India to Guyana Pharmaceutical Export
Bilateral Trade Intelligence · $29.1M Total Trade · 164 Exporters · 174 Buyers · DGFT Verified · Updated March 2026
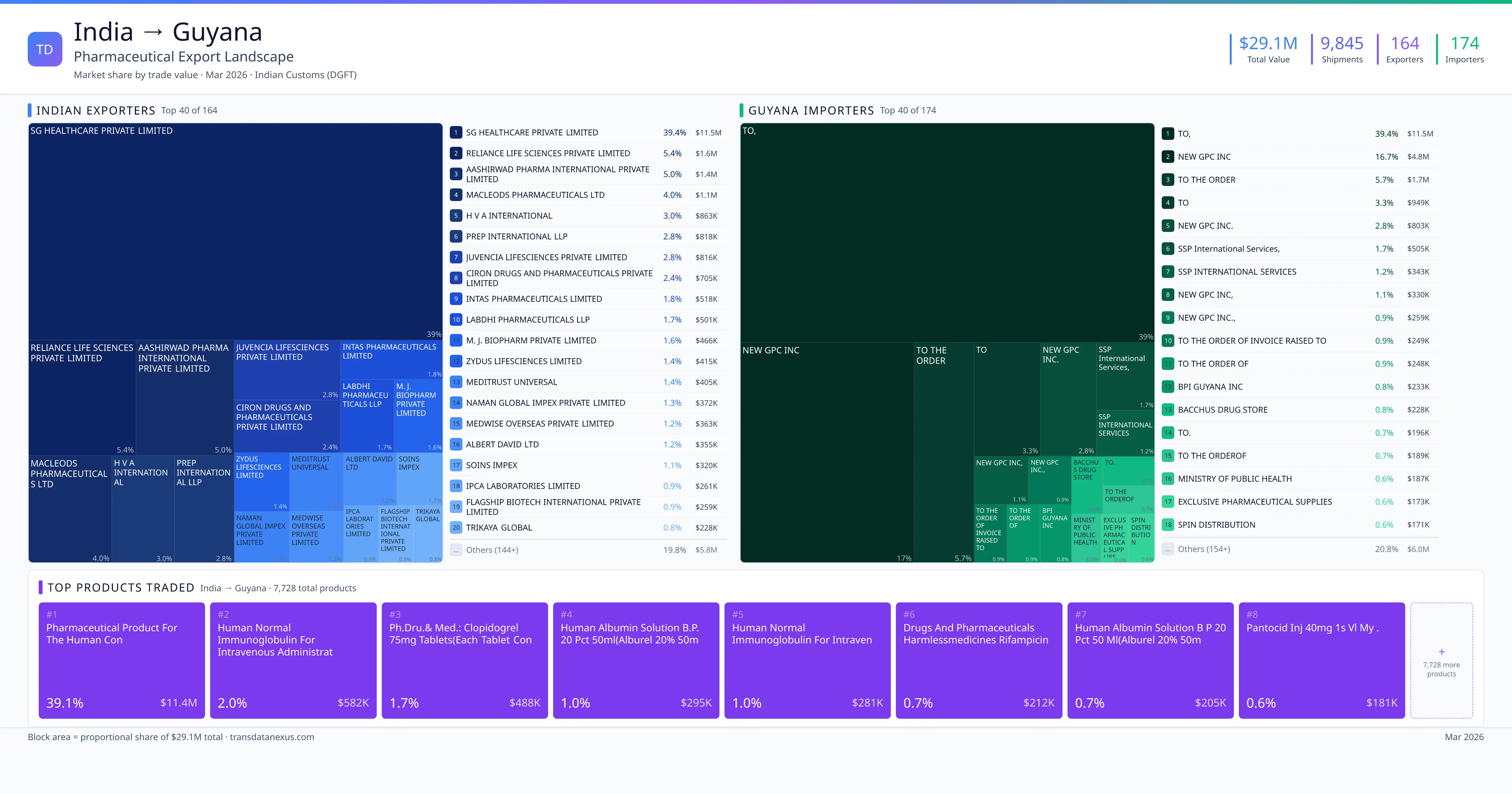
India exported $29.1M worth of pharmaceutical formulations to Guyana across 9,845 verified shipments, sourced from 164 Indian exporters supplying 174 Guyana buyers. The top exporters are SG HEALTHCARE PRIVATE LIMITED ($11.5M) and RELIANCE LIFE SCIENCES PRIVATE LIMITED ($1.6M). The leading products are Immunoglobulin ($348K) and Azithromycin ($130K). Average shipment value: $3.0K.
