In February 2026, the UK's MHRA issued a Class 3 medicines recall for specific batches of ibuprofen 200 mg tablets due to findings of foil perforations in some blisters. This precautionary measure underscores the importance of stringent quality control in pharmaceutical manufacturing. (gov.uk)
In May 2024, the EMA recommended the refusal of authorisation for Ibuprofen NVT 400 mg soft capsules, concluding that the benefits did not outweigh the risks. This decision highlights the critical role of demonstrating bioequivalence and safety in the approval process for generic medicines. (ema.europa.eu)
In February 2024, the EMA granted marketing authorisation for Ibuprofen Gen.Orph, a generic version of ibuprofen, indicating continued opportunities for generic medicines in the European market. (ema.europa.eu)
In July 2020, the EMA recommended the authorisation of Ibuprofen Kabi 400 mg solution for infusion, reflecting the agency's support for diverse formulations of ibuprofen to meet varying patient needs. (ema.europa.eu)
In May 2017, the EMA's CHMP concluded that the benefits of Paracetamol/Ibuprofen 500 mg/150 mg film-coated tablets outweigh the risks, leading to marketing authorisation in multiple EU Member States, including the United Kingdom. (ema.europa.eu)
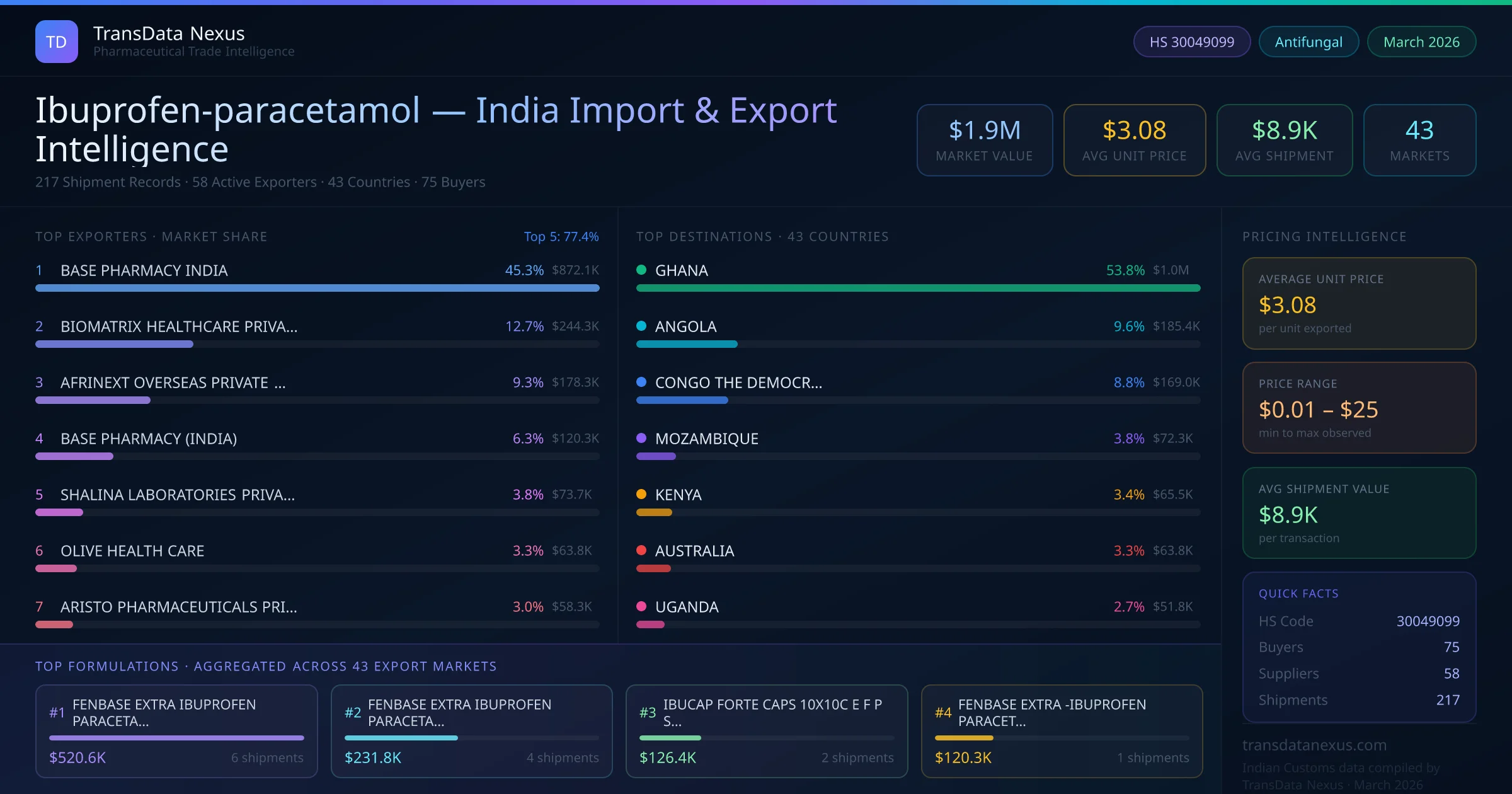
These developments underscore the dynamic nature of the pharmaceutical regulatory landscape and the importance of staying informed about regulatory decisions and quality standards to ensure successful market access and compliance.