In June 2025, the Indian Ministry of Health and Family Welfare revised the export NOC requirements, streamlining the process for pharmaceutical products, including Ornidazole, thereby facilitating faster export approvals.
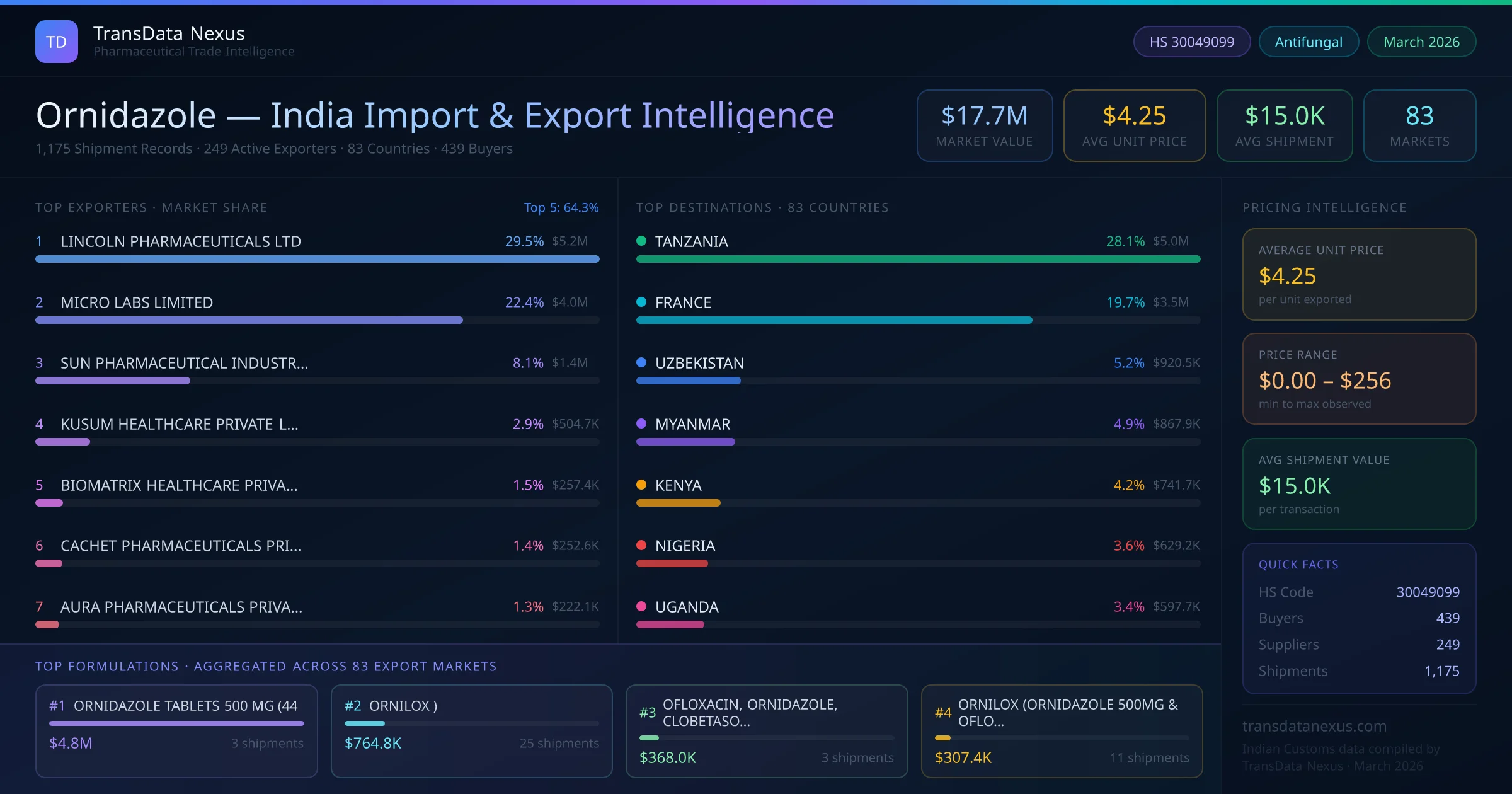
In September 2025, the European Medicines Agency (EMA) updated its GMP guidelines, emphasizing stricter compliance measures for active pharmaceutical ingredients (APIs). Indian manufacturers exporting to the EU, particularly to France, which accounts for 19.7% of Ornidazole exports, must align with these enhanced standards to maintain market access.
In December 2025, the World Health Organization (WHO) released a new edition of the Model List of Essential Medicines. Ornidazole was not included, indicating that while it remains a valuable therapeutic agent, it does not meet the current criteria for essential medicines designation.
In February 2026, the Indian Pharmacopoeia Commission (IPC) updated the monograph for Ornidazole, incorporating revised analytical methods to ensure better quality control. Manufacturers are advised to adopt these methods to comply with national standards.
In March 2026, the U.S. Food and Drug Administration (FDA) issued a draft guidance on the development of nitroimidazole derivatives, including Ornidazole, outlining the necessary clinical and non-clinical data requirements for potential approval. This guidance provides a clearer pathway for Indian exporters considering entry into the U.S. market.
These developments underscore the dynamic nature of the pharmaceutical regulatory landscape and the importance of continuous monitoring to ensure compliance and strategic market positioning.