Neoethicals, S.a,
Pharmaceutical Importer · Nicaragua · Analgesics & Antipyretics Focus · $4.5M Total Trade · DGFT Verified
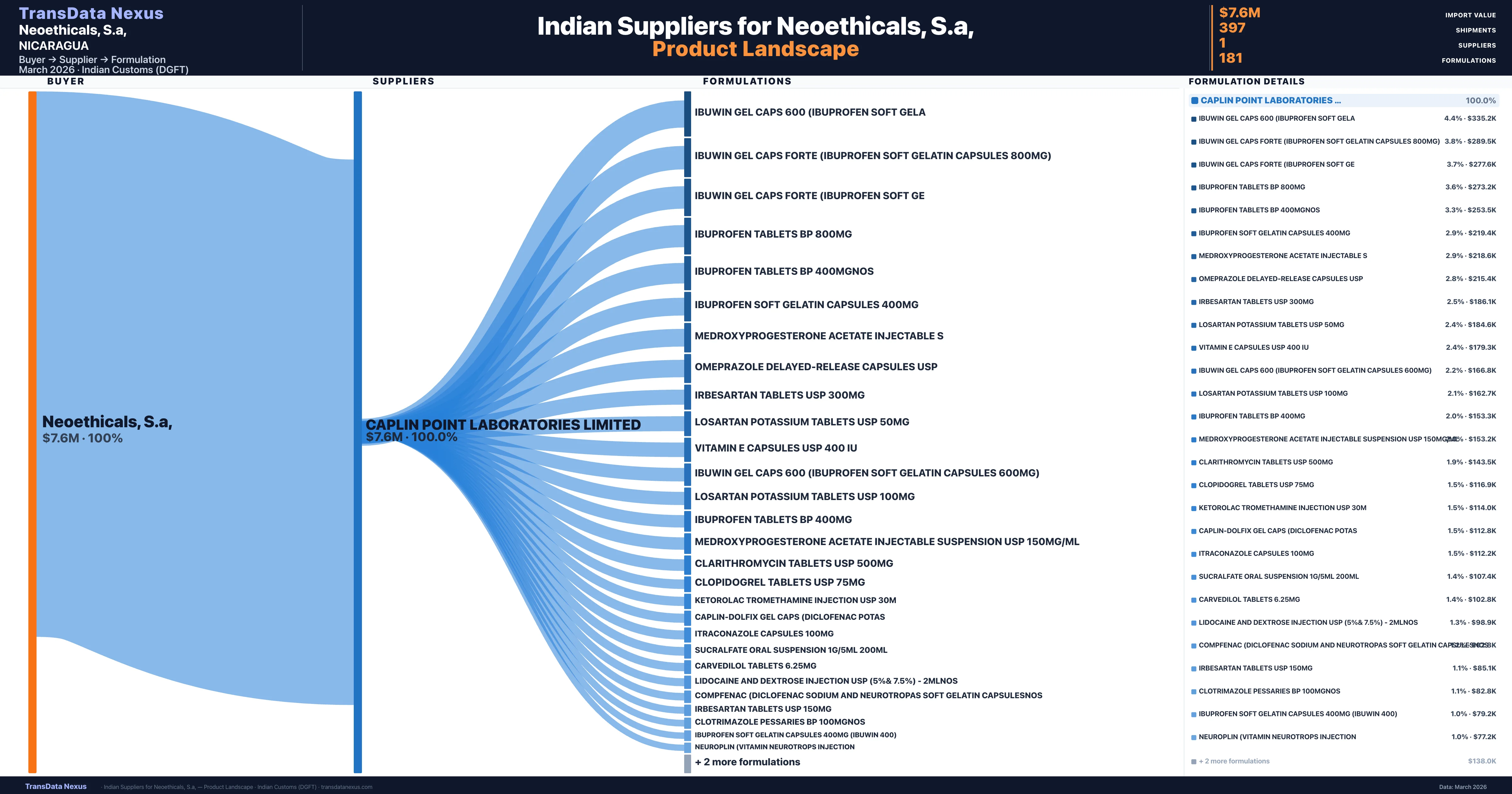
Neoethicals, S.a, is a pharmaceutical importer based in Nicaragua with a total trade value of $4.5M across 3 products in 3 therapeutic categories. Based on 89 verified import shipments from Indian Customs (DGFT) records, Neoethicals, S.a, is the #1 buyer in 1 product including Itraconazole. Neoethicals, S.a, sources from 1 verified Indian supplier, with Caplin Point Laboratories Limited accounting for 100.0% of imports.
