TO THE Consignee-
Pharmaceutical Importer · Kenya · Biologics & Immunotherapy Focus · $95.1K Total Trade · DGFT Verified
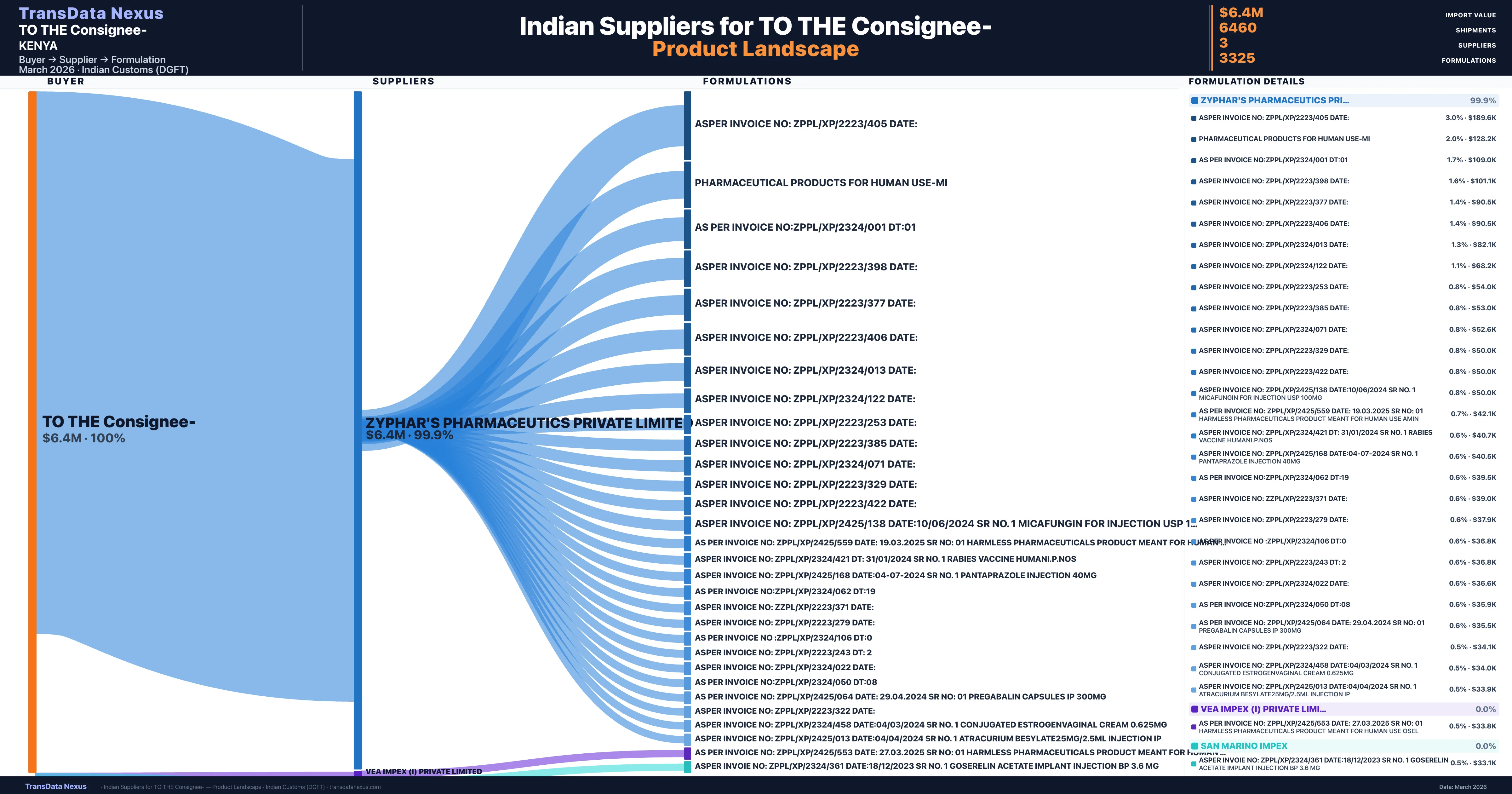
TO THE Consignee- is a pharmaceutical importer based in Kenya with a total trade value of $95.1K across 8 products in 4 therapeutic categories. Based on 46 verified import shipments from Indian Customs (DGFT) records, TO THE Consignee- is the #1 buyer in 1 product including Procarbazine. TO THE Consignee- sources from 3 verified Indian suppliers, with Zyphar's Pharmaceutics Private Limited accounting for 99.9% of imports.
