In June 2022, the EMA announced the withdrawal of the marketing authorization for Imatinib Actavis, following the request of the marketing authorization holder due to commercial reasons. This reflects the competitive landscape and market dynamics affecting generic imatinib products in the EU.
In August 2023, the EMA withdrew the marketing authorization for Imatinib Koanaa, also at the request of the marketing authorization holder, indicating ongoing shifts in the European market for generic imatinib.
In January 2026, the EMA's Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion for Iclusig (ponatinib), recommending its use in adult patients with chronic myeloid leukemia resistant to prior therapies, including imatinib. This development introduces additional therapeutic options in the treatment landscape.
In February 2026, the NPPA revised the ceiling price for imatinib under the DPCO, reflecting efforts to balance affordability with the sustainability of pharmaceutical manufacturing in India.
In March 2026, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of imatinib, which underscores its continued significance in global health.
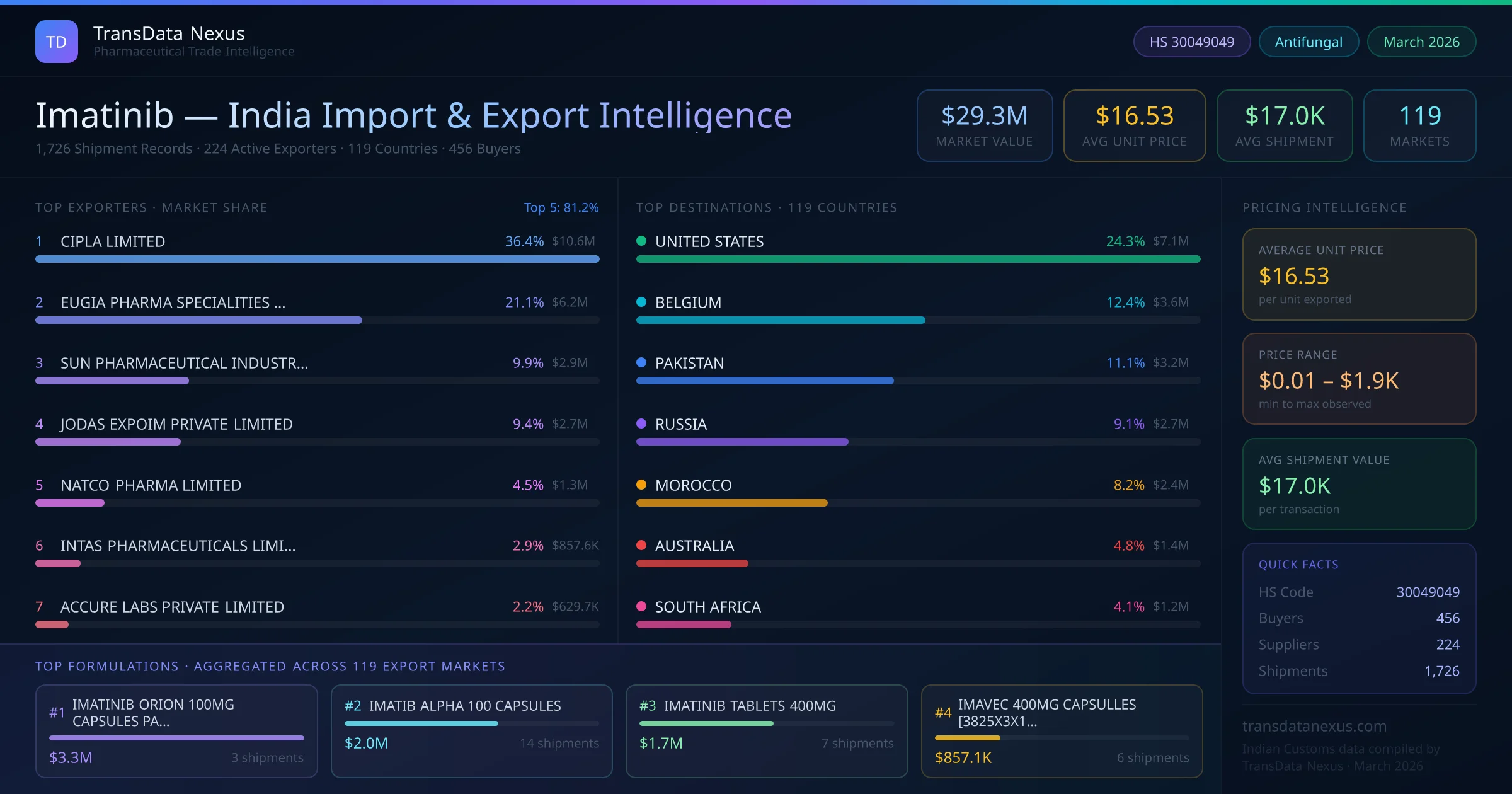
These developments highlight the dynamic regulatory and market environment surrounding imatinib, influencing both its global availability and the strategic decisions of Indian exporters.