Neoethicals, S.a, — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Neoethicals, S.a,?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Caplin Point Laboratories Limited | $7.6M | 397 | 100.0% |
Neoethicals, S.a, sources from 1 verified Indian supplier across 181 distinct formulations. The sourcing is highly concentrated — Caplin Point Laboratories Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Neoethicals, S.a, Import?
| Formulation | Value | Ships |
|---|---|---|
| Ibuwin gel CAPS 600 (ibuprofen soft | $335.2K | 8 |
| Ibuwin gel CAPS forte (ibuprofen soft gelatin capsules 800MG) | $289.5K | 8 |
| Ibuwin gel CAPS forte (ibuprofen soft | $277.6K | 6 |
| Ibuprofen tablets BP 800MG | $273.2K | 12 |
| Ibuprofen tablets BP | $253.5K | 12 |
| Ibuprofen soft gelatin capsules 400MG | $219.4K | 5 |
| Medroxyprogesterone acetate injectable | $218.6K | 5 |
| Omeprazole delayed-release capsules | $215.4K | 11 |
| Irbesartan tablets USP 300MG | $186.1K | 5 |
| Losartan potassium tablets USP 50MG | $184.6K | 9 |
| Vitamin e capsules USP 400 IU | $179.3K | 8 |
| Ibuwin gel CAPS 600 (ibuprofen soft gelatin capsules 600MG) | $166.8K | 6 |
| Losartan potassium tablets USP 100MG | $162.7K | 5 |
| Ibuprofen tablets BP 400MG | $153.3K | 10 |
| Medroxyprogesterone acetate injectable suspension USP 150MG/ML | $153.2K | 4 |
Neoethicals, S.a, imports 181 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Neoethicals, S.a, Import?
Top Products by Import Value
Neoethicals, S.a, Therapeutic Categories — 3 Specializations
Neoethicals, S.a, imports across 3 therapeutic categories, with Analgesics & Antipyretics (83.1%), Cardiovascular (13.5%), Antifungals (3.4%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Analgesics & Antipyretics
1 products · 83.1% · $3.7M
Cardiovascular
1 products · 13.5% · $600.0K
Antifungals
1 products · 3.4% · $150.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ibuprofen | Analgesics & Antipyretics | $3.7M | 74 | 0.6% | 4 |
| 2 | Irbesartan | Cardiovascular | $600.0K | 12 | 0.6% | 3 |
| 3 | Itraconazole | Antifungals | $150.0K | 3 | 0.5% | 1 |
Neoethicals, S.a, imports 3 pharmaceutical products across 3 categories into Nicaragua totaling $4.5M. The company is the #1 buyer for 1 product: Itraconazole.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Neoethicals, S.a,.
Request DemoNeoethicals, S.a, — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Neoethicals, S.A. is a Nicaraguan pharmaceutical importer and distributor headquartered in Managua, Nicaragua. The company specializes in sourcing and distributing high-quality pharmaceutical formulations, including tablets, capsules, syrups, and injections. Their mission is to enhance public health by providing innovative and reliable medical treatments. Neoethicals, S.A. operates as an independent entity without a publicly known parent company, playing a pivotal role in Nicaragua's pharmaceutical supply chain by ensuring the availability of essential medications. (neoethicals.online.com.ni)
2Distribution Network
Neoethicals, S.A. maintains its primary distribution center at Bodega Rap 8, Reparto Ulsa, Entrada del Instituto Ramirez Goyena, 1c. al Oeste 200 metros al Sur, Managua, Nicaragua. This strategic location facilitates efficient logistics and timely delivery of pharmaceutical products across the country. While specific details about additional warehouse locations and logistics capabilities are not publicly disclosed, the company's centralized operations suggest a focus on optimizing distribution within Nicaragua.
