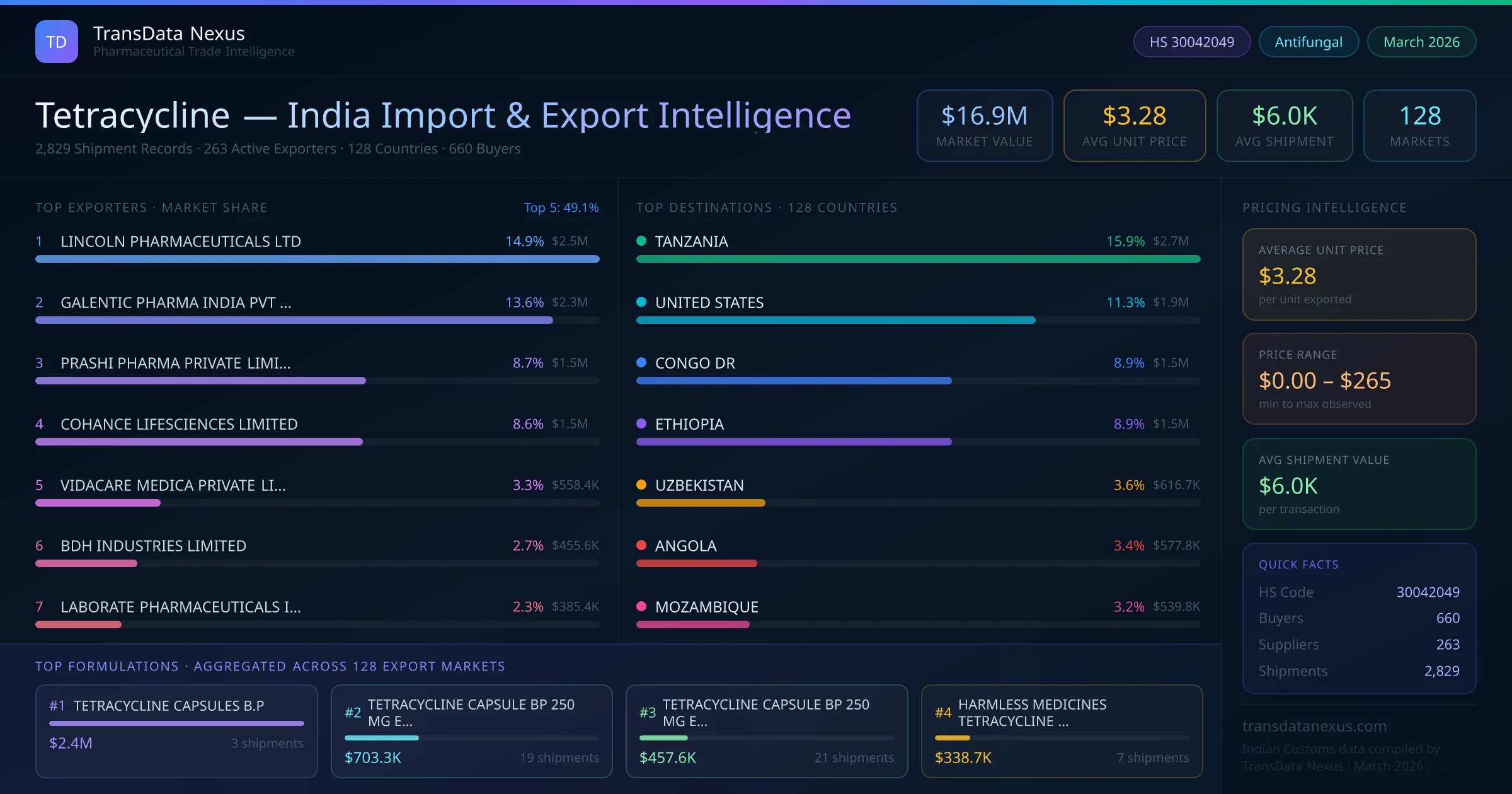
In August 2025, the NPPA revised the ceiling price for tetracycline, reducing it by 5% to enhance affordability for patients. This adjustment reflects the authority's commitment to making essential medicines more accessible.
In November 2025, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of tetracycline. This reaffirms the antibiotic's critical role in treating various bacterial infections globally.
In January 2026, the FDA issued Import Alert 36-04 concerning honey and blended syrup due to unsafe drug residues, including tetracycline. While this alert does not directly impact pharmaceutical tetracycline, it highlights the FDA's vigilance regarding antibiotic residues in imported products.
In February 2026, the EMA initiated a review of tetracycline-containing products to assess their benefit-risk balance, focusing on antimicrobial resistance concerns. This review aims to ensure the continued efficacy and safety of tetracycline in the European market.
In March 2026, the CDSCO issued new guidelines for the export of antibiotics, including tetracycline, emphasizing stringent quality control measures to align with international standards. These guidelines aim to bolster the credibility of Indian pharmaceutical exports.
These developments underscore the dynamic regulatory landscape surrounding tetracycline, reflecting ongoing efforts to balance accessibility, safety, and efficacy in the global pharmaceutical market.