India to Hungary Pharmaceutical Export
Bilateral Trade Intelligence · $393.7M Total Trade · 74 Exporters · 128 Buyers · DGFT Verified · Updated March 2026
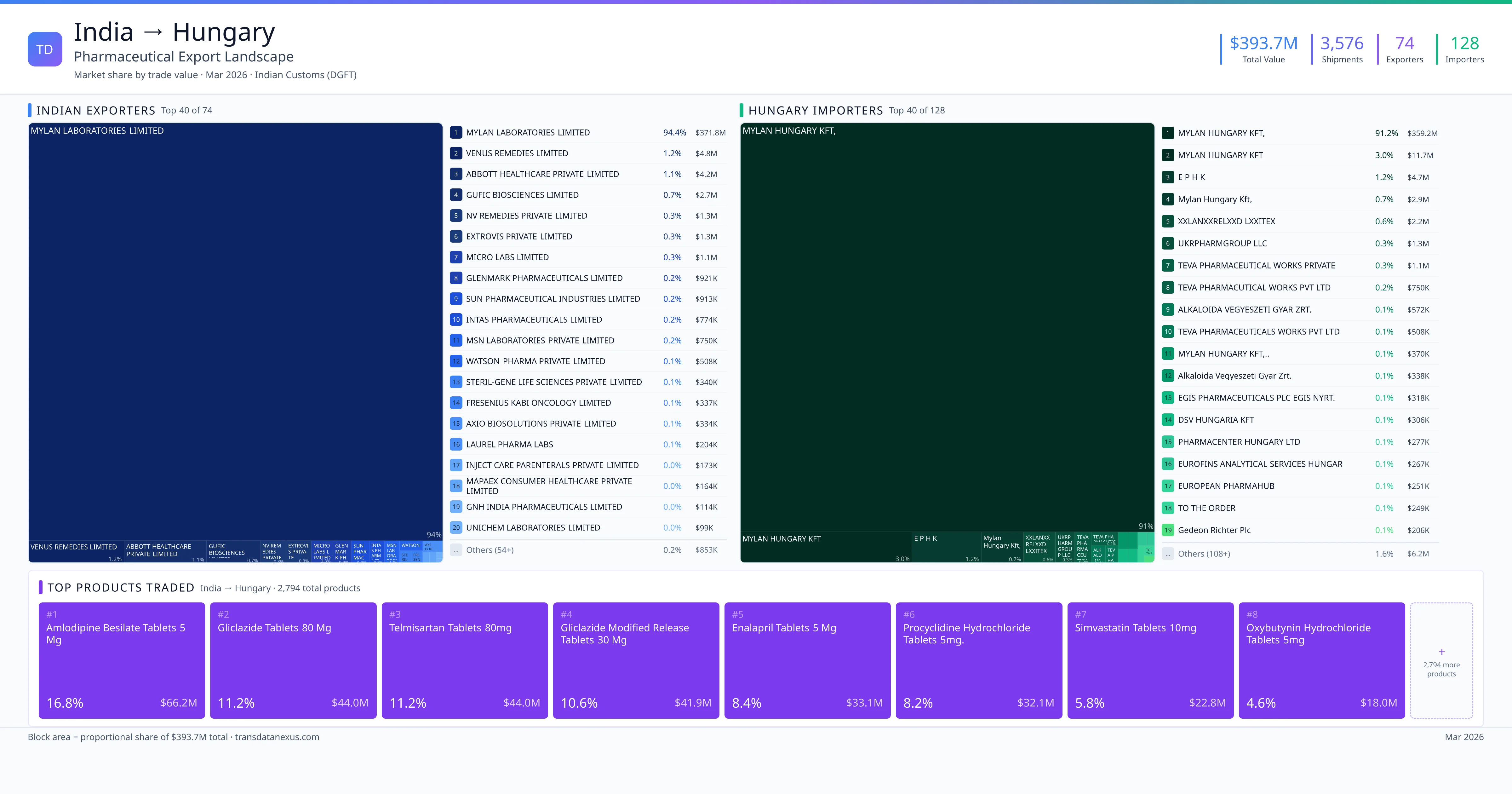
India exported $393.7M worth of pharmaceutical formulations to Hungary across 3,576 verified shipments, sourced from 74 Indian exporters supplying 128 Hungary buyers. The top exporters are MYLAN LABORATORIES LIMITED ($371.8M) and VENUS REMEDIES LIMITED ($4.8M). The leading products are Amlodipine ($66.2M) and Gliclazide ($44.0M). Average shipment value: $110.1K.