Top Pharmaceutical Formulations — India to Guyana
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Pharmaceutical Product for the Human Con | $11.4M | 39.1% |
| 2 | Human Normal Immunoglobulin for Intravenous Administration B.p.5% Solution 100m… | $581.6K | 2.0% |
| 3 | Ph.dru.& Med.: Clopidogrel 75mg Tablets(each Tablet Contains: Clopidogrel 75mg)… | $487.9K | 1.7% |
| 4 | Human Normal Immunoglobulin for Intraven | $281.3K | 1.0% |
| 5 | Drugs and Pharmaceuticals Harmlessmedicines Rifampicin Capsules BP 300 MG | $212.0K | 0.7% |
| 6 | Human Albumin Solution B P 20 Pct 50 Ml(alburel 20% 50ml) | $205.5K | 0.7% |
| 7 | Pantocid Injection 40mg 1s Vl My . | $180.6K | 0.6% |
| 8 | Human Albumin Solution B.p. 20 Pct 50ml(alburel 20% 50ml)vls | $172.4K | 0.6% |
| 9 | Hipen Clav 625 Tablet 1x10t Sale Afg (1x10 Tablets) | $162.5K | 0.6% |
| 10 | Pharmaceutical Product for the Human Use-zuclopenthixole Decanoate Injection Bp… | $139.5K | 0.5% |
| 11 | Pharmaceutical Medicines Fourderm Cream20 Gms | $139.2K | 0.5% |
| 12 | Phensod | $131.3K | 0.5% |
| 13 | Macromax 500 37543 Packs of 10x3 Tablets | $130.2K | 0.4% |
| 14 | Human Albumin Solution B.p. 20 Pct 50ml(alburel 20% 50ml) | $122.1K | 0.4% |
| 15 | Pharmaceutical Medicines Kamagra Oral Jelly 5gms | $111.8K | 0.4% |
India exports 19+ pharmaceutical formulations to Guyana with a combined trade value of $29.1M. Key products include Immunoglobulin ($348K), Azithromycin ($130K), Insulin ($94K), Tenofovir ($86K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 9,845 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Guyana Trade Routes
These are the top pharmaceutical products exported from India to Guyana, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Guyana buyers, regulatory requirements, and logistics for that specific product corridor. Products include Immunoglobulin ($348K), Azithromycin ($130K), Insulin ($94K), Tenofovir ($86K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Guyana
164 Indian pharmaceutical companies export finished formulations to Guyana. Leading exporters include Sg Healthcare Private Limited, Reliance Life Sciences Private Limited, Aashirwad Pharma International Private Limited, Macleods Pharmaceuticals Ltd. The top exporter accounts for 39.4% of total India–Guyana pharma exports. Source: Indian Customs (DGFT).
Top Guyana Buyers from India
174 companies in Guyana import pharmaceutical formulations from India. Top buyers include To,, New Gpc Inc, To The Order, To. The largest buyer accounts for 39.4% of India–Guyana pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Guyana Pharmaceutical Shipments
Indian Export Ports
Guyana Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Guyana Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Guyana has experienced significant growth over the past decade. In 2024, India exported approximately $13.64 million worth of pharmaceutical products to Guyana, marking a substantial increase from previous years. This surge reflects the strengthening bilateral relations and India's expanding role in supplying pharmaceutical products to the Caribbean region.
A pivotal milestone in this trade relationship occurred in November 2024, when India and Guyana formalized five Memoranda of Understanding (MoUs) aimed at enhancing cooperation in sectors including pharmaceuticals, healthcare, and agriculture. These agreements, signed during Prime Minister Narendra Modi's state visit to Guyana, are expected to further solidify the pharmaceutical trade corridor between the two nations. (dpi.gov.gy)
2India's Market Position
India's pharmaceutical exports to Guyana accounted for approximately 39% of Guyana's total pharmaceutical imports in 2024, positioning India as the leading supplier in this sector. This dominance underscores India's strategic importance in meeting Guyana's pharmaceutical needs and highlights the effectiveness of bilateral agreements in facilitating trade.