3Industry Role
In Nicaragua's pharmaceutical supply chain, Neoethicals, S.A. functions primarily as a wholesaler and distributor. By importing finished pharmaceutical formulations from international suppliers, the company ensures that a diverse range of medications is accessible to healthcare providers and patients throughout the nation. Their role is crucial in bridging the gap between global pharmaceutical manufacturers and the Nicaraguan market, contributing to the overall health infrastructure.
Supplier Relationship Intelligence — Neoethicals, S.a,
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Neoethicals, S.A. exhibits a high degree of supplier concentration, sourcing all its pharmaceutical imports from a single supplier: Caplin Point Laboratories Limited. Between 2022 and 2026, the company imported finished pharmaceutical formulations totaling $4.5 million USD across 89 shipments, with Caplin Point Laboratories Limited accounting for 100% of these imports. This exclusive sourcing strategy indicates a deep, strategic partnership between Neoethicals, S.A. and Caplin Point Laboratories Limited. While such concentration can lead to favorable terms and streamlined operations, it also poses risks related to supply chain disruptions, pricing fluctuations, and dependency on a single supplier's stability.
2Supply Chain Resilience
The resilience of Neoethicals, S.A.'s supply chain is closely tied to its exclusive relationship with Caplin Point Laboratories Limited. This singular sourcing approach may limit the company's ability to quickly adapt to supply chain disruptions or changes in market conditions. The lack of publicly available information on backup suppliers and the diversity of formulations imported suggests a potential vulnerability in the supply chain. Additionally, the exposure to shipping routes and the regulatory compliance of Caplin Point Laboratories Limited are critical factors influencing the overall resilience of Neoethicals, S.A.'s supply chain.
3Strategic Implications
Neoethicals, S.A.'s exclusive sourcing from Caplin Point Laboratories Limited positions the company to benefit from strong supplier relationships and potentially favorable terms. However, this strategy also exposes Neoethicals, S.A. to risks associated with supplier dependency, such as supply chain disruptions or pricing volatility. For Indian pharmaceutical exporters, this presents an opportunity to diversify Neoethicals, S.A.'s supplier base by offering competitive products and establishing relationships that could mitigate the risks associated with single-source dependency.
Importing Pharmaceuticals into Nicaragua — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Nicaragua
1Regulatory Authority & Framework
In Nicaragua, the Ministry of Health (MINSA) serves as the primary regulatory authority overseeing the importation, registration, and distribution of pharmaceutical products. The key legislation governing pharmaceutical imports includes the Medicines and Pharmacy Law (Law 292) and the General Health Law (Law 423). These laws establish the framework for ensuring the safety, efficacy, and quality of pharmaceutical products entering the Nicaraguan market. The marketing authorization pathway for Indian generics involves obtaining sanitary registration from MINSA, which requires comprehensive documentation demonstrating the product's safety and effectiveness.
2Import Licensing & GMP
Importers of pharmaceutical products into Nicaragua must obtain import permits from MINSA's Pharmaceutical Office. This process involves presenting documentation that demonstrates the safety and effectiveness of the products, as well as paying fees for sanitary registration and laboratory analysis. The recognition of Good Manufacturing Practice (GMP) certificates, such as EU GMP, WHO GMP, or PIC/S, is essential for the approval of pharmaceutical imports. Additionally, importers must secure wholesale distribution authorization to operate legally within the Nicaraguan market.
