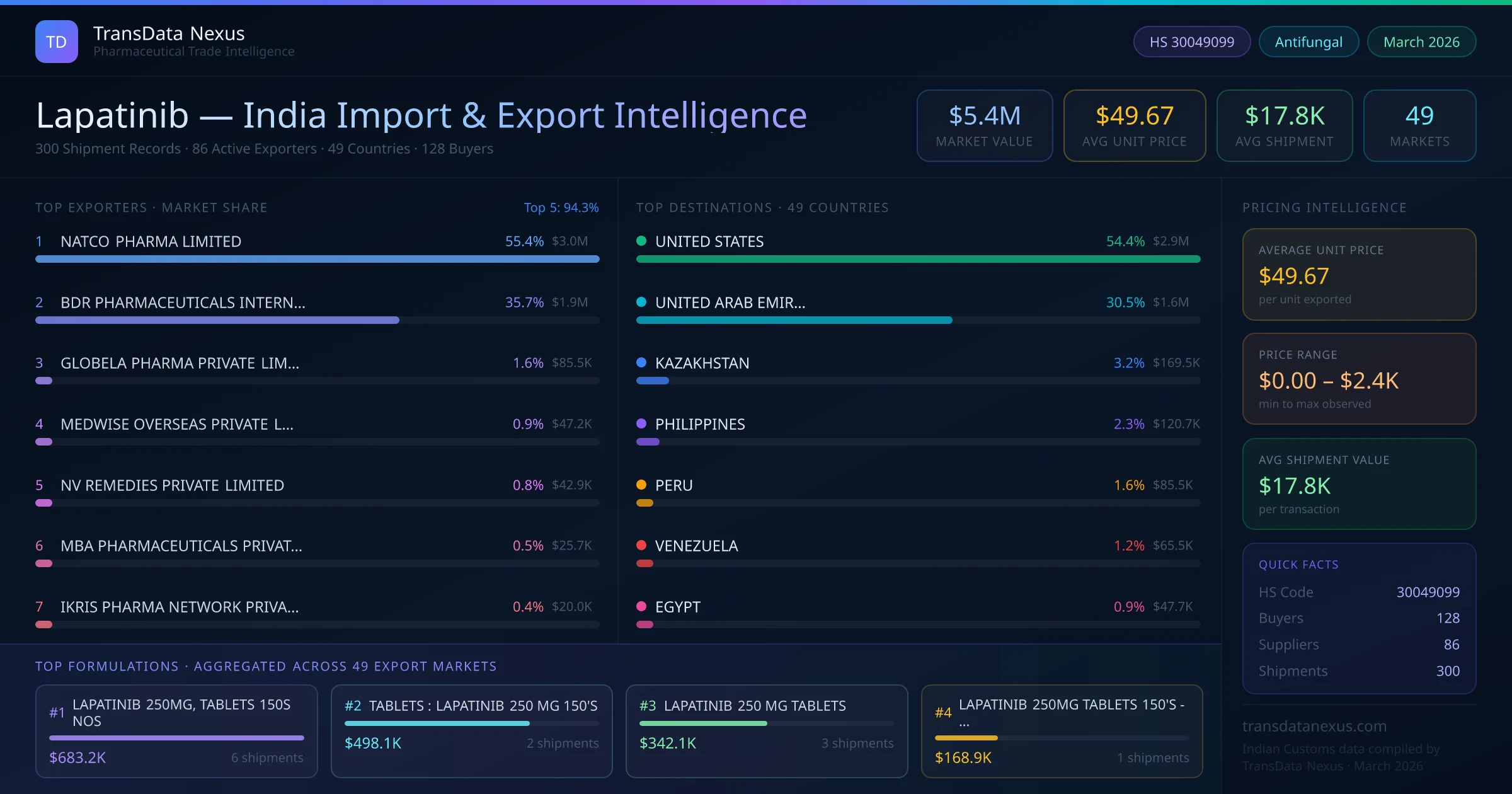
In April 2025, the World Health Organization unveiled a global repository for National Essential Medicines Lists (nEMLs), providing a centralized digital platform that compiles 150 national essential medicines lists from six global regions. This repository aims to enhance access to critical healthcare data, supporting policymakers, healthcare professionals, and researchers. While lapatinib is not included in the WHO Model List of Essential Medicines, its presence in various national lists could influence its global accessibility and adoption. (who.int)
In January 2026, the FDA updated its list of approved generic drugs, confirming the availability of generic lapatinib ditosylate tablets from multiple manufacturers, including those based in India. This development underscores the increasing competition in the U.S. market and the importance of maintaining stringent quality standards to ensure market share.
In February 2026, the WHO published a fact sheet on essential medicines, emphasizing the need for medicines that effectively and safely treat the priority healthcare needs of populations. The document highlights the importance of medicines being available, affordable, and of assured quality at all times. While lapatinib is not listed in the WHO Model List, its role in treating HER2-positive breast cancer remains significant. (who.int)
In March 2026, the WHO updated its list of National Regulatory Authorities (NRAs) operating at maturity levels 3 and 4, indicating robust regulatory systems. This update is crucial for exporters, as partnering with countries having mature regulatory systems can facilitate smoother market entry and compliance. (who.int)
In March 2026, the WHO published a report on the selection and use of essential medicines, providing updated guidance on the inclusion criteria and processes for the WHO Model List. This report serves as a valuable resource for manufacturers and policymakers in understanding global standards and expectations. (who.int)
These developments highlight the dynamic nature of the pharmaceutical regulatory landscape and underscore the importance for Indian exporters of lapatinib to stay informed and compliant with international standards and regulations.