3Recent Developments
In November 2024, during Prime Minister Narendra Modi's state visit to Guyana, the two countries signed 10 MoUs covering key sectors such as agriculture, hydrocarbons, digital payments, and defense. Notably, one of these agreements focused on enhancing cooperation in the pharmaceutical sector, aiming to strengthen the existing trade corridor and explore new avenues for collaboration.
These developments are expected to further bolster the pharmaceutical trade between India and Guyana, fostering a more robust and mutually beneficial economic relationship.
Guyana Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Guyana must navigate a structured registration process overseen by the Government Analyst-Food and Drug Department (GA-FDD), the regulatory authority responsible for drug registration in Guyana. The process involves several key steps:
1. Application Submission: Applicants are required to submit a comprehensive application to the GA-FDD, including detailed product information, manufacturing details, and evidence of the product's safety, efficacy, and quality. All labeling must be in English and include relevant information as stipulated by the GA-FDD.
2. Document Verification and Evaluation: The GA-FDD reviews the submitted documents to ensure compliance with the Food and Drugs Act and associated regulations. This evaluation assesses the product's compliance with safety, efficacy, and quality standards.
3. Approval and Registration: Upon successful evaluation, the GA-FDD issues a Certificate of Registration, authorizing the importation and sale of the pharmaceutical product in Guyana. This certificate is a prerequisite for market entry.
It's important to note that the GA-FDD relies on assessments from recognized reference authorities, such as the United States, Canada, the United Kingdom, and Australia, to ensure the quality of imported pharmaceutical products.
2GMP & Facility Requirements
The GA-FDD mandates that pharmaceutical products imported into Guyana adhere to Good Manufacturing Practice (GMP) standards to ensure product quality and safety. Manufacturing facilities in India must comply with internationally recognized GMP standards, such as those outlined by the World Health Organization (WHO). This includes maintaining stringent quality control measures, proper documentation, and regular inspections. (who.int)
While specific details regarding the inspection process and mutual recognition agreements between India and Guyana are not explicitly detailed in the available sources, it is generally understood that the GA-FDD conducts thorough evaluations of manufacturing facilities to verify compliance with GMP standards. Establishing mutual recognition agreements can facilitate smoother trade relations and expedite the approval process for pharmaceutical products.
3Import Documentation
Importers of pharmaceutical products into Guyana must ensure that all drugs are registered with the GA-FDD. The required documentation typically includes:
- Certificate of Pharmaceutical Product (CPP): A document issued by the regulatory authority in the exporting country, confirming that the product is authorized for sale and meets the necessary quality standards.
- Certificate of Analysis (CoA): A document that provides detailed information on the product's composition and quality, confirming that it meets the specified standards.
- Good Manufacturing Practice (GMP) Certificate: A certificate indicating that the manufacturing facility complies with GMP standards, ensuring the product's quality and safety.
Additionally, all labeling must be in English and include relevant information as stipulated by the GA-FDD.
Adhering to these documentation requirements is essential for the successful importation and sale of pharmaceutical products in Guyana.
Product Categories & Therapeutic Trends — India to Guyana
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Guyana are predominantly concentrated in the therapeutic areas of cardiovascular diseases, infectious diseases, and immunology. The leading product, "PHARMACEUTICAL PRODUCT FOR THE HUMAN CON," accounts for 39.1% of the total export value, indicating a significant demand for essential medications. Additionally, products like clopidogrel 75mg tablets and rifampicin capsules, which are used in the treatment of cardiovascular and infectious diseases respectively, contribute to 1.7% and 0.7% of the export value. This suggests that these therapeutic categories are well-represented in the export portfolio.
The dominance of these categories aligns with Guyana's healthcare needs, where non-communicable diseases (NCDs) such as cardiovascular conditions are prevalent. The Ministry of Health in Guyana has identified NCDs as major contributors to morbidity and mortality, emphasizing the need for effective management and treatment strategies. (health.gov.gy) Infectious diseases, including tuberculosis and malaria, continue to pose significant public health challenges, necessitating a steady supply of effective antimicrobial agents. Immunological treatments are also crucial, given the country's efforts to strengthen health resilience and address emerging health threats.
