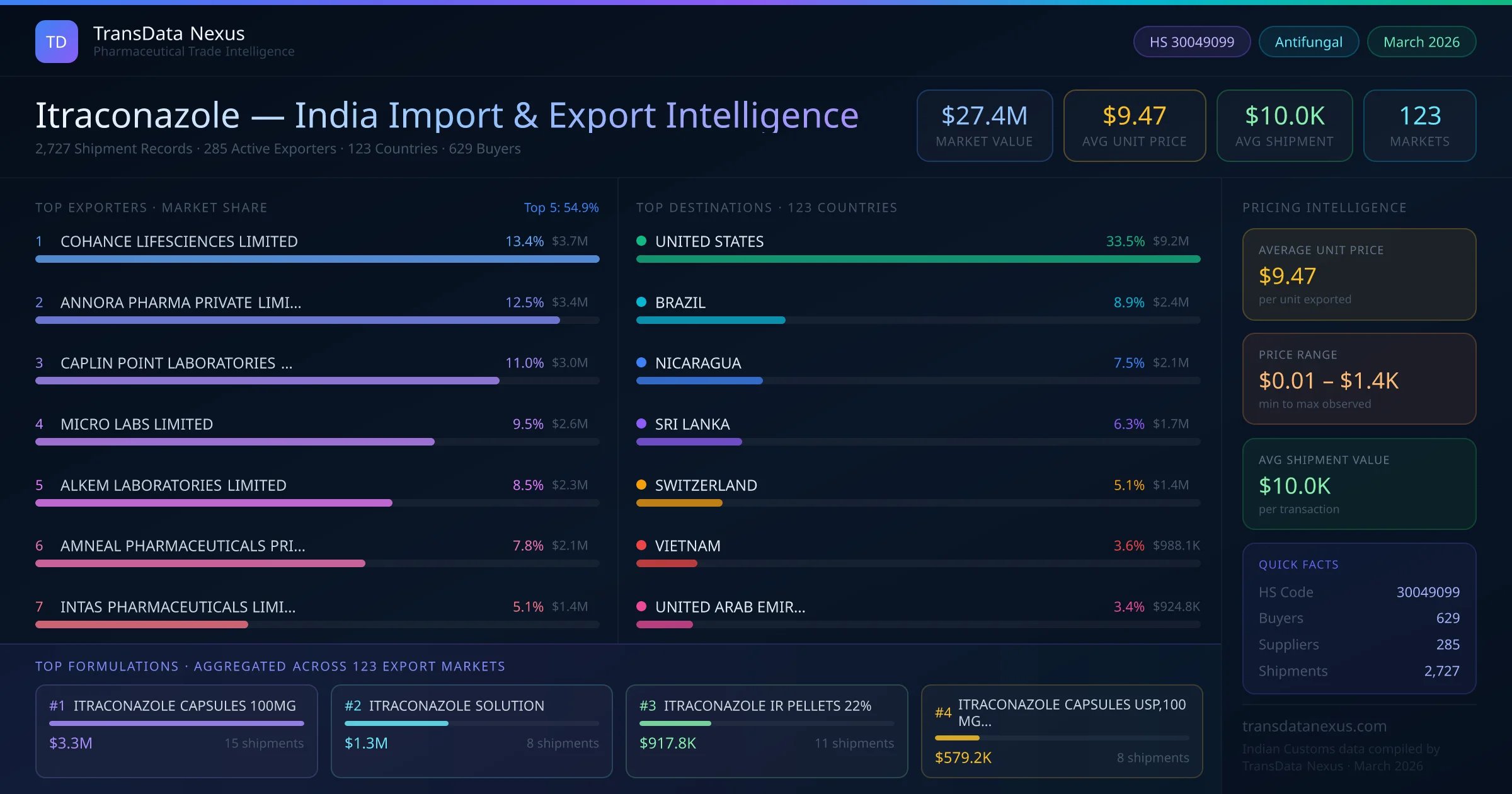
In January 2026, the EMA's Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion recommending an extension to the existing indication for Noxafil (posaconazole), a related antifungal agent, for the treatment of invasive aspergillosis in adults and pediatric patients from 2 years of age weighing more than 40 kg. (ema.europa.eu)
In July 2023, the EMA withdrew the orphan designation for itraconazole for the treatment of naevoid basal-cell carcinoma syndrome (Gorlin syndrome) at the sponsor's request. (ema.europa.eu)
In May 2018, the EMA granted orphan designation to itraconazole for the prevention of invasive aspergillosis, recognizing its potential in addressing this serious condition. (ema.europa.eu)
In April 2009, the EMA's CHMP concluded that the benefits of Prokanazol, an itraconazole-containing medicine, did not outweigh its risks, leading to the suspension of its marketing authorization in the Czech Republic. (ema.europa.eu)
In October 2005, the European Commission granted a marketing authorization for Posaconazole SP, an antifungal agent, for the treatment of various fungal infections in adults. However, the marketing authorization lapsed in November 2008 due to the product not being marketed within three years of authorization. (ema.europa.eu)
These developments underscore the dynamic nature of the antifungal pharmaceutical market and the importance of staying abreast of regulatory changes and market trends.