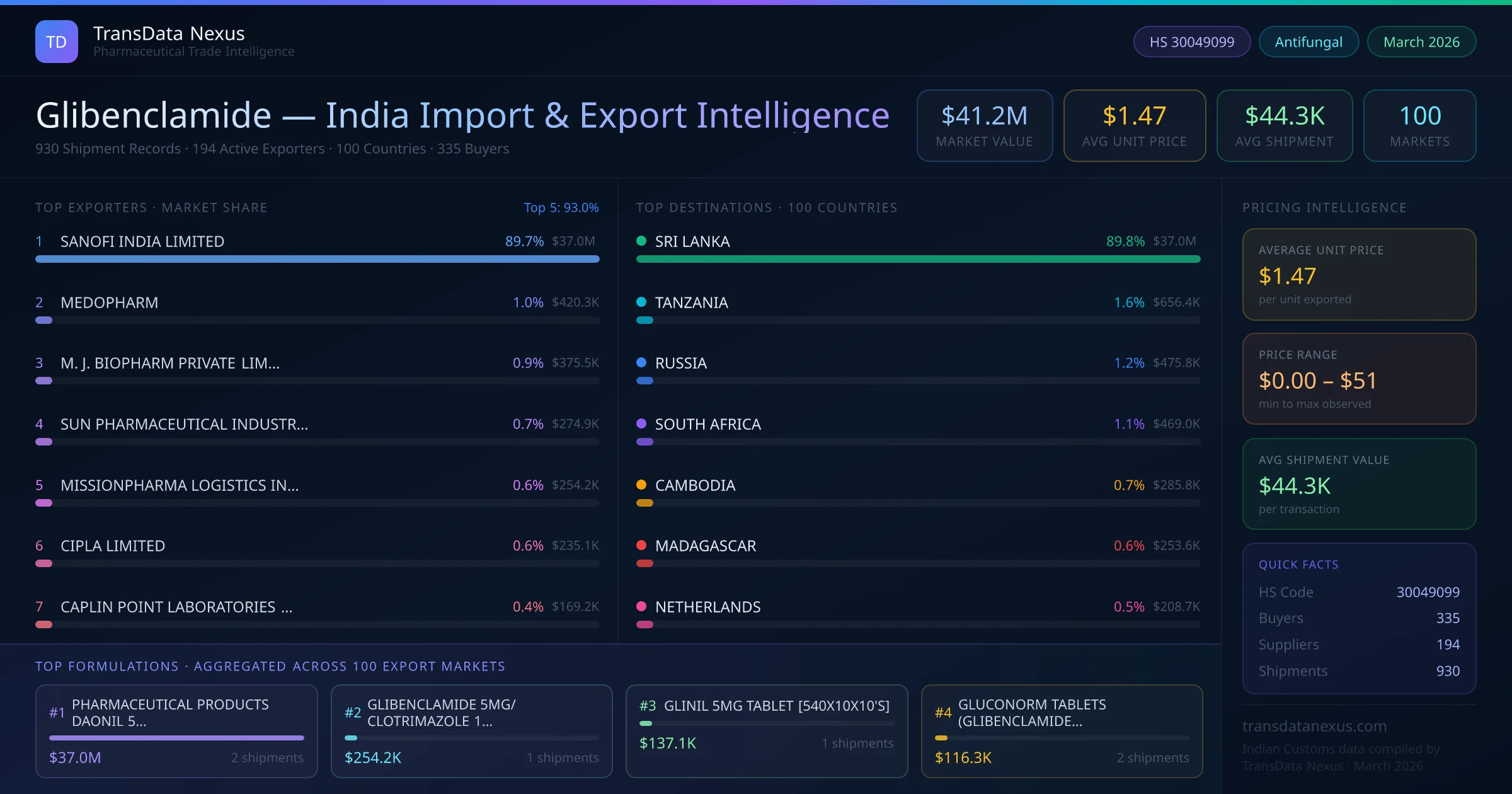
In June 2025, the NPPA revised the ceiling price for glibenclamide, reflecting changes in production costs and market dynamics. This adjustment aims to balance affordability for patients and sustainability for manufacturers.
In September 2025, the EMA updated its guidelines on GMP for active pharmaceutical ingredients (APIs), emphasizing stricter quality controls. Indian exporters must align with these updated standards to maintain access to the European market.
In December 2025, the WHO included glibenclamide in its updated Model List of Essential Medicines, reaffirming its importance in global diabetes management strategies.
In February 2026, the FDA issued a guidance document on the labeling requirements for generic drugs, including glibenclamide. This guidance aims to ensure consistency and clarity in drug information for healthcare providers and patients.
In March 2026, the Indian Ministry of Health and Family Welfare announced a policy initiative to streamline the export NOC process for pharmaceuticals, potentially reducing administrative burdens for exporters.
These developments underscore the dynamic nature of the pharmaceutical regulatory landscape, necessitating continuous monitoring and compliance by exporters to maintain market access and competitiveness.