2Emerging Opportunities
The pharmaceutical landscape in Guyana presents emerging opportunities for Indian exporters, particularly in the realm of biosimilars and the potential for generic drugs following patent expirations. The global pharmaceutical market is witnessing substantial growth, with projections indicating a compound annual growth rate (CAGR) of approximately 7% by 2032. This growth is driven by increasing demand for both generic and biosimilar products, as healthcare systems seek cost-effective alternatives to branded medications.
In Guyana, the government's focus on modernizing healthcare infrastructure and expanding access to essential medicines creates a conducive environment for the introduction of biosimilars and generics. The establishment of a Digital Health Training Institute and the expansion of health services indicate a commitment to improving healthcare delivery, which may include the adoption of cost-effective pharmaceutical solutions. (dpi.gov.gy)
3Demand Drivers
Guyana's disease burden, characterized by a high prevalence of non-communicable diseases (NCDs) and infectious diseases, significantly drives the demand for pharmaceutical imports. NCDs, including cardiovascular diseases, diabetes, and cancer, account for a substantial portion of the country's health challenges. The Ministry of Health has implemented programs to address these issues, focusing on prevention, detection, control, and treatment. (health.gov.gy)
The government's commitment to enhancing healthcare infrastructure, as evidenced by the allocation of $161.1 billion in the 2026 budget for modernizing hospitals and expanding digital care, further stimulates the demand for pharmaceutical products. (dpi.gov.gy) Additionally, the establishment of the Guyana One Health Project, which integrates human, animal, and environmental health systems, underscores the country's proactive approach to managing health emergencies and controlling infectious diseases.
These initiatives, coupled with an increasing healthcare budget and a focus on comprehensive health strategies, are expected to sustain and potentially increase the demand for pharmaceutical imports from India, particularly in the areas of NCDs and infectious diseases.
Trade Policy & Tariff Intelligence — India and Guyana
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
As of March 2026, Guyana's Most Favored Nation (MFN) tariff rates for pharmaceutical products classified under Harmonized System (HS) codes 3003 and 3004 are not publicly disclosed. This lack of transparency complicates the assessment of import duties on finished pharmaceutical formulations entering Guyana. Importers are advised to consult Guyana's National Drug Regulatory Authority (NDRA) for the most current and detailed tariff information.
Regarding preferential duty rates, there is no publicly available information indicating that Guyana has established preferential tariff rates for pharmaceutical imports from India under any trade agreements. Consequently, Indian pharmaceutical exporters may face standard MFN rates without the benefit of reduced tariffs.
In terms of trade defense measures, there is no evidence to suggest that Guyana has imposed anti-dumping or countervailing duties on Indian pharmaceutical products. This absence of trade remedies indicates a stable trade environment for Indian pharmaceutical exports to Guyana.
2Trade Agreements
Currently, there is no publicly available information indicating the existence of a Free Trade Agreement (FTA) or any bilateral trade agreements between India and Guyana that specifically address pharmaceutical trade. The absence of such agreements means that Indian pharmaceutical exporters do not benefit from preferential tariff rates or other trade facilitation measures that might be available under such agreements. Without current or upcoming negotiations for such agreements, the trade relationship remains governed by standard MFN provisions.
3IP & Patent Landscape
Guyana's intellectual property (IP) framework, particularly concerning patents and data exclusivity, is not extensively documented in publicly available sources. This lack of transparency makes it challenging to assess the specific provisions affecting the importation of generic drugs from India. However, in the absence of stringent IP protections, Indian generic pharmaceutical companies may find opportunities to enter the Guyanese market more readily. Nonetheless, the lack of clear IP guidelines also introduces potential risks related to patent disputes or regulatory challenges. Therefore, Indian exporters should exercise due diligence and consider seeking legal counsel to navigate the IP landscape effectively.
