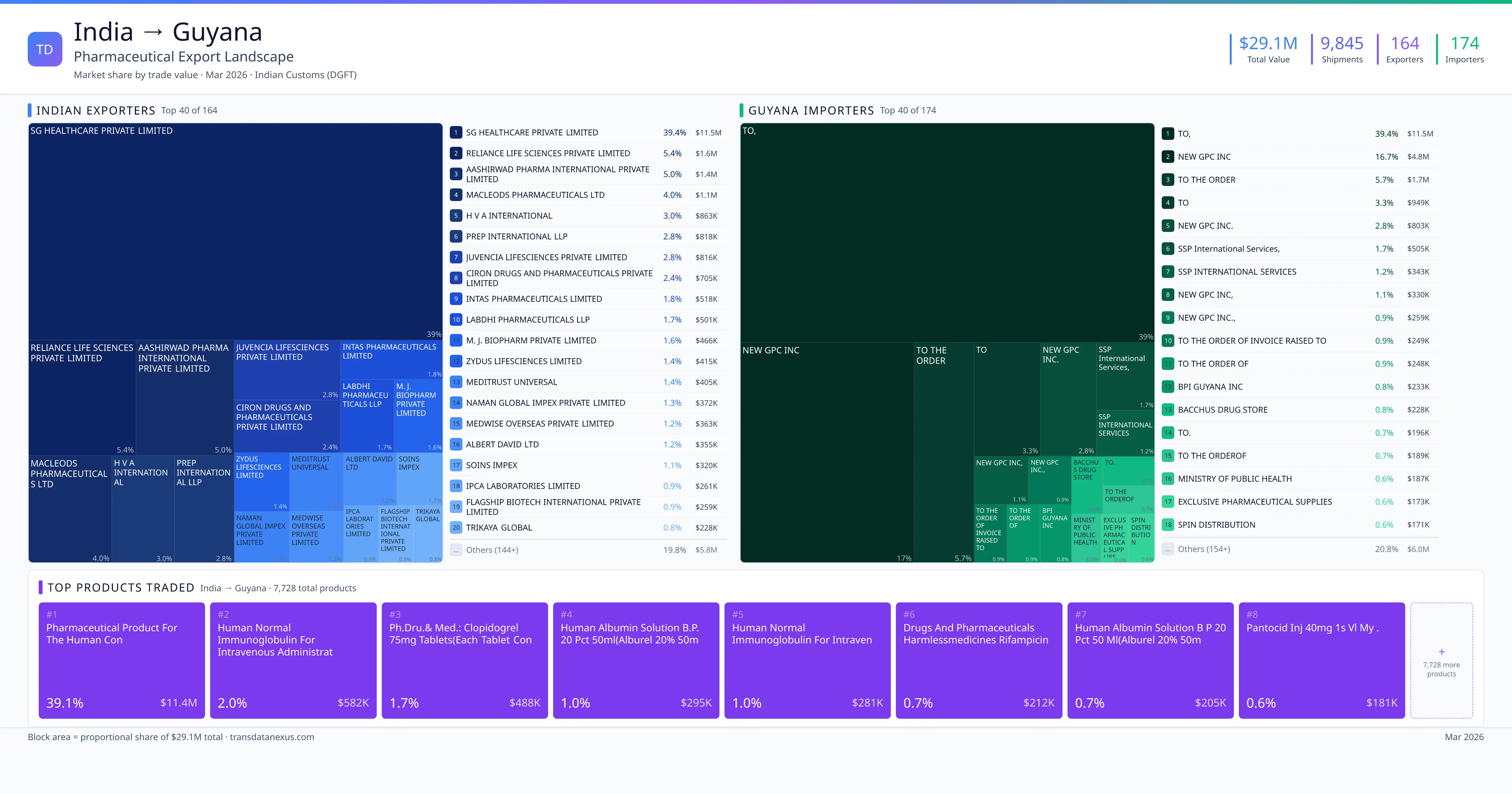
Indian pharmaceutical companies seeking to export finished formulations to Guyana must navigate a structured registration process overseen by the Government Analyst-Food and Drug Department (GA-FDD), the regulatory authority responsible for drug registration in Guyana. The process involves several key steps:
1. Application Submission: Applicants are required to submit a comprehensive application to the GA-FDD, including detailed product information, manufacturing details, and evidence of the product's safety, efficacy, and quality. All labeling must be in English and include relevant information as stipulated by the GA-FDD.
2. Document Verification and Evaluation: The GA-FDD reviews the submitted documents to ensure compliance with the Food and Drugs Act and associated regulations. This evaluation assesses the product's compliance with safety, efficacy, and quality standards.
3. Approval and Registration: Upon successful evaluation, the GA-FDD issues a Certificate of Registration, authorizing the importation and sale of the pharmaceutical product in Guyana. This certificate is a prerequisite for market entry.
It's important to note that the GA-FDD relies on assessments from recognized reference authorities, such as the United States, Canada, the United Kingdom, and Australia, to ensure the quality of imported pharmaceutical products.