3Quality & Labeling
Pharmaceutical products imported into Nicaragua are subject to batch testing and stability requirements to ensure their safety and efficacy. Labeling must comply with specific regulations, including the use of the Spanish language and adherence to established standards for information presentation. Serialization mandates may also be in place to enhance traceability and prevent counterfeit products from entering the market. These quality and labeling standards are enforced by MINSA to protect public health and maintain the integrity of the pharmaceutical supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, Nicaragua's pharmaceutical regulatory landscape has seen several developments. Notably, in March 2024, the National Health Regulation Authority (ANRS) issued Circular REF ANRS – DLCC FRUG001 2024, which introduced new procedures for the renewal of pharmaceutical product registrations. This circular emphasizes the need for updated documentation and samples for analysis, impacting the renewal process for various pharmaceutical products. Such regulatory changes underscore the importance for importers and exporters to stay informed and compliant with evolving standards. (latinalliance.co)
Neoethicals, S.a, — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Neoethicals, S.A. focuses its product strategy on three primary therapeutic categories: Analgesics & Antipyretics, Cardiovascular, and Antifungals. The top three imported products—Ibuprofen, Irbesartan, and Itraconazole—align with these categories, indicating a strategic emphasis on addressing common health concerns prevalent in the Nicaraguan population. The substantial import value of Ibuprofen ($3.7 million USD) suggests a high demand for pain and fever management solutions, while the inclusion of cardiovascular and antifungal medications reflects a comprehensive approach to healthcare needs.
2Sourcing Profile
Neoethicals, S.A. employs a sourcing strategy that relies exclusively on Caplin Point Laboratories Limited for its pharmaceutical imports. This approach indicates a preference for established suppliers with a proven track record in delivering quality products. The company's focus on finished pharmaceutical formulations, rather than raw Active Pharmaceutical Ingredients (APIs), suggests a strategy centered on ready-to-market products that meet the specific needs of the Nicaraguan market.
3Market Positioning
Based on its product mix, Neoethicals, S.A. serves a broad segment of the Nicaraguan pharmaceutical market, including retail pharmacies, hospitals, and wholesale distribution channels. The company's diverse portfolio, encompassing analgesics, cardiovascular drugs, and antifungals, positions it to meet a wide range of healthcare needs, thereby enhancing its market presence and competitiveness.
Seller's Guide — How to Become a Supplier to Neoethicals, S.a,
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter the Nicaraguan market by partnering with Neoethicals, S.A. The company's exclusive sourcing from Caplin Point Laboratories Limited indicates a potential openness to diversifying its supplier base. Indian exporters can leverage this opportunity by offering competitive products that align with Neoethicals, S.A.'s therapeutic focus and by demonstrating compliance with Nicaraguan regulatory standards.
2Requirements & Qualifications
Indian exporters aiming to supply Neoethicals, S.A. and the Nicaraguan market must ensure their products meet the regulatory requirements set by MINSA. This includes obtaining sanitary registration, providing comprehensive documentation on product safety and efficacy, and adhering to GMP standards recognized by Nicaraguan authorities. Additionally, compliance with labeling and packaging regulations, including the use of the Spanish language and adherence to established standards, is essential.
3How to Approach
To establish a relationship with Neoethicals, S.A., Indian exporters should initiate contact by presenting their product portfolios and demonstrating compliance with Nicaraguan regulatory standards. Participating in relevant tenders and industry events in Nicaragua can enhance visibility and facilitate connections. Developing a clear regulatory filing strategy, including obtaining necessary certifications and registrations, is crucial. Given the regulatory processes in Nicaragua, exporters should anticipate a timeline of several months for product registration and approval.
Frequently Asked Questions — Neoethicals, S.a,
What products does Neoethicals, S.a, import from India?
Neoethicals, S.a, imports 3 pharmaceutical products across 3 categories. Top imports: Ibuprofen ($3.7M), Irbesartan ($600.0K), Itraconazole ($150.0K).
Who supplies pharmaceuticals to Neoethicals, S.a, from India?
Neoethicals, S.a, sources from 1 verified Indian suppliers. The primary supplier is Caplin Point Laboratories Limited (100.0% of imports, $7.6M).
What is Neoethicals, S.a,'s total pharmaceutical import value?
Neoethicals, S.a,'s total pharmaceutical import value from India is $4.5M, based on 89 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Neoethicals, S.a, focus on?
Neoethicals, S.a, imports across 3 categories. The largest: Analgesics & Antipyretics (83.1%), Cardiovascular (13.5%), Antifungals (3.4%).
Get Full Neoethicals, S.a, Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Neoethicals, S.a, identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Neoethicals, S.a,'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 89 individual customs records matching Neoethicals, S.a,.
- 5.Supplier Verification: Neoethicals, S.a, sources from 1 verified Indian suppliers across 181 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.