Top Pharmaceutical Formulations — India to Hungary
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Amlodipine Besilate Tablets 5 MG | $66.2M | 16.8% |
| 2 | Gliclazide Tablets 80 MG | $44.0M | 11.2% |
| 3 | Telmisartan Tablets 80mg | $44.0M | 11.2% |
| 4 | Gliclazide Modified Release Tablets 30 MG | $41.9M | 10.6% |
| 5 | Enalapril Tablets 5 MG | $33.1M | 8.4% |
| 6 | Procyclidine Hydrochloride Tablets 5mg. | $32.1M | 8.2% |
| 7 | Simvastatin Tablets 10mg | $22.8M | 5.8% |
| 8 | Oxybutynin Hydrochloride Tablets 5mg | $18.0M | 4.6% |
| 9 | Zolpidem Tartrate 10mg | $15.9M | 4.0% |
| 10 | Harmless Medicines, Pravastatin Sodium Tablets 10 MG | $6.1M | 1.6% |
| 11 | Enalapril Tablets 20 MG | $5.2M | 1.3% |
| 12 | Ibuprofen Lysine Tablets 684mg (packs | $1.7M | 0.4% |
| 13 | Harmless Medicines, Atova/prog Tablets 250/100mg (eu) | $1.3M | 0.3% |
| 14 | Harmless Medicines, Ibuprofen Lysine Tab684mg (eu) | $855.7K | 0.2% |
| 15 | Propafenone Hydrochloride Film Coated Ta | $839.6K | 0.2% |
India exports 20+ pharmaceutical formulations to Hungary with a combined trade value of $393.7M. Key products include Amlodipine ($66.2M), Gliclazide ($44.0M), Telmisartan ($44.0M), Enalapril ($33.1M), Simvastatin ($22.8M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 3,576 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Hungary Trade Routes
These are the top pharmaceutical products exported from India to Hungary, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Hungary buyers, regulatory requirements, and logistics for that specific product corridor. Products include Amlodipine ($66.2M), Gliclazide ($44.0M), Telmisartan ($44.0M), Enalapril ($33.1M), Simvastatin ($22.8M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Hungary
74 Indian pharmaceutical companies export finished formulations to Hungary. Leading exporters include Mylan Laboratories Limited, Venus Remedies Limited, Abbott Healthcare Private Limited, Gufic Biosciences Limited. The top exporter accounts for 94.4% of total India–Hungary pharma exports. Source: Indian Customs (DGFT).
Top Hungary Buyers from India
128 companies in Hungary import pharmaceutical formulations from India. Top buyers include Mylan Hungary Kft,, Mylan Hungary Kft, E P H K, Mylan Hungary Kft,. The largest buyer accounts for 91.2% of India–Hungary pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Hungary Pharmaceutical Shipments
Indian Export Ports
Hungary Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Hungary Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Hungary has experienced significant growth over the past decade. In 2022, bilateral trade surpassed US$1 billion for the first time, driven by increased investments and cooperation in sectors such as energy, technology, and education. This expansion reflects a broader trend of strengthening economic ties between the two nations.
Hungary's pharmaceutical imports have been predominantly sourced from Germany, the Netherlands, and Belgium. In 2023, Germany and the Netherlands were the leading import partners, with Hungary importing nearly one billion euros' worth of pharmaceutical products from these countries. While specific data on India's share in Hungary's pharmaceutical imports is not readily available, the increasing trade volume suggests a growing presence of Indian pharmaceutical products in the Hungarian market.
