Natco Pharma Limited
Indian Pharmaceutical Exporter · Immunosuppressants Specialist · $36.0M Total Trade · DGFT Verified
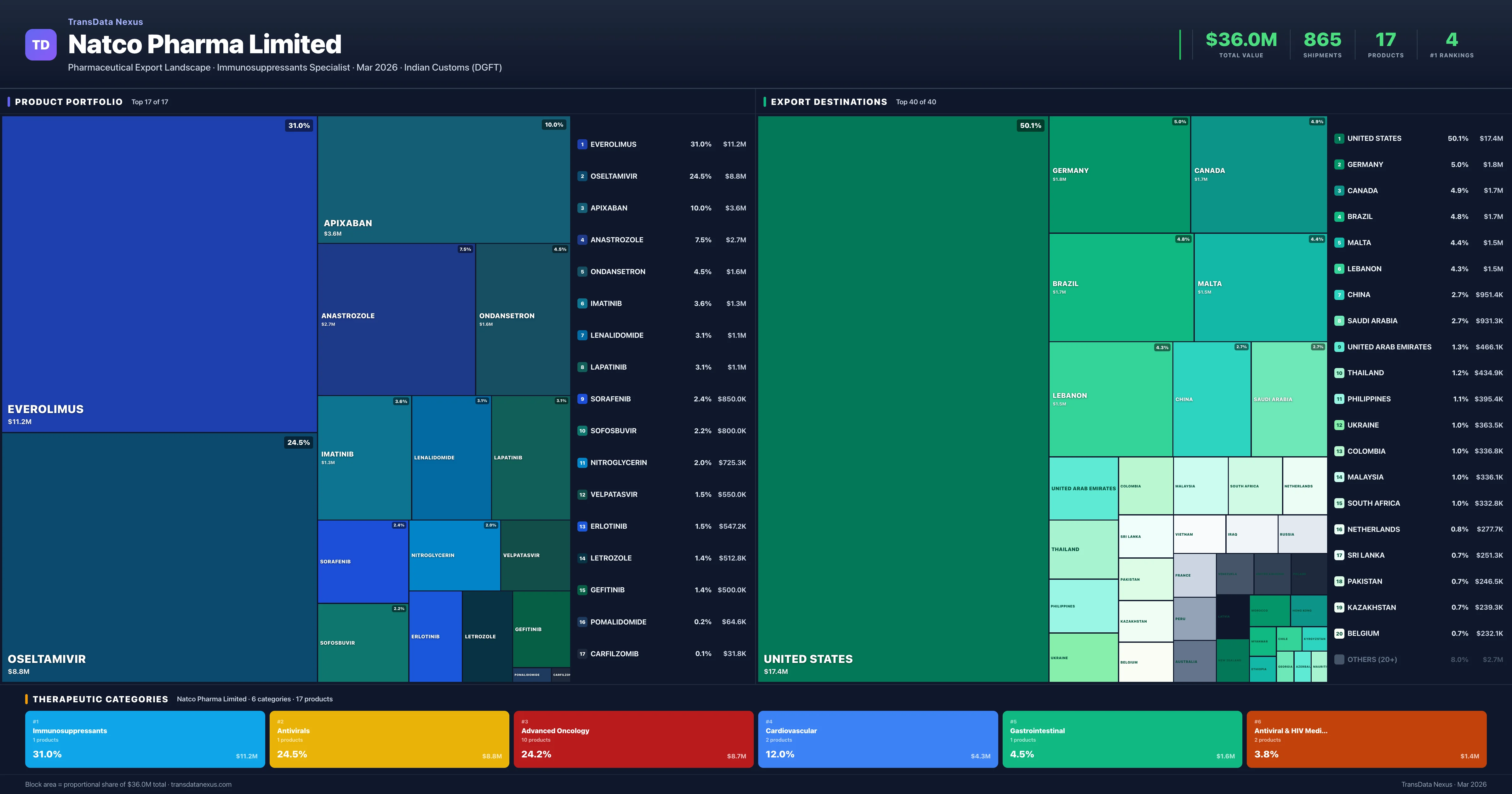
Natco Pharma Limited is an Indian pharmaceutical exporter with a total trade value of $36.0M across 17 products in 6 therapeutic categories. Based on 865 verified export shipments from Indian Customs (DGFT) records, Natco Pharma Limited is the #1 Indian exporter in 4 products including Everolimus, Oseltamivir, Lapatinib. Top exports include Everolimus ($11.2M), Oseltamivir ($8.8M), Apixaban ($3.6M).