Supply Chain & Logistics — India to Guyana Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Guyana primarily utilize sea and air freight, each with distinct routes and transit times. Sea freight typically involves transporting goods from major Indian ports to Georgetown, Guyana's capital. The journey encompasses transshipment points, often in the Caribbean or Central America, with total transit times ranging from 20 to 45 days, depending on specific routes and logistical factors.
Air freight offers a faster alternative, with direct flights from Indian airports to Cheddi Jagan International Airport in Georgetown. This mode significantly reduces transit times, making it suitable for urgent deliveries. However, air freight is generally more expensive than sea freight.
Recent disruptions in the Red Sea have impacted global shipping routes, potentially affecting transit times and costs for sea freight. While specific data on the extent of these disruptions is limited, it's advisable for stakeholders to monitor the situation closely and consult with logistics providers for the most current information.
2Port Infrastructure
In India, key export ports handling pharmaceutical shipments include Nhava Sheva Sea Port (INNSA1) and Sahar Air Cargo Complex. Nhava Sheva, located near Mumbai, is one of the busiest ports in India, offering extensive facilities for containerized cargo. Sahar Air Cargo Complex, situated in Mumbai, is a major hub for air freight, equipped with modern facilities to handle a variety of cargo types, including pharmaceuticals.
In Guyana, the primary import port is Georgetown, which serves as the main entry point for goods entering the country. The port is equipped to handle various types of cargo, including pharmaceuticals, and is supported by customs facilities to facilitate efficient clearance processes.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical formulations during transit is paramount. Cold chain logistics involve a series of temperature-controlled supply chain processes, including refrigerated storage and transportation, to ensure product efficacy upon arrival. Compliance with Good Distribution Practice (GDP) guidelines is essential, as outlined by the World Health Organization (WHO), to ensure that pharmaceutical products are consistently stored, transported, and handled under suitable conditions.
Packaging standards play a crucial role in cold chain logistics. Utilizing validated packaging solutions that can maintain the required temperature ranges for the duration of the transit is critical. For instance, reusable temperature-controlled pallet shippers are designed to provide high-performance, sustainable, and systematic solutions for long-duration cross-border pharmaceutical shipments.
Adhering to these standards ensures that pharmaceutical products arrive in Guyana in optimal condition, ready for distribution and use.
Market Opportunity Assessment — Guyana for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Guyana's pharmaceutical market is projected to generate a revenue of approximately $59.4 million USD in 2025, with an expected annual growth rate of 5.76% from 2025 to 2030, reaching a market volume of $78.58 million USD by 2030. This growth is driven by increasing healthcare demands and government initiatives aimed at improving public health.
The market is characterized by a significant reliance on imported pharmaceutical products, with generic drugs playing a crucial role due to their affordability and accessibility. The government's focus on universal health coverage and the introduction of the Universal Health Voucher Programme in March 2025, which provides citizens with a $10,000 voucher for medical tests, are expected to further stimulate demand for pharmaceutical products, including generics. (health.gov.gy)
2Healthcare System
Guyana operates a universal healthcare system, offering free public health services to all citizens and residents. However, certain specialized services, including diagnostic tests, are not covered under the public free care program. To address this, the government introduced the Universal Health Voucher Programme in March 2025, providing a $10,000 voucher to every citizen for essential medical tests, thereby enhancing access to healthcare services. (health.gov.gy)
The government has also signed Memorandums of Understanding (MOUs) with 75 private healthcare agencies to facilitate the rollout of voucher programs, ensuring comprehensive medical services are accessible to citizens across the country. (health.gov.gy)
3Opportunity for Indian Exporters
The growing demand for affordable and accessible healthcare in Guyana presents significant opportunities for Indian pharmaceutical exporters. With a total export value of $29.1 million USD in 2022, Indian exporters have established a strong presence in the Guyanese market, supplying a diverse range of finished pharmaceutical formulations. The top 10 products exported include essential medications such as clopidogrel 75mg tablets, human normal immunoglobulin, rifampicin capsules, and pantoprazole injections.
