of 17 manufacturers — many WHO-GMP and US FDA approved — combined with significantly lower production costs, well-developed API supply chains, and strong government support through Pharmexcil. India's pseudoephedrine exports reach 13 countries (7% of world markets), making it a dominant global supplier of respiratory & otc compounds.
Indian pseudoephedrine exporters typically require WHO-GMP certification for regulated markets, US FDA approval for the United States, and EU GMP certification for European markets. Additional requirements include Schedule M compliance under Indian drug laws, Free Sale Certificates from CDSCO, and country-specific approvals for markets in Africa, Asia, and Latin America.
25 buyers import pseudoephedrine from India across 13 countries. The repeat buyer rate is 76.0%, indicating strong ongoing trade relationships.
SUN PHARMACEUTICAL INDUSTRIES LIMITED is the leading pseudoephedrine exporter from India with a market share of 27.9% and export value of $3.3M across 45 shipments. The top 5 suppliers together hold 89.5% of the market.
Data on this page is sourced from Indian Customs (DGFT) shipping bill records. Verify regulatory status with the official agencies above.
Pharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormareAll trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
162 Verified Shipments
17 exporters to 13 countries
Expert-Reviewed
By pharmaceutical trade specialists