Natco Pharma Limited — Export Portfolio & Destination Treemap
Who is Natco Pharma Limited? — Company Overview & Market Position
Natco Pharma Limited, established in 1981, is a vertically integrated pharmaceutical company headquartered in Hyderabad, India. The company specializes in the research, development, manufacturing, and marketing of finished dosage formulations (FDFs) and active pharmaceutical ingredients (APIs). Its product portfolio spans various therapeutic areas, including oncology, cardiology, diabetology, and gastroenterology. Natco Pharma is recognized for its focus on niche therapeutic segments and complex products, positioning itself as a leader in branded oncology medicines in India and among the top producers of hepatitis C drugs.
As of March 2025, Natco Pharma reported a revenue of $529.4 million, reflecting a 10.8% increase from the previous fiscal year. The company employs approximately 4,199 individuals across its operations. Natco Pharma's stock is listed on the National Stock Exchange of India under the ticker symbol NATCOPHARM.
What Does Natco Pharma Limited Export? — Product Portfolio Analysis
Natco Pharma Limited Therapeutic Categories — 6 Specializations
Natco Pharma Limited operates across 6 therapeutic categories, with Immunosuppressants (31.0%), Antivirals (24.5%), Advanced Oncology (24.2%) representing the largest segments by export value. The portfolio is concentrated — the top 5 products account for 78% of total exports.
Immunosuppressants
1 products · 31.0% · $11.2M
Antivirals
1 products · 24.5% · $8.8M
Advanced Oncology
10 products · 24.2% · $8.7M
Cardiovascular
2 products · 12.0% · $4.3M
Gastrointestinal
1 products · 4.5% · $1.6M
Antiviral & HIV Medications
2 products · 3.8% · $1.4M
Product Portfolio — Top 17 by Export Value
Natco Pharma Limited exports 17 pharmaceutical products across 6 therapeutic categories. Market leader (#1 exporter) in 4 products.
| # | Product | Category | Export Value | Shipments | Market Share | Rank |
|---|---|---|---|---|---|---|
| 1 | Everolimus | Immunosuppressants | $11.2M | 223 | 29.4% | 1 |
| 2 | Oseltamivir | Antivirals | $8.8M | 176 | 7.2% | 1 |
| 3 | Apixaban | Cardiovascular | $3.6M | 72 | 2.0% | 6 |
| 4 | Anastrozole | Advanced Oncology | $2.7M | 68 | 2.4% | 5 |
| 5 | Ondansetron | Gastrointestinal | $1.6M | 90 | 1.3% | 14 |
| 6 | Imatinib | Advanced Oncology | $1.3M | 33 | 4.5% | 5 |
| 7 | Lenalidomide | Advanced Oncology | $1.1M | 31 | 0.1% | 7 |
| 8 | Lapatinib | Advanced Oncology | $1.1M | 22 | 20.5% | 1 |
| 9 | Sorafenib | Advanced Oncology | $850.0K | 17 | 5.1% | 2 |
| 10 | Sofosbuvir | Antiviral & HIV Medications | $800.0K | 16 | 2.0% | 4 |
| 11 | Nitroglycerin | Cardiovascular | $725.3K | 44 | 7.5% | 5 |
| 12 | Velpatasvir | Antiviral & HIV Medications | $550.0K | 11 | 2.2% | 4 |
| 13 | Erlotinib | Advanced Oncology | $547.2K | 11 | 20.4% | 1 |
| 14 | Letrozole | Advanced Oncology | $512.8K | 29 | 1.6% | 7 |
| 15 | Gefitinib | Advanced Oncology | $500.0K | 10 | 7.4% | 2 |
| 16 | Pomalidomide | Advanced Oncology | $64.6K | 5 | 1.5% | 2 |
| 17 | Carfilzomib | Advanced Oncology | $31.8K | 7 | 2.6% | 3 |
Natco Pharma Limited exports 17 pharmaceutical products across 6 therapeutic categories with a total export value of $36.0M. The company is the #1 Indian exporter in 4 products: Everolimus, Oseltamivir, Lapatinib, Erlotinib. The top category is Immunosuppressants (31.0% of portfolio), followed by Antivirals (24.5%), indicating a concentrated portfolio with the top 5 products accounting for 77.5% of total export value.
Key Metrics
Top Categories
Top Export Routes
Related Analysis
Need Detailed Data?
Shipment-level records, pricing & buyer contacts for Natco Pharma Limited.
