In November 2024, the EMA adopted a positive opinion for Sarclisa (isatuximab) in combination with Bortezomib, lenalidomide, and dexamethasone for the treatment of newly diagnosed multiple myeloma patients ineligible for autologous stem cell transplant. This development may influence the demand for Bortezomib in combination therapies.
In July 2023, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of Bortezomib, highlighting its continued importance in global health.
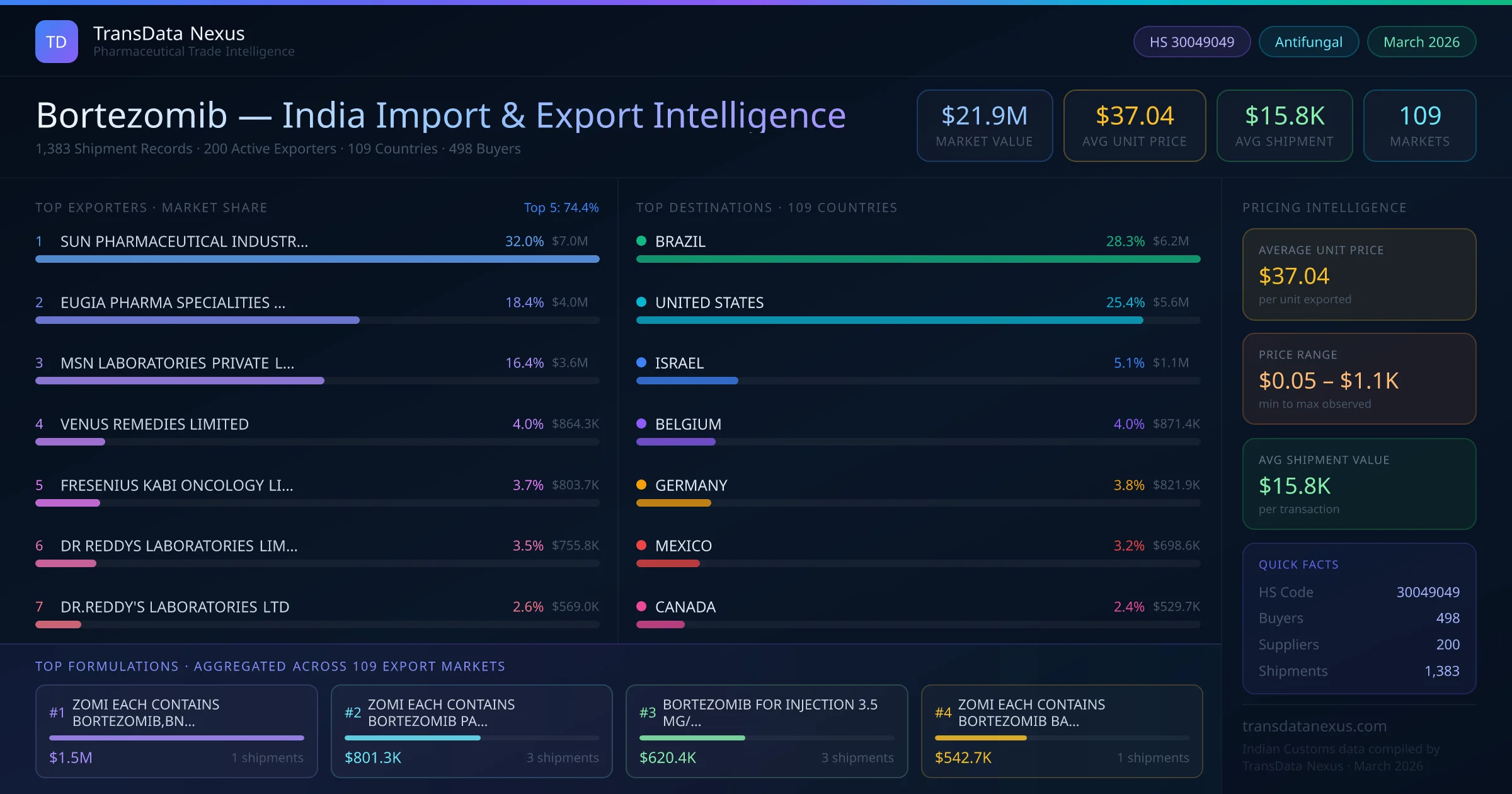
In April 2024, the NPPA conducted a review of anti-cancer drug prices, including Bortezomib, to assess market dynamics and ensure affordability. While no price ceiling was imposed, the review emphasized the need for price monitoring to prevent exorbitant pricing.
In December 2024, the CDSCO issued revised guidelines for the export of pharmaceutical products, streamlining the NOC process to facilitate faster approvals for manufacturers, including those exporting Bortezomib.
In February 2025, the FDA issued a warning letter to an Indian manufacturer of Bortezomib, citing deviations from cGMP regulations. This underscores the importance of maintaining stringent quality control measures to ensure compliance with international standards.
These developments reflect the dynamic nature of the pharmaceutical industry and the need for continuous monitoring of regulatory changes, market trends, and compliance requirements to maintain a competitive edge in the global Bortezomib market.