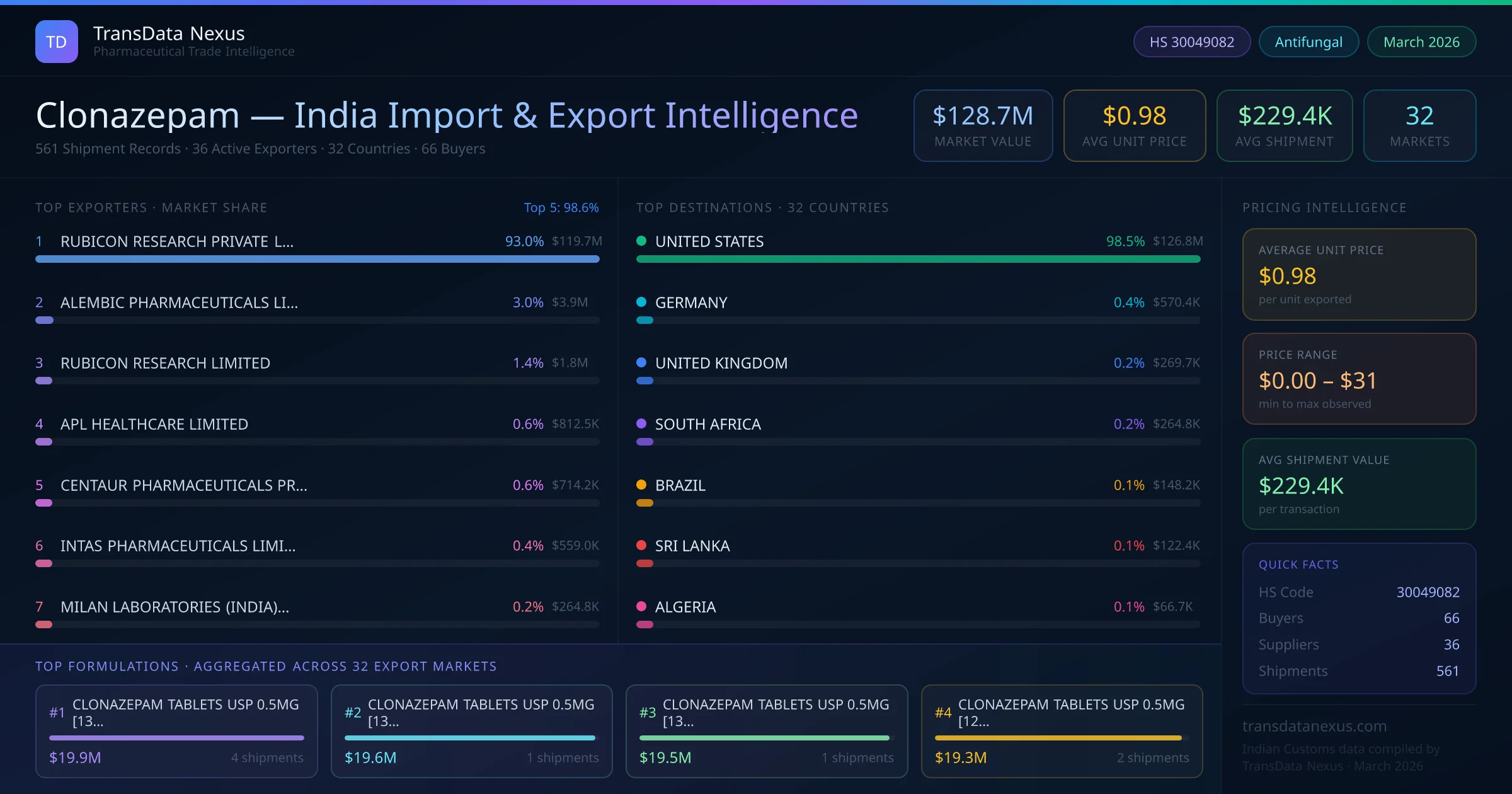
In January 2025, the European Medicines Agency (EMA) confirmed the recommendation to suspend certain medicines over flawed studies conducted by GVK Biosciences, a contract research organization based in India. While Clonazepam was not directly implicated, this development highlights the critical importance of compliance with Good Clinical Practice (GCP) standards for Indian exporters. (ema.europa.eu)
In March 2025, the U.S. Food and Drug Administration (FDA) revoked the orphan drug designation for Clonazepam for the treatment of hyperekplexia, a rare neurological disorder. This revocation indicates a shift in the regulatory status of Clonazepam for this specific indication.
In November 2025, the Indian government, through the NPPA, revised the ceiling price for Clonazepam formulations under the DPCO. This revision aims to balance affordability for patients while ensuring fair compensation for manufacturers.
In December 2025, the WHO updated its Model List of Essential Medicines, reaffirming the inclusion of Clonazepam. This reaffirms its essential status in the treatment of seizure disorders globally.
In February 2026, the EMA conducted a Periodic Safety Update Report Single Assessment (PSUSA) for Clonazepam, concluding maintenance of its authorization. This assessment underscores the ongoing monitoring of Clonazepam's safety profile within the EU. (ema.europa.eu)
These developments underscore the dynamic regulatory environment surrounding Clonazepam, emphasizing the need for continuous compliance and vigilance by manufacturers and exporters.