In May 2024, Exelixis announced a settlement with Cipla Limited and Cipla USA regarding patent litigation over Cabometyx. This agreement permits Cipla to launch a generic version of cabozantinib in the U.S. market beginning January 1, 2031, following the expiration of the primary patent.
In April 2025, the World Health Organization (WHO) unveiled a global repository for National Essential Medicines Lists (nEMLs), compiling 150 national lists from six global regions. This initiative aims to enhance access to critical healthcare data, supporting policymakers and healthcare professionals in decision-making processes. (who.int)
In September 2025, the WHO published the 24th edition of its Model List of Essential Medicines, which did not include cabozantinib. The exclusion may reflect considerations such as cost-effectiveness, availability of alternative treatments, or specific public health priorities. (who.int)
In February 2026, the FDA updated its list of approved drugs, confirming that no generic versions of cabozantinib have been approved to date. This underscores the ongoing patent protection and the absence of generic competition in the U.S. market.
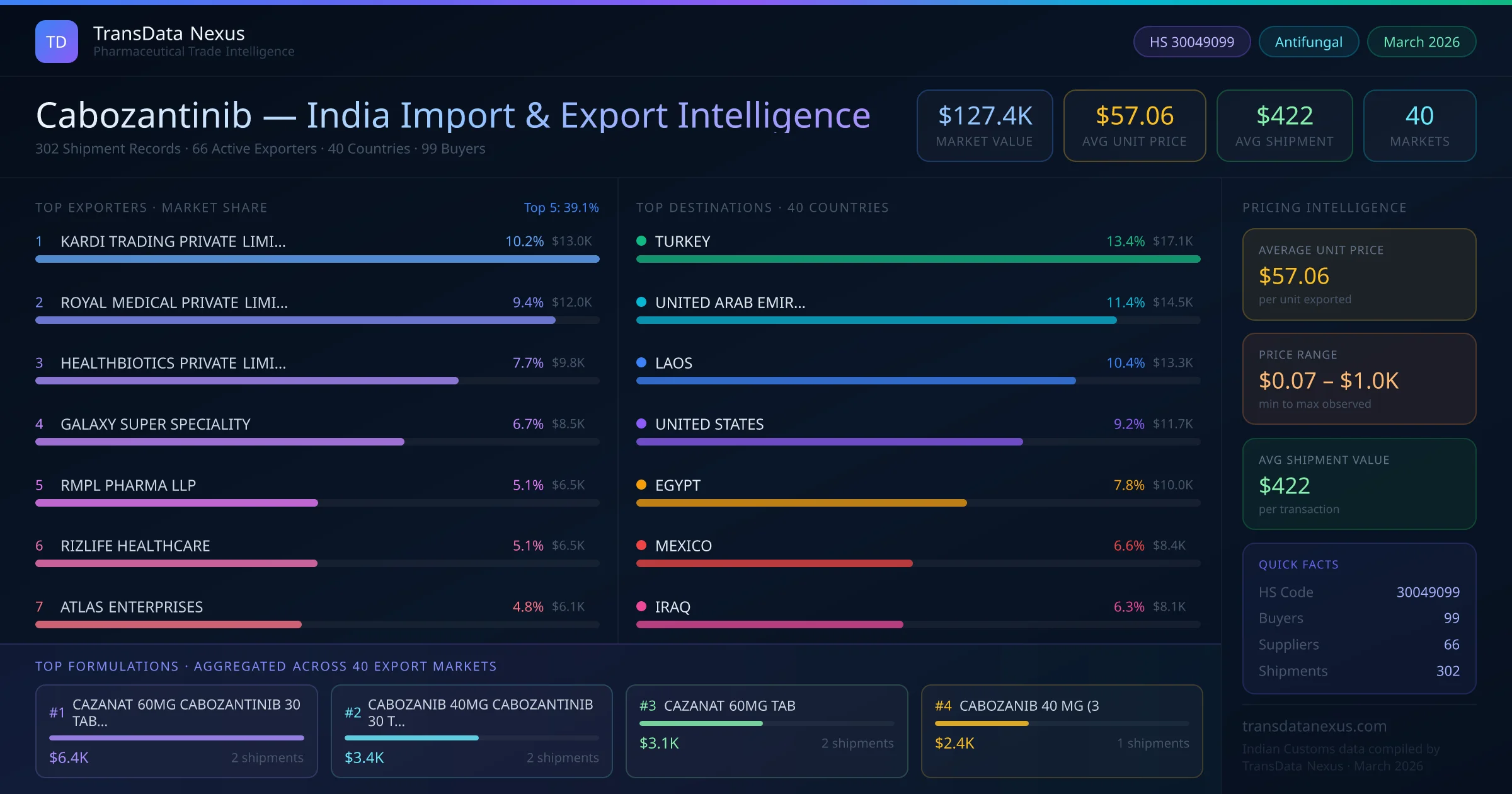
In March 2026, the Indian pharmaceutical export data indicated that cabozantinib exports totaled $0.1 million USD, with 302 shipment records and 66 active exporters. The primary destinations included Turkey (13.4%), United Arab Emirates (11.4%), Laos (10.4%), United States (9.2%), and Egypt (7.8%). The average Free on Board (FOB) unit price was $57.06, reflecting the competitive pricing strategies of Indian exporters.
These developments highlight the dynamic regulatory and market landscape for cabozantinib, influenced by patent protections, international regulatory decisions, and global health priorities.