In the United States, the Food and Drug Administration (FDA) granted initial approval for osimertinib on November 13, 2015, for the treatment of patients with metastatic EGFR T790M mutation-positive NSCLC who have progressed on or after EGFR TKI therapy. Subsequent approvals expanded its indications, including adjuvant therapy after tumor resection in adult patients with NSCLC harboring EGFR exon 19 deletions or exon 21 L858R mutations, approved on December 18, 2020. The most recent approval, dated February 16, 2024, authorized its use in combination with pemetrexed and platinum-based chemotherapy for the first-line treatment of adult patients with locally advanced or metastatic NSCLC with specific EGFR mutations.
As of March 2026, the FDA's Orange Book lists one approved Abbreviated New Drug Application (ANDA) for osimertinib tablets, submitted by Alembic Pharmaceuticals Limited. This ANDA received tentative approval on May 31, 2023, indicating that while the generic product meets the necessary requirements, final approval is pending due to existing patent protections or exclusivity periods.
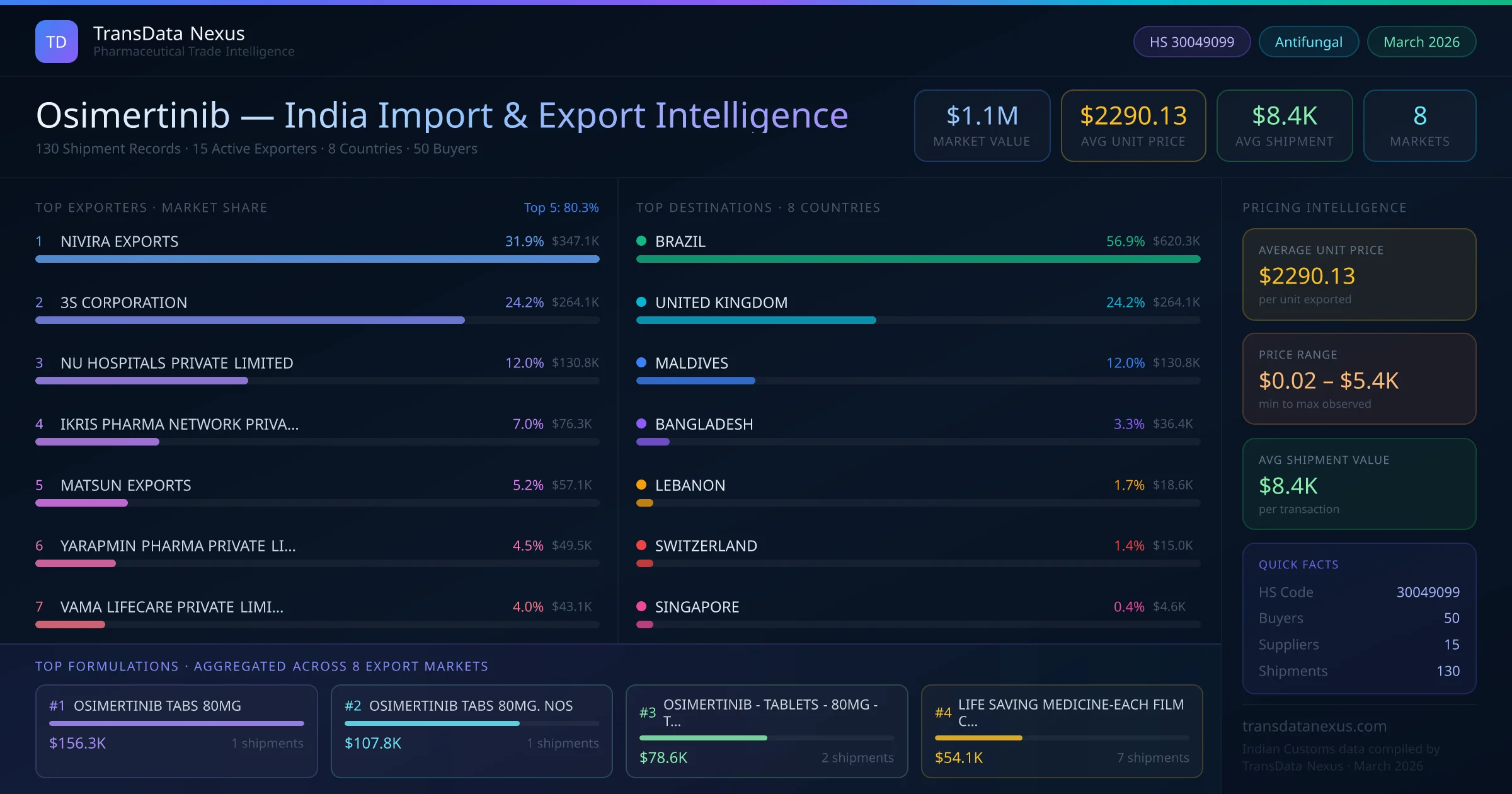
Given the limited number of approved ANDAs, the U.S. market for osimertinib remains predominantly brand-driven. This scenario presents opportunities for Indian exporters, especially considering the 15 active exporters from India and the 50% repeat buyer rate, to establish a foothold in the U.S. market upon the expiration of existing patents and exclusivities.