Request DemoNatco Pharma Limited — Corporate Profile & Information
Headquarters, ownership, manufacturing facilities, and key leadership
1Corporate Overview
Natco Pharma Limited, established in 1981, is a vertically integrated pharmaceutical company headquartered in Hyderabad, India. The company specializes in the research, development, manufacturing, and marketing of finished dosage formulations (FDFs) and active pharmaceutical ingredients (APIs). Its product portfolio spans various therapeutic areas, including oncology, cardiology, diabetology, and gastroenterology. Natco Pharma is recognized for its focus on niche therapeutic segments and complex products, positioning itself as a leader in branded oncology medicines in India and among the top producers of hepatitis C drugs.
As of March 2025, Natco Pharma reported a revenue of $529.4 million, reflecting a 10.8% increase from the previous fiscal year. The company employs approximately 4,199 individuals across its operations. Natco Pharma's stock is listed on the National Stock Exchange of India under the ticker symbol NATCOPHARM.
2Manufacturing Facilities
Natco Pharma operates multiple manufacturing facilities across India, each designed to meet stringent international quality standards. These facilities are equipped with modern research laboratories and capabilities in new drug development. The company's manufacturing plants are strategically located to serve both domestic and international markets, ensuring efficient production and distribution of its pharmaceutical products.
3Key Leadership
The leadership team at Natco Pharma is headed by Vice Chairman and CEO Mr. Rajeev Nannapaneni. Mr. Nannapaneni has been instrumental in steering the company's strategic direction and growth. The Board of Directors includes Chairman and Managing Director Mr. V.C. Nannapaneni, who founded the company in 1981. Other key executives include Dr. Donthineni Linga Rao, President of Technical Affairs and Whole Time Director, and Mr. Chekuri Venkat Ramesh, Company Secretary and Compliance Officer.
Where Does Natco Pharma Limited Export? — Destination Intelligence
Regulated market presence, emerging market penetration, and geographic strategy
1Regulated Markets
Natco Pharma has established a significant presence in regulated markets, including the United States, Europe, the United Kingdom, Australia, and Japan. In the U.S., the company has achieved multiple regulatory milestones, including the launch of generic versions of Tamiflu (oseltamivir) and Pomalidomide, and the first generic version of Revlimid (lenalidomide) in 2022. In Europe, Natco Pharma has received approvals for various products, expanding its footprint in the region. The company's commitment to quality and compliance has facilitated its market access in these regions, enabling it to offer affordable alternatives to branded medications.
2Emerging Markets
Natco Pharma has strategically expanded its reach into emerging markets, including Africa, Latin America, and Southeast Asia. The company's focus on affordable medicines has resonated well in these regions, where access to essential drugs is often limited. By obtaining WHO prequalification for certain products, Natco Pharma has enhanced its credibility and facilitated smoother market entry in these territories. This approach aligns with the company's mission to increase access to high-quality, affordable medicines globally.
3Geographic Strategy
Natco Pharma's geographic strategy emphasizes diversification to mitigate risks associated with overreliance on any single market. The company's operations span across India, the United States, Europe, and emerging markets, reflecting a balanced approach to global expansion. This diversification strategy not only spreads risk but also leverages growth opportunities in various regions, aligning with the company's long-term objectives.
Natco Pharma Limited — Manufacturing & Regulatory Profile
FDA status, WHO-GMP certification, EU GMP compliance, and regulatory track record
1FDA Status
Natco Pharma's manufacturing facility in Kothur, Telangana, India, has been registered with the U.S. Food and Drug Administration (FDA). The facility has undergone inspections, with the most recent occurring from October 9 to 18, 2023. Following this inspection, the FDA issued a warning letter on April 8, 2024, citing significant violations of Current Good Manufacturing Practice (CGMP) regulations. The company has been working to address these issues to maintain its market presence in the U.S.
2WHO & EU GMP
Natco Pharma has obtained Good Manufacturing Practice (GMP) certifications from the World Health Organization (WHO) and the European Union (EU). These certifications affirm the company's commitment to maintaining high-quality manufacturing standards for its pharmaceutical products. The EU GMP certification, in particular, facilitates the company's market access within the European Union, enabling it to offer its products across member states.
3CDSCO & Indian Regulatory
In India, Natco Pharma holds manufacturing licenses from the Central Drugs Standard Control Organisation (CDSCO), the national regulatory authority. The company complies with state drug controller approvals and has obtained export No Objection Certificates (NOCs), facilitating its export activities. These regulatory approvals underscore Natco Pharma's adherence to national standards and its capability to supply pharmaceutical products both domestically and internationally.
4Recent Regulatory Actions
On April 8, 2024, the FDA issued a warning letter to Natco Pharma Limited, referencing inspection findings from October 9 to 18, 2023, at the Kothur facility. The letter highlighted significant violations of CGMP regulations. The company has been addressing these concerns to ensure compliance and maintain its standing in the U.S. market.
