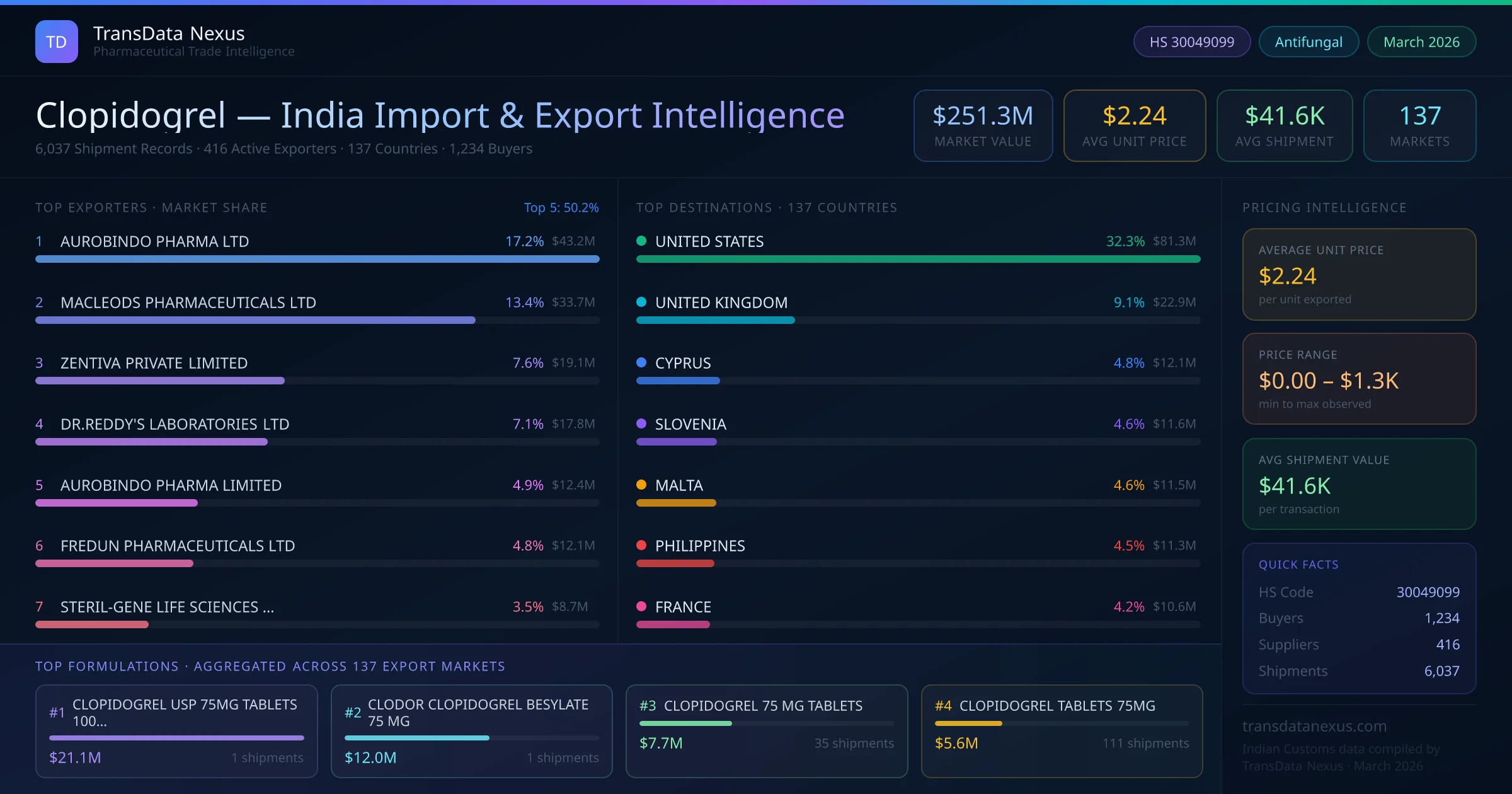
In July 2025, the NPPA revised the ceiling price for clopidogrel, reducing it by 5% to enhance affordability for patients. This adjustment reflects the authority's commitment to making essential medicines more accessible.
In October 2025, the EMA issued a new guideline on the bioequivalence requirements for generic clopidogrel products. This guideline aims to streamline the approval process for generics while ensuring therapeutic equivalence to the reference product.
In December 2025, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of clopidogrel. This update underscores the drug's continued importance in global health initiatives.
In February 2026, the FDA approved a new generic version of clopidogrel, further increasing competition in the U.S. market. This approval is expected to contribute to lower prices and improved patient access.
In March 2026, the CDSCO implemented stricter quality control measures for the manufacturing of clopidogrel, aiming to enhance product safety and efficacy. These measures include more rigorous inspections and compliance requirements for manufacturers.
These developments highlight the dynamic regulatory landscape surrounding clopidogrel, emphasizing the need for manufacturers and exporters to stay informed and compliant with evolving standards and policies.