2India's Market Position
India's pharmaceutical exports to Hungary have been substantial, with a total export value of US$393.7 million over 3,576 shipments from 2022 to 2026. The average shipment value stands at US$110,000, indicating a consistent demand for Indian pharmaceutical formulations in Hungary. The top 10 products exported include amlodipine besilate tablets, gliclazide tablets, and telmisartan tablets, highlighting India's strength in cardiovascular and diabetes medications.
The dominance of Mylan Laboratories Limited, accounting for US$371.8 million of the total export value, underscores the strategic importance of this corridor for Indian pharmaceutical companies. Hungary's well-established pharmaceutical sector, with significant investments in research and development, provides a conducive environment for Indian exporters to expand their market share.
3Recent Developments
In March 2024, India and the European Free Trade Association (EFTA) signed the Trade and Economic Partnership Agreement (TEPA), aiming to eliminate or reduce tariffs on the majority of goods traded between India and EFTA countries, including Hungary. The agreement, effective from October 2025, is expected to enhance trade relations and provide preferential access for Indian pharmaceutical products to the European market.
Additionally, Hungary's commitment to expanding ties with India, particularly in sectors like energy and technology, is expected to further strengthen bilateral relations and create new avenues for pharmaceutical trade.
Hungary Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Hungary must navigate the European Medicines Agency (EMA) and Hungary's National Competent Authority (NCA) for marketing authorization. The EMA evaluates applications for generic medicines, which must demonstrate bioequivalence to a reference product authorized in the EU. This process typically involves providing quality data and bioavailability studies, while pre-clinical and clinical trials may be waived if the generic is identical in composition and strength to the reference product. (ema.europa.eu)
The Common Technical Document (CTD) format is the standard for dossier submissions in the EU, encompassing modules on administrative information, quality, non-clinical and clinical data. The approval timeline for generic medicines can vary but generally ranges from 210 to 270 days, depending on the complexity of the application and the efficiency of the regulatory process. Fees associated with the application process are subject to EMA's fee structure and may vary based on the type of application and the applicant's status.
2GMP & Facility Requirements
Hungary requires that manufacturing facilities comply with Good Manufacturing Practice (GMP) standards equivalent to those established by the World Health Organization (WHO), Pharmaceutical Inspection Convention (PIC/S), or the International Council for Harmonisation (ICH). This includes adherence to quality systems, validation of equipment, and implementation of a pharmaceutical quality system (PQS). The inspection process involves thorough evaluations to ensure compliance with these standards, and mutual recognition agreements may facilitate the acceptance of GMP certifications from recognized authorities. (bfda.gov.bt)
3Import Documentation
For importing pharmaceutical products into Hungary, Indian companies must obtain an import license and provide necessary documentation, including a Certificate of Pharmaceutical Product (CPP), Certificate of Analysis (CoA), and proof of GMP compliance. Customs procedures require detailed documentation to ensure that imported medicines meet Hungary's regulatory standards and are authorized for sale within the country. (portal.nebih.gov.hu)
Product Categories & Therapeutic Trends — India to Hungary
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Hungary are predominantly concentrated in cardiovascular and diabetes management medications. The top three products—amlodipine besilate tablets (16.8%), gliclazide tablets (11.2%), and telmisartan tablets (11.2%)—collectively account for 39.2% of the total export value. This emphasis aligns with Hungary's significant burden of cardiovascular diseases (CVDs) and diabetes. In 2021, Hungary reported 57,964 deaths due to CVDs, with an age-standardized mortality rate of 273 per 100,000 population. Additionally, the prevalence of hypertension among adults was notably high, affecting 55.9% of men and 40.9% of women in 2019. These statistics underscore a substantial demand for effective cardiovascular and diabetes treatments, driving the dominance of these therapeutic categories in India's pharmaceutical exports to Hungary.
