In the United States, regorafenib is marketed under the brand name Stivarga. The FDA approved Stivarga for the treatment of metastatic colorectal cancer in September 2012, gastrointestinal stromal tumors in February 2013, and hepatocellular carcinoma in April 2017. The FDA's Orange Book lists several patents for Stivarga, including U.S. Patent Numbers 8,637,553 (expiring February 16, 2031), 8,680,124 (expiring June 2, 2030), 9,458,107 (expiring April 8, 2031), and 9,957,232 (expiring July 9, 2032). These patents have influenced the approval of generic versions. For instance, Actavis Laboratories FL, Inc. received FDA approval for its generic regorafenib tablets (40 mg) on December 10, 2020, under ANDA 209728. This approval reflects the FDA's commitment to increasing generic competition in the oncology sector.
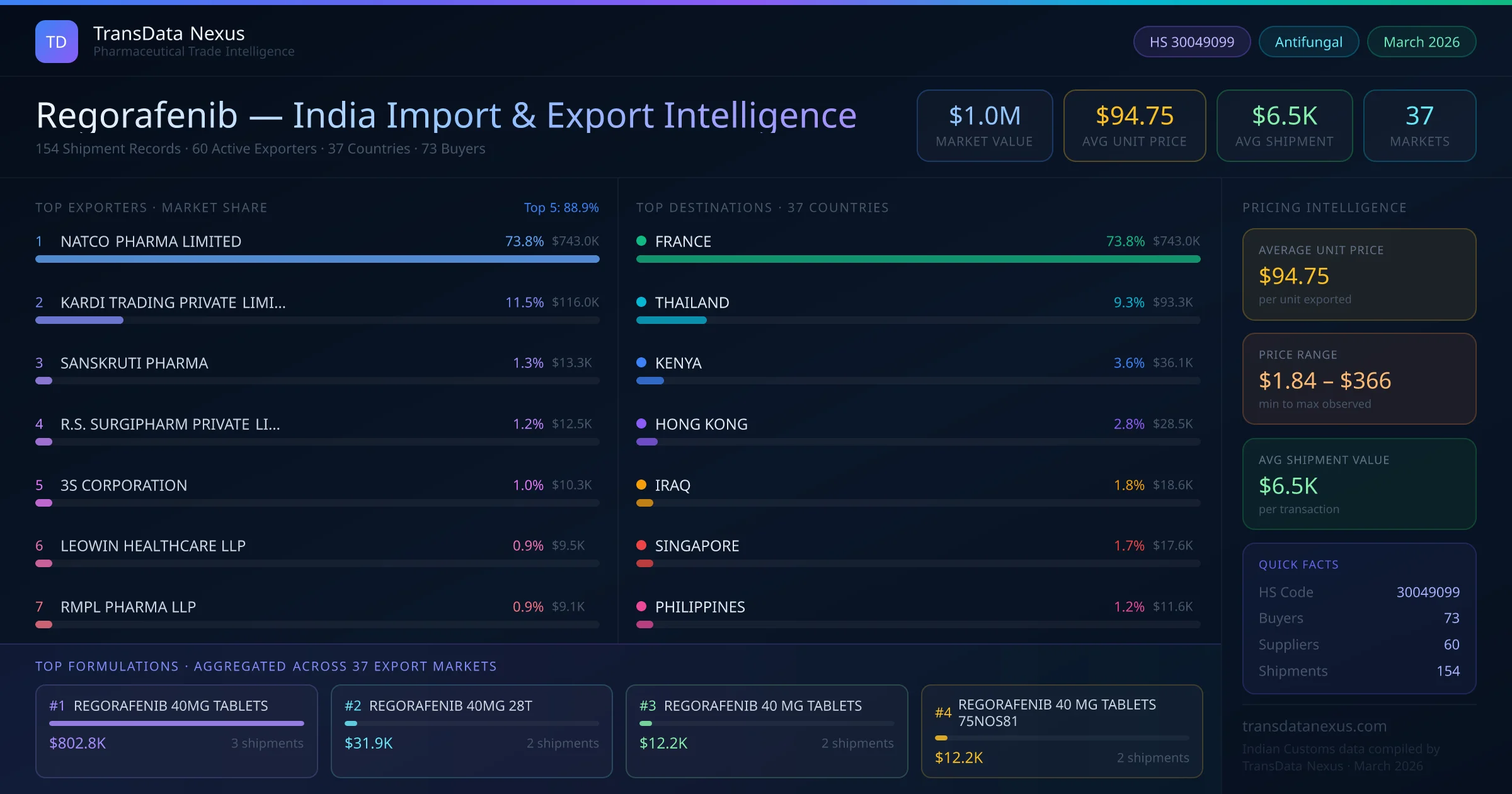
The presence of 60 active Indian exporters indicates a robust supply chain catering to the U.S. market. However, exporters must navigate the FDA's stringent regulatory requirements, including compliance with Current Good Manufacturing Practices (cGMP) and potential import alerts. As of March 2026, there are no active FDA import alerts specifically targeting regorafenib from India, suggesting adherence to regulatory standards by Indian manufacturers.