In June 2025, the EMA's Pharmacovigilance Risk Assessment Committee (PRAC) identified non-arteritic anterior ischemic optic neuropathy (NAION) as a very rare side effect of GLP-1 receptor agonists, including empagliflozin-containing products. This finding necessitates updated safety information and increased vigilance among healthcare providers. (ema.europa.eu)
In July 2025, the EMA's CHMP adopted a positive opinion recommending the extension of the indication for Invokana (canagliflozin) to include treatment of children aged 10 years and older with type 2 diabetes mellitus. This decision reflects a broader trend of expanding indications for SGLT2 inhibitors, potentially influencing the market dynamics for empagliflozin. (ema.europa.eu)
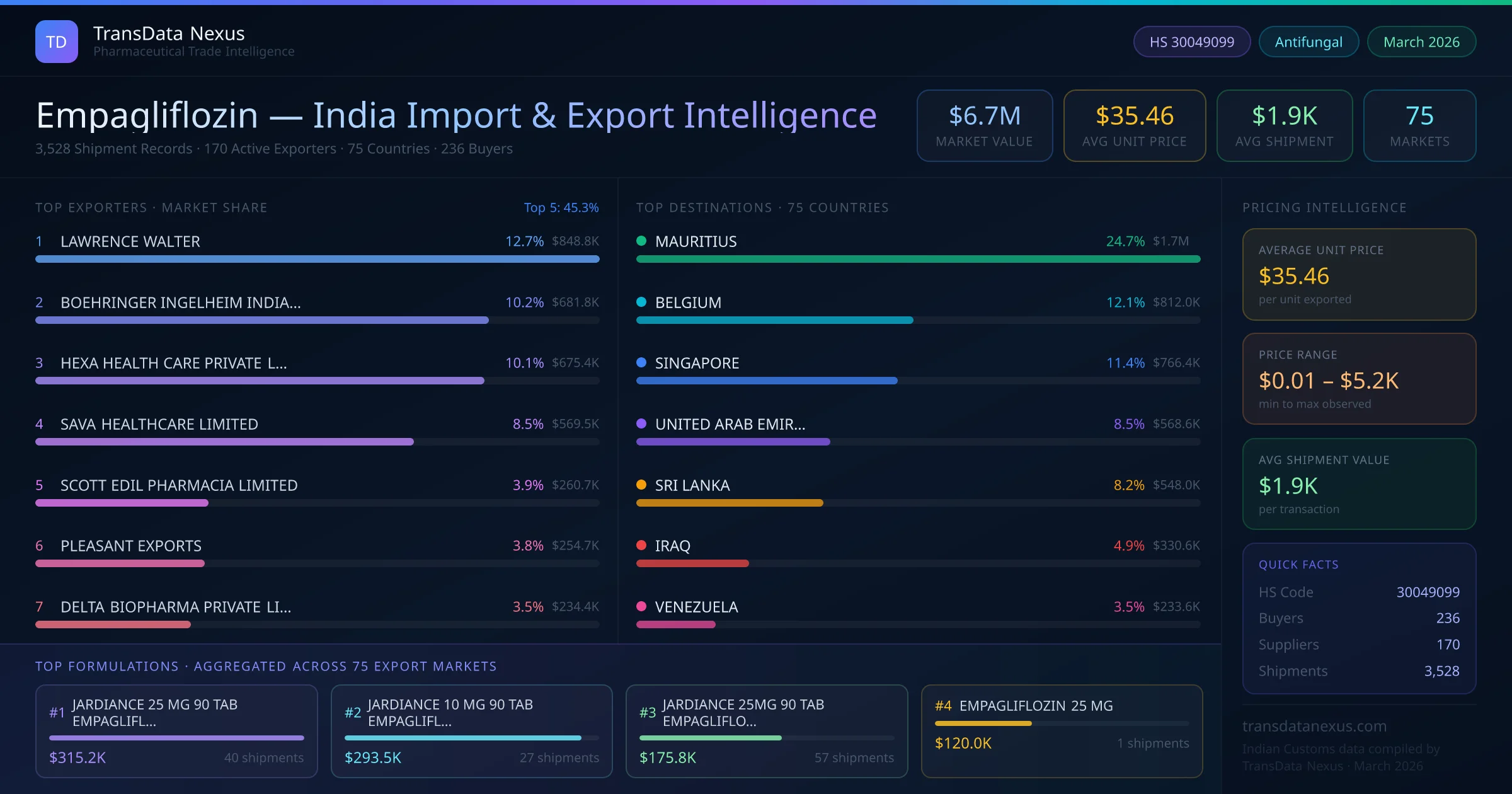
In November 2023, the EMA's CHMP adopted a positive opinion recommending an extension of the indication for Jardiance (empagliflozin) to include treatment of chronic kidney disease. This expansion underscores the evolving therapeutic applications of empagliflozin and may impact its market demand. (ema.europa.eu)
In August 2025, the FDA approved a new combination therapy involving empagliflozin and metformin for the treatment of type 2 diabetes mellitus. This approval introduces additional competition in the market, affecting Indian exporters.
In December 2025, the NPPA conducted a review of anti-diabetic drug prices, including empagliflozin, to assess market trends and ensure affordability. While no immediate price controls were implemented, the review indicates potential regulatory interventions that could impact pricing strategies for Indian exporters.