2Emerging Opportunities
The Hungarian pharmaceutical market is experiencing a shift towards generic medications, driven by government initiatives aimed at reducing healthcare costs. This trend presents opportunities for Indian exporters to supply cost-effective generic drugs, particularly in therapeutic areas where patent expirations are imminent. For instance, the oncology sector is projected to grow by 11% annually until 2032, indicating a rising demand for cancer treatments. (hungarytoday.hu) Additionally, Hungary's pharmaceutical industry invested approximately EUR 382 million in research and development in 2022, accounting for 2.4% of total EU pharmaceutical R&D expenditure. This investment suggests a focus on innovation and the potential for introducing new therapeutic products, including biosimilars, to meet evolving healthcare needs.
3Demand Drivers
Hungary's aging population is a significant driver of pharmaceutical import demand. In 2021, 20.4% of the population was aged 65 and over, with projections indicating an increase to 27.8% by 2050. This demographic shift is expected to disproportionately increase the burden of age-related diseases, particularly cancer. A study projected that the incidence and mortality of cancer in Hungary would rise up to 2030, exacerbated by the aging population. These factors contribute to a growing demand for pharmaceutical products, especially those addressing chronic and age-related conditions, thereby driving the need for imports from countries like India.
Trade Policy & Tariff Intelligence — India and Hungary
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Hungary, as a member of the European Union (EU), applies the EU's Common Customs Tariff (CCT) to imports, including pharmaceutical formulations classified under Harmonized System (HS) codes 3003 and 3004. The Most Favored Nation (MFN) tariff rates for these products are generally set at 0%, reflecting the EU's commitment to facilitating the import of pharmaceutical goods. This zero-duty rate is consistent across all non-EU countries, including India.
Under the EU's Generalized Scheme of Preferences (GSP), developing countries may benefit from reduced or zero tariffs on certain products. However, India is classified as a "graduated" country under the GSP, meaning it does not receive preferential tariff treatment for pharmaceutical products. Consequently, Indian pharmaceutical exports to Hungary are subject to the standard MFN tariff rates.
Hungary does not impose anti-dumping or countervailing duties on Indian pharmaceutical products. The EU's trade defense measures are applied uniformly across member states, and there are no specific duties targeting Indian pharmaceutical imports. This absence of additional duties ensures that Indian pharmaceutical formulations can enter the Hungarian market without facing trade barriers beyond standard customs procedures.
2Trade Agreements
As of March 2026, there are no bilateral Free Trade Agreements (FTAs) between India and Hungary. Hungary, being an EU member, participates in the EU's trade agreements, but these do not extend to India in the pharmaceutical sector. The EU's trade relations with India are governed by the EU-India Trade and Cooperation Agreement, which does not currently include specific provisions for pharmaceutical products. Therefore, Indian pharmaceutical exports to Hungary are not subject to preferential treatment under any existing trade agreements.
3IP & Patent Landscape
Hungary's intellectual property (IP) framework aligns with EU regulations, providing robust protection for pharmaceutical patents. Patents granted in Hungary are enforceable within its jurisdiction, offering protection for innovative pharmaceutical products. However, Hungary does not have specific provisions that facilitate the entry of Indian generic drugs into its market. The EU's patent system does not include mechanisms such as compulsory licensing or data exclusivity periods that would allow for the expedited entry of generic versions of patented drugs. Consequently, Indian generic pharmaceutical companies may face challenges in entering the Hungarian market due to the stringent patent protections in place.
Supply Chain & Logistics — India to Hungary Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Hungary primarily utilize sea and air transport, each with distinct transit times and logistical considerations.
Sea Freight: The most common sea route involves departing from major Indian ports such as Mumbai (Nhava Sheva) and Chennai (Ennore), proceeding through the Suez Canal, and arriving at European ports like Rotterdam. For instance, a direct service from Ennore to Rotterdam takes approximately 33 days, with vessels departing 1-2 times a week. From Rotterdam, goods are typically transported by rail or road to Hungary, adding an additional 2-3 days to the overall transit time.