The introduction of the Universal Health Voucher Programme is expected to further increase the demand for pharmaceutical products, particularly generics, aligning with India's strengths in this segment. Additionally, the government's efforts to achieve universal health coverage and improve healthcare infrastructure indicate a favorable environment for expanding pharmaceutical exports. The growth forecast for Indian pharmaceutical imports into Guyana from 2025 to 2028 is positive, driven by these developments and the increasing need for quality healthcare products.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Guyana
Competing origins, India's edge, challenges and threats
1Competing Origins
India is a significant supplier of finished pharmaceutical formulations to Guyana, with exports totaling $29.1 million USD over 9,845 shipments from 2022 to 2026. This positions India as a key player in Guyana's pharmaceutical market. Other notable suppliers include domestic manufacturers like New GPC Inc., which has been a major contributor to the Caribbean's pharmaceutical industry since the 1920s, and international distributors such as Global Healthcare Supplies Inc., which has been supplying Guyana with medical equipment and pharmaceuticals since 2012. While specific market share data for China, EU manufacturers, and other emerging suppliers is limited, India's substantial export figures suggest a dominant position in the market.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages in supplying Guyana:
- Cost Competitiveness: Indian manufacturers provide high-quality pharmaceuticals at competitive prices, making them attractive to cost-conscious buyers in Guyana.
- Regulatory Compliance: Many Indian pharmaceutical companies adhere to international standards, including WHO-GMP, ensuring product quality and safety.
- English-Speaking Workforce: The widespread use of English in India facilitates clear communication and efficient business transactions with Guyanese partners.
- Broad Product Portfolio: Indian exporters offer a wide range of pharmaceutical products, catering to diverse medical needs in Guyana.
3Challenges & Threats
Despite its strengths, India's pharmaceutical exports to Guyana face several challenges:
- Regulatory Tightening: Changes in Guyana's regulatory environment could impact the approval and importation of foreign pharmaceuticals.
- Competition from China: Chinese pharmaceutical companies are increasingly entering the Caribbean market, potentially offering lower-priced alternatives.
- Quality Perception: Ensuring consistent product quality is crucial, as any lapses could affect India's reputation in the Guyanese market.
Addressing these challenges is essential for maintaining and enhancing India's position in Guyana's pharmaceutical sector.
FAQ — India to Guyana Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Guyana?
India exported pharmaceuticals worth $29.1M to Guyana across 9,845 verified shipments.
Who are the top Indian pharmaceutical exporters to Guyana?
1. SG HEALTHCARE PRIVATE LIMITED — $11.5M. 2. RELIANCE LIFE SCIENCES PRIVATE LIMITED — $1.6M. 3. AASHIRWAD PHARMA INTERNATIONAL PRIVATE LIMITED — $1.4M. Total: 164 suppliers.
Which companies in Guyana import pharmaceuticals from India?
1. TO, — $11.5M. 2. NEW GPC INC — $4.8M. 3. TO THE ORDER — $1.7M. 174 buyers total.
What pharmaceutical products does India export most to Guyana?
1. Pharmaceutical Product for the Human Con ($11.4M, 39.1%); 2. Human Normal Immunoglobulin for Intravenous Administration B.p.5% Solution 100m… ($581.6K, 2.0%); 3. Ph.dru.& Med.: Clopidogrel 75mg Tablets(each Tablet Contains: Clopidogrel 75mg)… ($487.9K, 1.7%); 4. Human Normal Immunoglobulin for Intraven ($281.3K, 1.0%); 5. Drugs and Pharmaceuticals Harmlessmedicines Rifampicin Capsules BP 300 MG ($212.0K, 0.7%)
Which ports handle pharmaceutical shipments from India to Guyana?
Export: AHEMDABAD ICD, NHAVA SHEVA SEA (INNSA1), SAHAR AIR, SAHAR AIR CARGO ACC (INBOM4), JNPT. Import: Georgetown, GEORGETOWN, GEORGR TOWN, OGLE, BARTICA.
Why does Guyana import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $29.1M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Guyana?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Guyana pharmaceutical trade?
$3.0K per consignment across 9,845 shipments.
How many Indian pharmaceutical companies export to Guyana?
164 Indian companies. Largest: SG HEALTHCARE PRIVATE LIMITED with $11.5M.
How can I find verified Indian pharmaceutical suppliers for Guyana?
TransData Nexus covers 164 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Guyana Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 9,845 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Guyana identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 9,845 verified shipments from 164 Indian exporters to 174 Guyana buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
164 Exporters
174 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists