Ministry OF Health OF THE Republic
Pharmaceutical Importer · Zambia · Tuberculosis Medications Focus · $4.1M Total Trade · DGFT Verified
Ministry OF Health OF THE Republic is a pharmaceutical importer based in Zambia with a total trade value of $4.1M across 4 products in 2 therapeutic categories. Based on 82 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Ministry OF Health OF THE Republic sources from 5 verified Indian suppliers, with Macleods Pharmaceuticals Limited accounting for 75.0% of imports.
Ministry OF Health OF THE Republic — Import Portfolio & Supplier Network
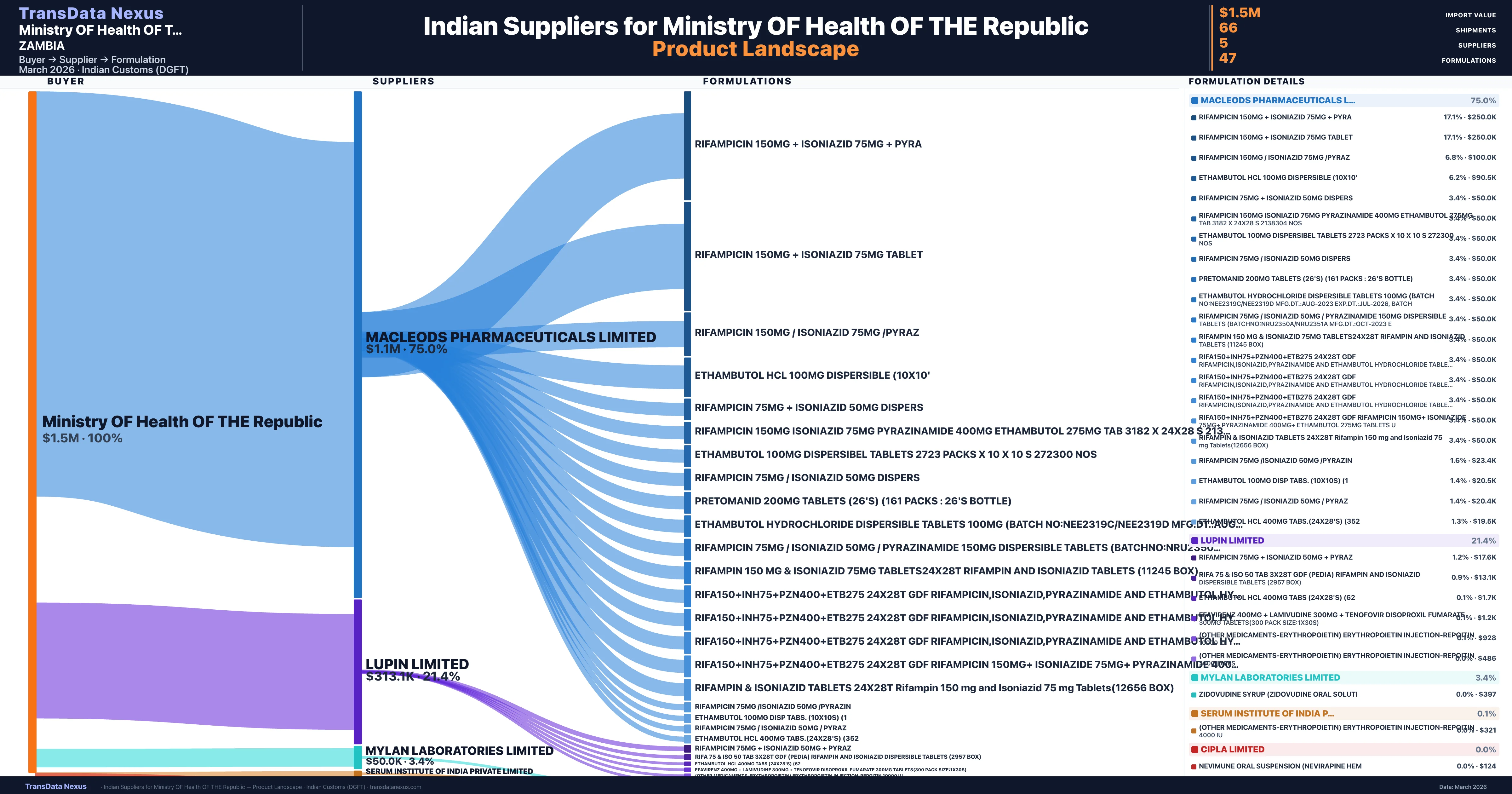
Who Are the Verified Indian Suppliers to Ministry OF Health OF THE Republic?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Macleods Pharmaceuticals Limited | $1.1M | 41 | 75.0% |
| Lupin Limited | $313.1K | 7 | 21.4% |
| Mylan Laboratories Limited | $50.0K | 1 | 3.4% |
| Serum Institute Of India Private Limited | $1.7K | 3 | 0.1% |
| Cipla Limited | $520 | 14 | 0.0% |
Ministry OF Health OF THE Republic sources from 5 verified Indian suppliers across 47 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does Ministry OF Health OF THE Republic Import?
| Formulation | Value | Ships |
|---|---|---|
| Rifampicin 150MG + isoniazid 75MG + | $250.0K | 5 |
| Rifampicin 150MG + isoniazid 75MG | $250.0K | 5 |
| Rifampicin 150MG / isoniazid 75MG | $100.0K | 2 |
| Ethambutol hcl 100MG dispersible | $90.5K | 2 |
| Rifampicin 75MG + isoniazid 50MG | $50.0K | 1 |
| Rifampicin 150MG isoniazid 75MG pyrazinamide 400MG ethambutol 275MG TAB 3182 x 24x28 s 2138304 | $50.0K | 1 |
| Ethambutol 100MG dispersibel tablets 2723 packs x 10 x 10 s 272300 | $50.0K | 1 |
| Rifampicin 75MG / isoniazid 50MG | $50.0K | 1 |
| Pretomanid 200MG tablets (26's) (161 packs : 26's bottle) | $50.0K | 1 |
| Ethambutol hydrochloride dispersible tablets 100MG (batch no:nee2319c/nee2319d mfg.dt.:aug-2023 exp.dt.:jul-2026 | $50.0K | 1 |
| Rifampicin 75MG / isoniazid 50MG / pyrazinamide 150MG dispersible tablets (batchno:nru2350a/nru2351a mfg.dt.:oct-2023 | $50.0K | 1 |
| Rifampin 150 MG & isoniazid 75MG tablets24x28t rifampin and isoniazid tablets (11245 Box) | $50.0K | 1 |
| Rifa150+inh75+pzn400+etb275 24x28t gdf rifampicin,isoniazid,pyrazinamide and ethambutol hydrochloride tablets USP(6231 | $50.0K | 1 |
| Rifa150+inh75+pzn400+etb275 24x28t gdf rifampicin,isoniazid,pyrazinamide and ethambutol hydrochloride tablets USP(4961 | $50.0K | 1 |
| Rifa150+inh75+pzn400+etb275 24x28t gdf rifampicin,isoniazid,pyrazinamide and ethambutol hydrochloride tablets USP(5846 | $50.0K | 1 |
Ministry OF Health OF THE Republic imports 47 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Ministry OF Health OF THE Republic Import?
Top Products by Import Value
Ministry OF Health OF THE Republic Therapeutic Categories — 2 Specializations
Ministry OF Health OF THE Republic imports across 2 therapeutic categories, with Tuberculosis Medications (56.1%), Advanced Antibiotics (43.9%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Tuberculosis Medications
3 products · 56.1% · $2.3M
Advanced Antibiotics
1 products · 43.9% · $1.8M
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Rifampicin | Advanced Antibiotics | $1.8M | 36 | 0.1% | 10 |
| 2 | Isoniazid | Tuberculosis Medications | $1.4M | 27 | 0.1% | 12 |
| 3 | Ethambutol | Tuberculosis Medications | $650.0K | 13 | 0.1% | 7 |
| 4 | Pyrazinamide | Tuberculosis Medications | $300.0K | 6 | 0.0% | 4 |
Ministry OF Health OF THE Republic imports 4 pharmaceutical products across 2 categories into Zambia totaling $4.1M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Ministry OF Health OF THE Republic.
Request DemoMinistry OF Health OF THE Republic — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
The Ministry of Health of the Republic of Zambia is the governmental body responsible for overseeing the nation's healthcare system, including the procurement and distribution of medical supplies. As a government entity, it operates without a parent company and plays a pivotal role in ensuring the availability of essential medicines and medical supplies across Zambia. Its operations encompass policy formulation, resource allocation, and the management of healthcare services to meet the needs of the Zambian population.
2Distribution Network
The Ministry of Health's distribution network is extensive, aiming to ensure equitable access to medical supplies throughout Zambia. While specific warehouse locations are not publicly detailed, the ministry collaborates with agencies like the Zambia Medicines and Medical Supplies Agency (ZAMMSA), which operates regional hubs in Mansa, Mpika, Chipata, Choma, Mongu, Kabompo, and Luanshya. These hubs are equipped with modern storage facilities, including cold storage for sensitive products, and are supported by advanced inventory systems and dedicated transportation fleets to facilitate timely delivery to health facilities, even in remote areas. (zammsa.co.zm)
3Industry Role
In Zambia's pharmaceutical supply chain, the Ministry of Health functions as the primary wholesaler and central procurement authority. It is responsible for sourcing, importing, and distributing medical supplies to public health institutions nationwide. This centralization ensures standardized procurement processes and equitable distribution of essential medicines, aligning with the ministry's mandate to provide quality healthcare services to all Zambians.
Supplier Relationship Intelligence — Ministry OF Health OF THE Republic
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The Ministry of Health's sourcing strategy reveals a significant concentration on a limited number of suppliers. Notably, 75% of the total import value from India is attributed to MACLEODS PHARMACEUTICALS LIMITED, with 41 shipments. This heavy reliance on a single supplier could pose risks related to supply chain disruptions, such as production delays or quality control issues. However, the consistent volume of shipments suggests a stable and ongoing relationship, indicating that the ministry may prioritize reliability and cost-effectiveness in its procurement decisions.
2Supply Chain Resilience
The Ministry of Health's supply chain resilience is closely tied to its dependence on MACLEODS PHARMACEUTICALS LIMITED for a substantial portion of its pharmaceutical imports. While this concentration can lead to efficiencies and cost savings, it also exposes the ministry to potential risks if the supplier faces operational challenges. The presence of additional suppliers, such as LUPIN LIMITED and MYLAN LABORATORIES LIMITED, provides a degree of diversification, but the overall supply chain could benefit from further diversification to enhance resilience. Ensuring that all suppliers adhere to international Good Manufacturing Practices (GMP) and maintaining robust quality assurance protocols are essential steps to mitigate potential risks.
3Strategic Implications
The Ministry of Health's concentrated sourcing pattern positions it to negotiate favorable terms with its primary supplier, potentially securing better pricing and consistent supply. For Indian exporters, this presents an opportunity to strengthen relationships with the ministry by demonstrating reliability, quality compliance, and the ability to meet large-scale procurement needs. However, the existing supplier's established presence means that new entrants must offer compelling value propositions to gain a foothold in the market.
Importing Pharmaceuticals into Zambia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Zambia
1Regulatory Authority & Framework
The Zambia Medicines Regulatory Authority (ZAMRA) is the principal body responsible for regulating the pharmaceutical industry in Zambia. Established under the Medicines and Allied Substances Act No. 3 of 2013, ZAMRA oversees the registration, licensing, and monitoring of pharmaceutical products and establishments. The authority ensures that all medicines and medical supplies meet the required standards of quality, safety, and efficacy before they are marketed in Zambia. This regulatory framework is aligned with international standards, including those set by the World Health Organization (WHO), to safeguard public health. (zamra.co.zm)
2Import Licensing & GMP
To import pharmaceutical products into Zambia, companies must obtain a Pharmaceutical License from ZAMRA, which involves demonstrating compliance with Good Manufacturing Practices (GMP). ZAMRA recognizes GMP certifications from reputable international bodies, including the European Union (EU), the World Health Organization (WHO), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers are also required to secure an import permit for each consignment, ensuring that all imported medicines meet ZAMRA's standards. Additionally, wholesale distributors must obtain authorization from ZAMRA to operate legally within Zambia. (zamra.co.zm)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing and stability assessments to confirm their quality and efficacy. ZAMRA mandates that all medicines comply with its quality control standards, which may include laboratory testing of critical commodities such as antiretrovirals, anti-TB drugs, anti-malarials, and injectable antibiotics. Labeling requirements stipulate that information be provided in English, the official language of Zambia, and include details such as the product name, dosage form, strength, batch number, manufacturing date, expiry date, and storage conditions. Serialization mandates may also be enforced to enhance traceability and prevent counterfeit products. (zamra.co.zm)
4Recent Regulatory Changes
Between 2024 and 2026, Zambia has undertaken legislative amendments to streamline drug procurement processes. In June 2023, Health Minister Sylvia Masebo called for urgent amendments to the Zambia Medicines and Medical Supplies Agency (ZAMMSA) Act and the Zambia Public Procurement Act (ZPPA) to address challenges in drug procurement. These proposed changes aim to expedite the acquisition of essential medical resources, including ambulances and health center kits, by removing legislative constraints that have previously delayed procurement efforts.
Ministry OF Health OF THE Republic — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
The Ministry of Health's focus on importing tuberculosis (TB) medications and advanced antibiotics aligns with Zambia's public health priorities. TB remains a significant health concern in the country, necessitating a steady supply of effective treatments. The importation of advanced antibiotics addresses the need for specialized medications to combat resistant infections. The market demand for these products is driven by the prevalence of infectious diseases and the ministry's commitment to providing comprehensive healthcare services to the population.
2Sourcing Profile
The Ministry of Health's sourcing strategy emphasizes the procurement of generic drugs, particularly in the therapeutic areas of tuberculosis and advanced antibiotics. This approach is cost-effective and ensures the availability of essential medicines. India's pharmaceutical industry, known for its large-scale production of generics, plays a crucial role in meeting Zambia's procurement needs. The ministry's focus on finished pharmaceutical formulations, rather than raw active pharmaceutical ingredients (APIs), indicates a preference for ready-to-use products that meet international quality standards.
3Market Positioning
By importing a significant volume of tuberculosis medications and advanced antibiotics, the Ministry of Health positions itself as a central supplier to public health institutions across Zambia. This strategy ensures that government health facilities have access to essential medicines, supporting the ministry's role in delivering healthcare services through government tenders and wholesale distribution channels. The focus on these therapeutic areas underscores the ministry's commitment to addressing critical health challenges in the country.
Seller's Guide — How to Become a Supplier to Ministry OF Health OF THE Republic
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a potential opportunity for new Indian suppliers to enter the Zambian market by offering high-quality, cost-effective generic drugs that meet ZAMRA's regulatory standards. To succeed, suppliers must demonstrate reliability, compliance with international GMP standards, and the capacity to meet large-scale procurement requirements. Additionally, establishing strong relationships with the Ministry of Health and understanding the specific needs of the Zambian healthcare system will be crucial for gaining a competitive edge.
2Requirements & Qualifications
Indian exporters aiming to supply the Ministry of Health must obtain GMP certifications recognized by ZAMRA, such as those from the EU, WHO, or PIC/S. They must also secure a Pharmaceutical License from ZAMRA and comply with all import regulations, including obtaining import permits for each consignment. Products must meet ZAMRA's quality control standards, undergo necessary testing, and adhere to labeling requirements in English. Additionally, exporters should be prepared to participate in Zambia's public procurement processes, which may involve competitive bidding and compliance with the Public Procurement Act.
3How to Approach
To establish a relationship with the Ministry of Health, Indian exporters should first ensure compliance with ZAMRA's regulatory requirements, including obtaining the necessary licenses and certifications. Engaging with the ministry through official channels, such as responding to public tenders and participating in procurement processes, is essential. Building a reputation for reliability, quality, and adherence to delivery schedules will enhance the likelihood of successful partnerships. Understanding the specific needs of the Zambian healthcare system and aligning product offerings accordingly will further strengthen the exporter’s position in the market.
Frequently Asked Questions — Ministry OF Health OF THE Republic
What products does Ministry OF Health OF THE Republic import from India?
Ministry OF Health OF THE Republic imports 4 pharmaceutical products across 2 categories. Top imports: Rifampicin ($1.8M), Isoniazid ($1.4M), Ethambutol ($650.0K), Pyrazinamide ($300.0K).
Who supplies pharmaceuticals to Ministry OF Health OF THE Republic from India?
Ministry OF Health OF THE Republic sources from 5 verified Indian suppliers. The primary supplier is Macleods Pharmaceuticals Limited (75.0% of imports, $1.1M).
What is Ministry OF Health OF THE Republic's total pharmaceutical import value?
Ministry OF Health OF THE Republic's total pharmaceutical import value from India is $4.1M, based on 82 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Ministry OF Health OF THE Republic focus on?
Ministry OF Health OF THE Republic imports across 2 categories. The largest: Tuberculosis Medications (56.1%), Advanced Antibiotics (43.9%).
Get Full Ministry OF Health OF THE Republic Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Ministry OF Health OF THE Republic identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Ministry OF Health OF THE Republic's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 82 individual customs records matching Ministry OF Health OF THE Republic.
- 5.Supplier Verification: Ministry OF Health OF THE Republic sources from 5 verified Indian suppliers across 47 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.