Air Freight: Air transport offers a faster alternative, with transit times ranging from 2 to 6 business days, depending on the specific route and carrier. Budapest Ferenc Liszt International Airport (BUD) serves as Hungary's primary cargo hub, handling a significant volume of pharmaceutical imports. Air freight is particularly advantageous for time-sensitive or high-value pharmaceutical products.
Impact of Red Sea Disruptions: Disruptions in the Red Sea, such as the blockage of the Suez Canal, can significantly impact sea freight routes, leading to delays and increased costs. For example, the 2021 Suez Canal blockage resulted in substantial shipping delays worldwide. While air freight is less susceptible to such disruptions, it may experience indirect effects due to increased demand and potential congestion at alternative ports.
2Port Infrastructure
In India, key export ports for pharmaceutical shipments include Delhi Air, Cochin Sea, and LCS Jaigaon. Delhi Air accounts for 5.2% of the shipments, Cochin Sea 0.8%, and LCS Jaigaon 0.3%. These ports are equipped to handle pharmaceutical exports, with Delhi Air serving as a significant air cargo hub.
In Hungary, Budapest is the primary import port, handling 69.9% of the shipments, with additional facilities accounting for 16.9% and 5.0%. Budapest's infrastructure supports efficient processing of pharmaceutical imports, ensuring timely delivery to the domestic market.
3Cold Chain & Compliance
Temperature-sensitive pharmaceutical formulations require strict cold chain logistics to maintain product efficacy. This involves temperature-controlled storage and transportation, with continuous monitoring to ensure compliance with Good Distribution Practice (GDP) standards. Packaging must be designed to maintain the required temperature range throughout the supply chain. In Hungary, the National Institute of Pharmacy and Nutrition (OGYÉI) regulates pharmaceutical imports, ensuring that all shipments meet the necessary safety and quality standards.
Market Opportunity Assessment — Hungary for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Hungary's pharmaceutical market is projected to achieve a revenue of US$1.17 billion in 2025, with an anticipated annual growth rate of 4.55% from 2025 to 2030, reaching US$1.47 billion by 2030. The market is characterized by a significant presence of generic drugs, which have been gaining traction due to government initiatives aimed at reducing healthcare costs. In 2023, Hungary's active pharmaceutical ingredients market generated a revenue of US$998.2 million and is expected to grow at a compound annual growth rate (CAGR) of 5.9% from 2024 to 2030.
2Healthcare System
Hungary operates a universal healthcare system managed by the National Health Insurance Fund Administration (NEAK), which provides comprehensive coverage to residents through a single health insurance fund. (eurohealthobservatory.who.int) The system is financed through tax and social health insurance contributions, with the NEAK overseeing the management of the Health Insurance Fund, including funding, reimbursement accounting, and maintaining records. (neak.gov.hu) In May 2025, the Hungarian government introduced Government Decree 110/2025, centralizing the inventory management of medical consumables used by public healthcare institutions.
3Opportunity for Indian Exporters
The Hungarian pharmaceutical market presents significant opportunities for Indian exporters, particularly in the generic drug segment. In 2022, India exported finished pharmaceutical formulations valued at US$393.7 million to Hungary, with 74 Indian exporters supplying 128 Hungarian buyers. The top products included Amlodipine Besilate Tablets 5 mg, Gliclazide Tablets 80 mg, and Telmisartan Tablets 80 mg, indicating a strong demand for cardiovascular and diabetes medications. The market's projected growth and the increasing emphasis on generics align with India's strengths in this sector, suggesting a favorable environment for expanding exports.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Hungary
Competing origins, India's edge, challenges and threats
1Competing Origins
Hungary's pharmaceutical market is predominantly supplied by European Union (EU) countries, with Germany and the Netherlands being the leading import partners. In 2023, Hungary imported nearly one billion euros' worth of pharmaceutical products from these two nations, making them the largest suppliers. Belgium also plays a significant role, exporting pharmaceutical products to Hungary valued at 680 million euros during the same period.