TO THE Consignee- — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to TO THE Consignee-?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Zyphar's Pharmaceutics Private Limited | $6.4M | 6,316 | 99.9% |
| Vea Impex (i) Private Limited | $3.0K | 48 | 0.0% |
| San Marino Impex | $2.2K | 96 | 0.0% |
TO THE Consignee- sources from 3 verified Indian suppliers across 3,325 distinct formulations. The sourcing is highly concentrated — Zyphar's Pharmaceutics Private Limited accounts for 99.9% of total imports, indicating a strategic single-source relationship.
What Formulations Does TO THE Consignee- Import?
| Formulation | Value | Ships |
|---|---|---|
| Asper invoice no: zppl/xp/2223/405 | $189.6K | 4 |
| Pharmaceutical products for human | $128.2K | 171 |
| As per invoice no:zppl/xp/2324/001 | $109.0K | 165 |
| Asper invoice no: zppl/xp/2223/398 | $101.1K | 6 |
| Asper invoice no: zppl/xp/2223/377 | $90.5K | 34 |
| Asper invoice no: zppl/xp/2223/406 | $90.5K | 111 |
| Asper invoice no: zppl/xp/2324/013 | $82.1K | 3 |
| Asper invoice no: zppl/xp/2324/122 | $68.2K | 36 |
| Asper invoice no: zppl/xp/2223/253 | $54.0K | 15 |
| Asper invoice no: zppl/xp/2223/385 | $53.0K | 3 |
| Asper invoice no: zppl/xp/2324/071 | $52.6K | 3 |
| Asper invoice no: zppl/xp/2223/329 | $50.0K | 1 |
| Asper invoice no: zppl/xp/2223/422 | $50.0K | 1 |
| Asper invoice no: zppl/xp/2425/138 date:10/06/2024 sr no. 1 micafungin for injection USP 100MG | $50.0K | 1 |
| As per invoice no: zppl/xp/2425/559 date: 19.03.2025 sr no: 01 harmless pharmaceuticals product meant for human use | $42.1K | 1 |
TO THE Consignee- imports 3,325 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does TO THE Consignee- Import?
TO THE Consignee- Therapeutic Categories — 4 Specializations
TO THE Consignee- imports across 4 therapeutic categories, with Biologics & Immunotherapy (57.2%), Advanced Oncology (22.4%), Antimalarial & Antiparasitic (18.9%) representing the largest segments. The portfolio is concentrated — top 5 products = 92% of total imports.
Biologics & Immunotherapy
2 products · 57.2% · $54.4K
Advanced Oncology
4 products · 22.4% · $21.3K
Antimalarial & Antiparasitic
1 products · 18.9% · $18.0K
Advanced Diabetes Medications
1 products · 1.5% · $1.5K
Import Portfolio — Top 8 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ustekinumab | Biologics & Immunotherapy | $46.6K | 2 | 3.7% | 4 |
| 2 | Mefloquine | Antimalarial & Antiparasitic | $18.0K | 3 | 0.7% | 10 |
| 3 | Procarbazine | Advanced Oncology | $8.6K | 7 | 3.3% | 1 |
| 4 | Adalimumab | Biologics & Immunotherapy | $7.8K | 4 | 0.7% | 9 |
| 5 | Daunorubicin | Advanced Oncology | $6.4K | 7 | 0.9% | 9 |
| 6 | Lomustine | Advanced Oncology | $4.3K | 16 | 2.2% | 4 |
| 7 | Cabozantinib | Advanced Oncology | $2.0K | 5 | 1.5% | 7 |
| 8 | Saxagliptin | Advanced Diabetes Medications | $1.5K | 2 | 0.0% | 13 |
TO THE Consignee- imports 8 pharmaceutical products across 4 categories into Kenya totaling $95.1K. The company is the #1 buyer for 1 product: Procarbazine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for TO THE Consignee-.
Request DemoTO THE Consignee- — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
TO THE Consignee- is a pharmaceutical importer and buyer based in Kenya, specializing in the acquisition of finished pharmaceutical formulations from international suppliers. The company plays a pivotal role in the Kenyan pharmaceutical distribution network, ensuring the availability of a diverse range of medications to meet the country's healthcare needs. While specific details about its headquarters and parent company are not publicly disclosed, TO THE Consignee- is recognized for its significant contributions to the importation and distribution of pharmaceutical products within Kenya.
2Distribution Network
TO THE Consignee- operates a comprehensive distribution network across Kenya, facilitating the efficient delivery of pharmaceutical products to various healthcare providers, including hospitals, clinics, and pharmacies. The company's logistics capabilities are designed to ensure timely and reliable supply chains, adhering to the stringent standards set by the Pharmacy and Poisons Board (PPB) of Kenya. This network is integral to maintaining the availability of essential medicines throughout the country.
3Industry Role
In Kenya's pharmaceutical supply chain, TO THE Consignee- functions primarily as a wholesaler and importer. By sourcing finished pharmaceutical formulations from international suppliers, the company bridges the gap between global manufacturers and the Kenyan market, ensuring that a wide array of medications is accessible to meet the diverse healthcare needs of the population.
Supplier Relationship Intelligence — TO THE Consignee-
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
TO THE Consignee- demonstrates a high degree of sourcing concentration, with its top five imported products accounting for 91.9% of its total import value. This concentration indicates a strategic focus on specific high-demand medications, which can lead to cost efficiencies and streamlined operations. However, such dependency also poses risks, including potential supply chain disruptions if any of these key products face shortages or regulatory challenges. The company's reliance on a limited number of suppliers for these products suggests a need for robust contingency planning to mitigate potential risks associated with supplier dependency.
2Supply Chain Resilience
The resilience of TO THE Consignee-'s supply chain is closely tied to its sourcing strategy. The company's concentration on a select group of products and suppliers may limit its ability to adapt quickly to changes in the global pharmaceutical market. To enhance supply chain resilience, it would be prudent for TO THE Consignee- to diversify its supplier base and consider alternative sourcing options. Additionally, ensuring compliance with the PPB's regulations and maintaining strong relationships with suppliers can further strengthen the supply chain's robustness.
3Strategic Implications
The current sourcing pattern of TO THE Consignee- positions the company as a key player in the Kenyan pharmaceutical market, particularly in the importation of specific high-demand medications. For Indian exporters, this presents an opportunity to establish partnerships with TO THE Consignee- by offering products that align with the company's import profile. By understanding the therapeutic areas and specific products that TO THE Consignee- focuses on, Indian exporters can tailor their offerings to meet the company's needs, potentially gaining a foothold in the Kenyan market.
Importing Pharmaceuticals into Kenya — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Kenya
1Regulatory Authority & Framework
In Kenya, the Pharmacy and Poisons Board (PPB) serves as the primary regulatory authority overseeing the importation, distribution, and sale of pharmaceutical products. The PPB is responsible for ensuring that all pharmaceutical products meet the required standards of quality, safety, and efficacy before they are permitted for use in the country. Key legislation governing pharmaceutical imports includes the Pharmacy and Poisons Act, which outlines the procedures for drug registration, importation, and distribution. Additionally, the Pharmacy and Poisons (Registration of Health Products and Technologies) Rules provide detailed guidelines on the registration process for health products, including pharmaceuticals. These regulations are designed to protect public health by ensuring that only safe and effective medicines are available in the Kenyan market.
2Import Licensing & GMP
Importers of pharmaceutical products into Kenya are required to obtain an import license from the PPB. This license is issued upon the successful registration of the pharmaceutical product, which involves submitting a sample for evaluation and obtaining a certificate of analysis from a recognized laboratory. The PPB also mandates that imported pharmaceutical products comply with Good Manufacturing Practice (GMP) standards. While the PPB does not specify which GMP certifications are acceptable, it is generally understood that certifications from reputable authorities such as the World Health Organization (WHO) are recognized. Additionally, importers must possess a wholesale dealer's license, which is issued to registered pharmacists and is subject to annual renewal.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing and meet stability requirements as part of the registration process with the PPB. Labeling requirements include providing clear and accurate information on the product's name, active ingredients, dosage, and expiry date. The PPB specifies that labeling should be in English and Kiswahili, the official languages of Kenya. Serialization mandates may apply to certain products to enhance traceability and prevent counterfeit drugs from entering the market.
4Recent Regulatory Changes
In January 2025, the PPB issued a directive banning the importation, sale, and distribution of unregistered pharmaceutical products, particularly those referencing the Indian Pharmacopoeia. This move was aimed at curbing the influx of unregistered drugs into the Kenyan market and ensuring that all pharmaceutical products meet the country's regulatory standards. The directive emphasized that any pharmaceutical product not duly registered by the PPB is strictly prohibited for importation, distribution, sale, or handling in Kenya. (pulse.co.ke)
TO THE Consignee- — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
TO THE Consignee- focuses on importing pharmaceutical products across four therapeutic categories, with a significant emphasis on Biologics & Immunotherapy (57.2%), Advanced Oncology (22.4%), and Antimalarial & Antiparasitic (18.9%). This strategic focus aligns with the pressing healthcare needs in Kenya, where there is a high demand for treatments in these areas. The company's importation of specific products such as Ustekinumab, Mefloquine, and Procarbazine indicates a targeted approach to addressing prevalent health conditions in the country.
2Sourcing Profile
TO THE Consignee- primarily sources generic pharmaceutical products from India, focusing on finished formulations rather than active pharmaceutical ingredients (APIs) or bulk drugs. This sourcing strategy allows the company to offer a wide range of medications that are both cost-effective and readily available. India's well-established pharmaceutical manufacturing industry, known for its adherence to international quality standards, makes it a reliable source for these products.
3Market Positioning
Based on its product mix, TO THE Consignee- serves a broad segment of the Kenyan pharmaceutical market, including retail pharmacies, hospitals, and government tenders. The company's focus on high-demand therapeutic areas positions it as a key supplier of essential medicines, catering to the diverse healthcare needs of the Kenyan population.
Seller's Guide — How to Become a Supplier to TO THE Consignee-
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with TO THE Consignee- by offering products that align with the company's import profile, particularly in the therapeutic areas of Biologics & Immunotherapy, Advanced Oncology, and Antimalarial & Antiparasitic treatments. By understanding the specific products and formulations that TO THE Consignee- imports, Indian exporters can tailor their offerings to meet the company's needs, potentially gaining access to the Kenyan market.
2Requirements & Qualifications
Indian exporters seeking to supply TO THE Consignee- and the Kenyan market must ensure that their products are registered with the PPB and comply with the regulatory requirements set forth by the board. This includes obtaining the necessary import licenses, adhering to GMP standards recognized by the PPB, and ensuring that products meet the labeling and packaging requirements specified by the board.
3How to Approach
To establish a partnership with TO THE Consignee-, Indian exporters should initiate contact to understand the company's specific product needs and regulatory requirements. Participating in relevant tenders and trade fairs can also provide opportunities to showcase products. It is essential to ensure that all regulatory filings are completed accurately and submitted within the timelines set by the PPB. Building a strong relationship with TO THE Consignee- through consistent communication and demonstrating compliance with all regulatory standards will be crucial for a successful partnership.
Frequently Asked Questions — TO THE Consignee-
What products does TO THE Consignee- import from India?
TO THE Consignee- imports 8 pharmaceutical products across 4 categories. Top imports: Ustekinumab ($46.6K), Mefloquine ($18.0K), Procarbazine ($8.6K), Adalimumab ($7.8K), Daunorubicin ($6.4K).
Who supplies pharmaceuticals to TO THE Consignee- from India?
TO THE Consignee- sources from 3 verified Indian suppliers. The primary supplier is Zyphar's Pharmaceutics Private Limited (99.9% of imports, $6.4M).
What is TO THE Consignee-'s total pharmaceutical import value?
TO THE Consignee-'s total pharmaceutical import value from India is $95.1K, based on 46 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does TO THE Consignee- focus on?
TO THE Consignee- imports across 4 categories. The largest: Biologics & Immunotherapy (57.2%), Advanced Oncology (22.4%), Antimalarial & Antiparasitic (18.9%).
Get Full TO THE Consignee- Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: TO THE Consignee- identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as TO THE Consignee-'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 46 individual customs records matching TO THE Consignee-.
- 5.Supplier Verification: TO THE Consignee- sources from 3 verified Indian suppliers across 3,325 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.