Natco Pharma Limited — Competitive Positioning Among Indian Pharma Exporters
Market share comparison, key differentiators, and strategic analysis
1Competitive Landscape
Natco Pharma operates in a competitive landscape with several key players in the pharmaceutical industry. In the oncology segment, the company faces competition from firms like Cipla and Dr. Reddy's Laboratories. In the generics market, it competes with companies such as Lupin Pharmaceuticals and Sun Pharmaceutical Industries. Despite this competition, Natco Pharma has established a strong market presence through its focus on niche therapeutic areas and complex products, enabling it to capture significant market share in various segments.
2Key Differentiators
Natco Pharma's key differentiators include its focus on niche therapeutic areas, such as oncology, cardiology, and diabetology, and its expertise in developing complex generic formulations. The company's vertically integrated operations, encompassing both FDFs and APIs, allow for greater control over product quality and supply chain efficiency. Additionally, Natco Pharma's commitment to affordability and access to medicines distinguishes it in the global pharmaceutical market.
3Strategic Position
Natco Pharma's current strategic direction emphasizes the development and marketing of generic pharmaceuticals, with a particular focus on complex and niche products. The company is also exploring opportunities in the biosimilars and contract development and manufacturing organization (CDMO) sectors to diversify its portfolio and revenue streams. Looking ahead, Natco Pharma aims to expand its global footprint, particularly in emerging markets, while continuing to strengthen its position in established markets through strategic partnerships and product innovations.
Buyer Due Diligence Brief — Evaluating Natco Pharma Limited as a Supplier
Track record assessment, certifications to verify, and pre-order checks
1Supplier Assessment
Natco Pharma has a strong track record as a supplier, with a diverse portfolio of over 140 marketed drugs and a presence in more than 50 countries. The company's commitment to quality is evidenced by its in-house R&D capabilities and adherence to international manufacturing standards. Its reliability is further demonstrated by partnerships with leading global pharmaceutical companies, underscoring trust in its products and services.
2Certifications to Verify
Importers should verify the following certifications when considering Natco Pharma as a supplier:
- FDA Registration: Confirm the company's registration status with the U.S. Food and Drug Administration to ensure compliance with U.S. regulations.
- WHO-GMP Certification: Verify the World Health Organization's Good Manufacturing Practice certification to ensure adherence to international quality standards.
- EU GMP Certification: Check for European Union Good Manufacturing Practice certification to facilitate market access within EU member states.
- ISO Certifications: Review any ISO certifications related to quality management systems to assess the company's commitment to quality.
These certifications can typically be verified through the respective regulatory bodies' official websites or by contacting Natco Pharma directly.
3Due Diligence Checklist
When conducting due diligence on Natco Pharma, consider the following steps:
- Regulatory Compliance: Review the company's compliance with international regulatory standards, including FDA, WHO-GMP, and EU GMP certifications.
- Quality Assurance: Assess the company's quality management systems and any ISO certifications to ensure product quality.
- Financial Stability: Examine financial statements and reports to evaluate the company's financial health and stability.
Frequently Asked Questions — Natco Pharma Limited
How many pharmaceutical products does Natco Pharma Limited export from India?
Natco Pharma Limited exports 17 pharmaceutical products across 6 therapeutic categories. The top exports are Everolimus ($11.2M), Oseltamivir ($8.8M), Apixaban ($3.6M), Anastrozole ($2.7M), Ondansetron ($1.6M). Total export value is $36.0M.
What is Natco Pharma Limited's total pharmaceutical export value?
Natco Pharma Limited's total pharmaceutical export value is $36.0M, based on 865 verified shipments recorded in Indian Customs (DGFT) data.
In which products is Natco Pharma Limited the #1 Indian exporter?
Natco Pharma Limited is the #1 Indian exporter in 4 products: Everolimus (29.4% market share), Oseltamivir (7.2% market share), Lapatinib (20.5% market share), Erlotinib (20.4% market share).
What therapeutic categories does Natco Pharma Limited cover?
Natco Pharma Limited exports across 6 therapeutic categories. The largest are Immunosuppressants (31.0%, 1 products), Antivirals (24.5%, 1 products), Advanced Oncology (24.2%, 10 products).
Get Full Natco Pharma Limited Export Intelligence
Access shipment-level details, pricing data, buyer connections, product breakdown, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify manufacturer licensing, GMP certification, and export registration with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Matching: Natco Pharma Limited identified across shipments using name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 3.Market Share: Calculated per product as Natco Pharma Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 865 individual customs records matching Natco Pharma Limited.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
17 Products Tracked
6 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports per product category. For current shipment-level data, contact TransData Nexus.