China and other emerging markets contribute to Hungary's pharmaceutical imports, but their share remains relatively smaller compared to EU suppliers. For instance, in 2023, China accounted for approximately 9.35% of Hungary's pharmaceutical imports, amounting to 570 million euros.
India's pharmaceutical exports to Hungary are substantial, with a total export value of $393.7 million USD over 3,576 shipments from 74 Indian exporters to 128 Hungarian buyers. This positions India as a significant supplier in Hungary's pharmaceutical market, though it still trails behind the leading EU countries.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages:
- Cost Competitiveness: India is renowned for providing high-quality pharmaceuticals at affordable prices, making it an attractive option for cost-conscious markets.
- Regulatory Compliance: A significant number of Indian pharmaceutical manufacturing plants are approved by the U.S. Food and Drug Administration (FDA), underscoring the industry's adherence to stringent international standards.
- Product Variety: India's pharmaceutical sector boasts a broad portfolio, including over-the-counter medicines, generics, active pharmaceutical ingredients (APIs), vaccines, biosimilars, and custom research manufacturing (CRM).
- English-Speaking Workforce: The widespread use of English in India facilitates effective communication and collaboration with international partners.
3Challenges & Threats
Despite its strengths, India's pharmaceutical exports to Hungary face several challenges:
- Regulatory Compliance: Navigating the complex regulatory landscape of the European Medicines Agency (EMA) and Hungary's National Competent Authority requires meticulous attention to detail and can be time-consuming.
- Competition from China: China's growing pharmaceutical industry poses a competitive threat, especially in terms of pricing and increasing market presence.
- Quality Perception: While India is known for cost-effective solutions, there may be perceptions regarding the quality of its pharmaceutical products compared to those from established EU manufacturers.
Addressing these challenges is crucial for India to strengthen its position in Hungary's pharmaceutical market.
FAQ — India to Hungary Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Hungary?
India exported pharmaceuticals worth $393.7M to Hungary across 3,576 verified shipments.
Who are the top Indian pharmaceutical exporters to Hungary?
1. MYLAN LABORATORIES LIMITED — $371.8M. 2. VENUS REMEDIES LIMITED — $4.8M. 3. ABBOTT HEALTHCARE PRIVATE LIMITED — $4.2M. Total: 74 suppliers.
Which companies in Hungary import pharmaceuticals from India?
1. MYLAN HUNGARY KFT, — $359.2M. 2. MYLAN HUNGARY KFT — $11.7M. 3. E P H K — $4.7M. 128 buyers total.
What pharmaceutical products does India export most to Hungary?
1. Amlodipine Besilate Tablets 5 MG ($66.2M, 16.8%); 2. Gliclazide Tablets 80 MG ($44.0M, 11.2%); 3. Telmisartan Tablets 80mg ($44.0M, 11.2%); 4. Gliclazide Modified Release Tablets 30 MG ($41.9M, 10.6%); 5. Enalapril Tablets 5 MG ($33.1M, 8.4%)
Which ports handle pharmaceutical shipments from India to Hungary?
Export: DELHI AIR, COCHIN SEA, LCS JAIGAON, NEPALGUNJ ICD, HAZIRA PORT SURAT ICD. Import: BUDAPEST,, BUDAPEST, Budapest, Koper, KOPER.
Why does Hungary import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $393.7M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Hungary?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Hungary pharmaceutical trade?
$110.1K per consignment across 3,576 shipments.
How many Indian pharmaceutical companies export to Hungary?
74 Indian companies. Largest: MYLAN LABORATORIES LIMITED with $371.8M.
How can I find verified Indian pharmaceutical suppliers for Hungary?
TransData Nexus covers 74 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Hungary Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 3,576 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Hungary identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 3,576 verified shipments from 74 Indian exporters to 128 Hungary buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
74 Exporters
128 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists