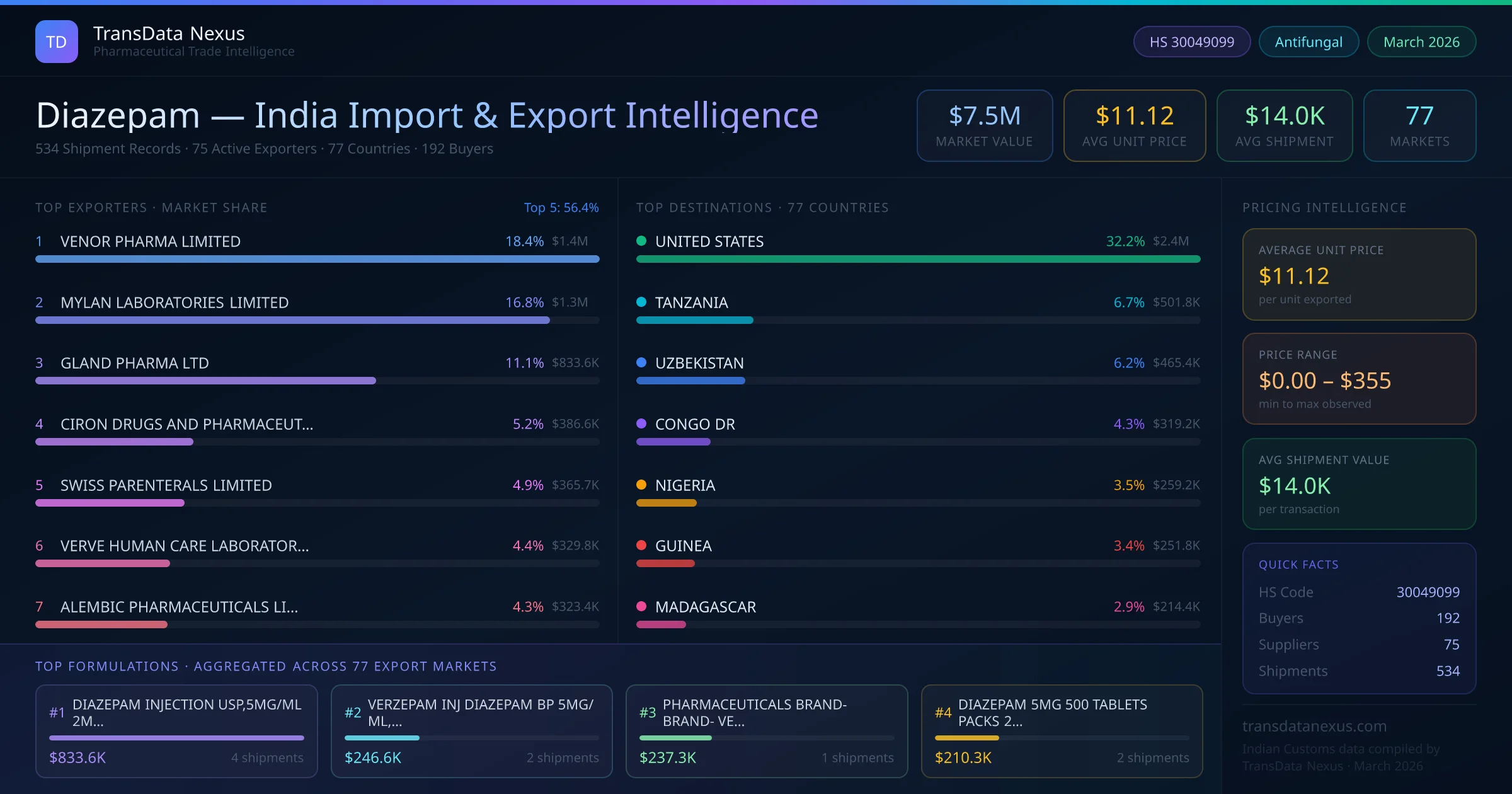
In May 2025, the FDA approved a generic version of Diastat AcuDial, a diazepam rectal gel, manufactured by Novel Labs Inc., expanding treatment options for acute repetitive seizures.
In September 2025, the WHO updated its Model List of Essential Medicines to its 24th edition, reaffirming the inclusion of diazepam in various formulations, highlighting its continued importance in global health. (who.int)
In November 2025, the FDA approved a generic version of Diastat, a diazepam rectal gel, manufactured by Navinta LLC, further increasing the availability of generic diazepam products in the U.S. market.
In December 2025, the WHO released updated guidelines on the management of acute convulsive seizures, emphasizing the use of non-parenteral routes for benzodiazepine administration, including rectal diazepam, when intravenous access is not available. (who.int)
In February 2026, the FDA approved a generic version of Valtoco, a diazepam nasal spray, expanding the range of administration routes available for diazepam.
These developments reflect ongoing efforts to enhance the accessibility and affordability of diazepam, both in the U.S. and globally, through the approval of generic formulations and the reinforcement of its essential status in medical treatment protocols.