Jamp Pharma Corporation
Pharmaceutical Importer · Canada · Cardiovascular Focus · $13.3M Total Trade · DGFT Verified
Jamp Pharma Corporation is a pharmaceutical importer based in Canada with a total trade value of $13.3M across 14 products in 9 therapeutic categories. Based on 401 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Jamp Pharma Corporation sources from 36 verified Indian suppliers, with Medreich Limited accounting for 26.5% of imports.
Jamp Pharma Corporation — Import Portfolio & Supplier Network
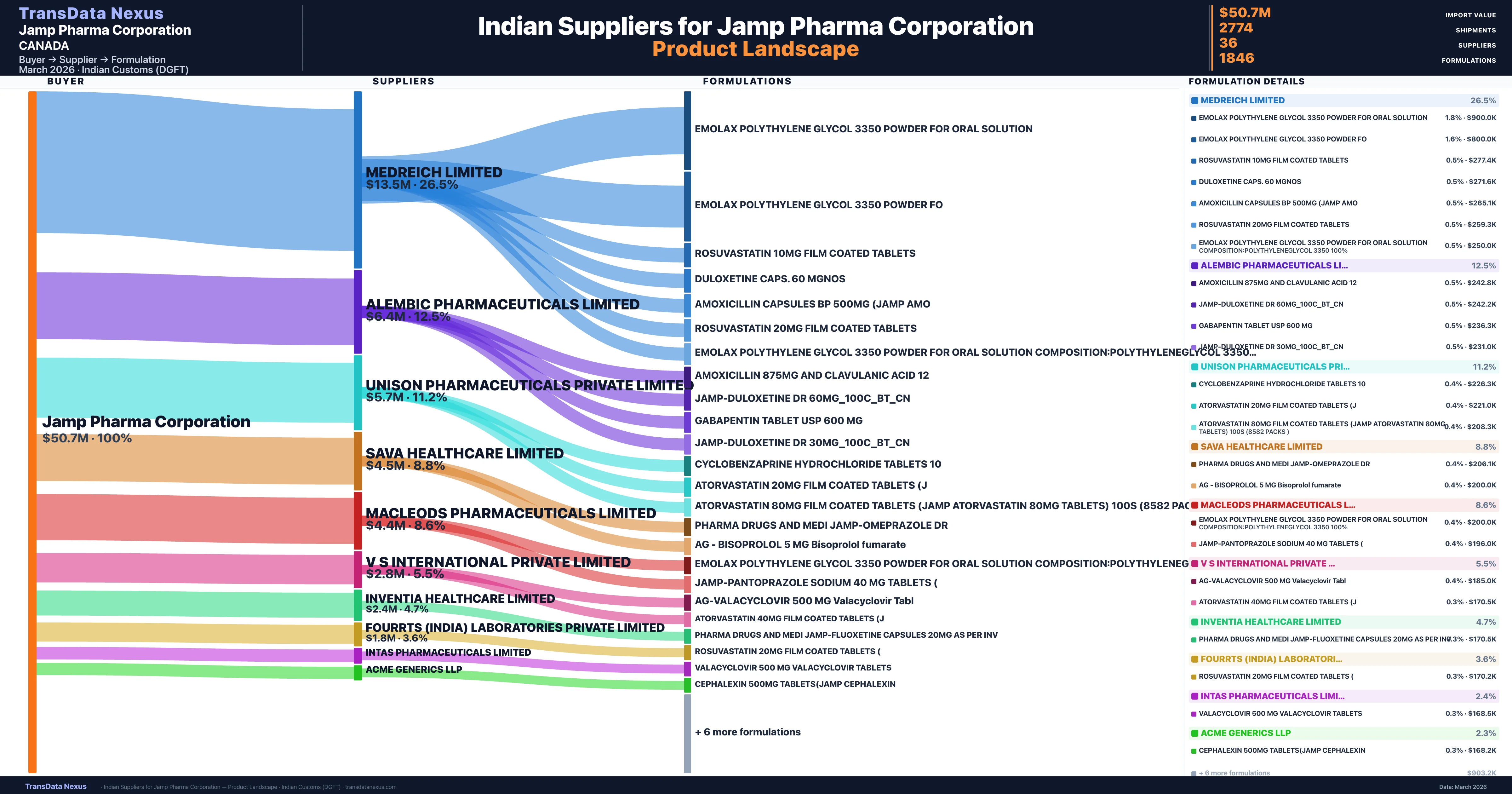
Who Are the Verified Indian Suppliers to Jamp Pharma Corporation?
Customs-verified supplier relationships from Indian DGFT records
Jamp Pharma Corporation sources from 36 verified Indian suppliers across 1,846 distinct formulations. The supply base is diversified across 36 suppliers, reducing single-source dependency risk.
What Formulations Does Jamp Pharma Corporation Import?
| Formulation | Value | Ships |
|---|---|---|
| Emolax polythylene glycol 3350 powder for oral | $900.0K | 18 |
| Emolax polythylene glycol 3350 powder | $800.0K | 16 |
| Rosuvastatin 10MG film coated | $277.4K | 9 |
| Duloxetine CAPS. 60 | $271.6K | 12 |
| Amoxicillin capsules BP 500MG (jamp | $265.1K | 7 |
| Rosuvastatin 20MG film coated | $259.3K | 11 |
| Emolax polythylene glycol 3350 powder for oral solution composition:polythyleneglycol 3350 | $250.0K | 5 |
| Amoxicillin 875MG and clavulanic acid | $242.8K | 8 |
| Jamp-duloxetine dr | $242.2K | 6 |
| Gabapentin tablet USP 600 MG | $236.3K | 9 |
| Jamp-duloxetine dr | $231.0K | 5 |
| Cyclobenzaprine hydrochloride tablets | $226.3K | 6 |
| Atorvastatin 20MG film coated tablets | $221.0K | 11 |
| Atorvastatin 80MG film coated tablets (jamp atorvastatin 80MG tablets) 100s (8582 packs ) | $208.3K | 5 |
| Pharma drugs and medi jamp-omeprazole | $206.1K | 7 |
Jamp Pharma Corporation imports 1,846 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Jamp Pharma Corporation Import?
Jamp Pharma Corporation Therapeutic Categories — 9 Specializations
Jamp Pharma Corporation imports across 9 therapeutic categories, with Cardiovascular (31.1%), Advanced Diabetes Medications (22.9%), Antivirals (19.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 69% of total imports.
Cardiovascular
4 products · 31.1% · $4.1M
Advanced Diabetes Medications
1 products · 22.9% · $3.0M
Antivirals
2 products · 19.8% · $2.6M
Combination Drugs
2 products · 10.4% · $1.4M
Ayurvedic & Herbal Products
1 products · 8.1% · $1.1M
Advanced Oncology
1 products · 2.4% · $321.0K
Lipid & Metabolism
1 products · 2.3% · $300.0K
Advanced Antibiotics
1 products · 1.7% · $220.4K
Antimalarial & Antiparasitic
1 products · 1.4% · $180.5K
Import Portfolio — Top 14 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Dapagliflozin | Advanced Diabetes Medications | $3.0M | 72 | 4.6% | 4 |
| 2 | Apixaban | Cardiovascular | $2.3M | 71 | 1.3% | 12 |
| 3 | Acyclovir | Antivirals | $1.3M | 37 | 1.0% | 13 |
| 4 | Valacyclovir | Antivirals | $1.3M | 37 | 1.4% | 8 |
| 5 | Bisoprolol | Cardiovascular | $1.1M | 28 | 1.3% | 9 |
| 6 | Natural | Ayurvedic & Herbal Products | $1.1M | 25 | 3.1% | 8 |
| 7 | Salicylic | Combination Drugs | $841.7K | 25 | 3.5% | 7 |
| 8 | Simvastatin | Cardiovascular | $569.3K | 51 | 0.5% | 17 |
| 9 | Acetylsalicylic | Combination Drugs | $542.8K | 14 | 8.3% | 3 |
| 10 | Temozolomide | Advanced Oncology | $321.0K | 12 | 1.4% | 14 |
| 11 | Febuxostat | Lipid & Metabolism | $300.0K | 6 | 0.6% | 18 |
| 12 | Nitrofurantoin | Advanced Antibiotics | $220.4K | 9 | 0.6% | 19 |
| 13 | Pyrantel | Antimalarial & Antiparasitic | $180.5K | 6 | 5.5% | 5 |
| 14 | Digoxin | Cardiovascular | $95.5K | 8 | 3.3% | 4 |
Jamp Pharma Corporation imports 14 pharmaceutical products across 9 categories into Canada totaling $13.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Jamp Pharma Corporation.
Request DemoJamp Pharma Corporation — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
JAMP Pharma Corporation, established in 1988, is a privately held Canadian pharmaceutical company headquartered in Boucherville, Quebec. Initially founded as a contract manufacturing organization (CMO), JAMP Pharma has evolved into a comprehensive provider of high-quality medicines and healthcare solutions for patients and consumers across Canada. (jamppharma.ca)
Under the leadership of CEO Louis Pilon, who acquired the company in 2006, JAMP Pharma has experienced significant growth, expanding its portfolio to include over 350 molecules and becoming a leader in the Canadian generic pharmaceutical industry. The company operates through various divisions, such as Orimed Pharma, BioJAMP, Wampole, Laboratoire Suisse, and Cosmetic Import, offering a diverse range of products, including generic medications, biosimilars, over-the-counter products, and natural health products. (jamppharma.ca)
2Distribution Network
JAMP Pharma's distribution network is robust, ensuring nationwide coverage across Canada. The company maintains a fully integrated commercial operation, including warehousing, quality assurance laboratories, and cold chain maintenance capabilities. This infrastructure supports the supply of over 11,500 pharmacies from coast to coast, demonstrating the company's commitment to providing reliable access to its products. (jamppharma.ca)
3Industry Role
In Canada's pharmaceutical supply chain, JAMP Pharma Corporation functions primarily as a wholesaler and distributor. The company imports a diverse range of pharmaceutical formulations, including tablets, capsules, syrups, and injections, from various international suppliers, notably from India. JAMP Pharma's extensive portfolio and distribution capabilities position it as a significant player in the Canadian pharmaceutical market.
Supplier Relationship Intelligence — Jamp Pharma Corporation
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
JAMP Pharma's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 68.8% of its total import value from India. This concentration indicates a strategic focus on specific high-demand therapeutic areas, such as cardiovascular, advanced diabetes medications, and antivirals. The company's reliance on a select group of suppliers, including MEDREICH LIMITED, ALEMBIC PHARMACEUTICALS LIMITED, and UNISON PHARMACEUTICALS PRIVATE LIMITED, suggests a stable and established relationship with these partners. However, this dependency also presents potential risks, such as supply chain disruptions or price fluctuations.
2Supply Chain Resilience
JAMP Pharma's supply chain resilience is supported by its diversified portfolio of 1,846 unique formulations and a network of 36 verified Indian suppliers. This diversity mitigates risks associated with over-reliance on a single supplier or product. The company's established relationships with key suppliers, such as MEDREICH LIMITED and ALEMBIC PHARMACEUTICALS LIMITED, enhance supply chain stability. Additionally, JAMP Pharma's integrated operations, including warehousing and quality assurance laboratories, contribute to its ability to maintain a reliable supply of pharmaceutical products.
3Strategic Implications
JAMP Pharma's sourcing pattern, characterized by a concentrated focus on specific therapeutic areas and key suppliers, reinforces its competitive position in the Canadian pharmaceutical market. This strategy enables the company to leverage economies of scale and establish strong partnerships with suppliers. For Indian exporters, understanding JAMP Pharma's product focus and supplier relationships is crucial for identifying opportunities to become alternative suppliers, particularly in the cardiovascular, diabetes, and antiviral segments.
Importing Pharmaceuticals into Canada — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Canada
1Regulatory Authority & Framework
In Canada, the importation and sale of pharmaceutical products are regulated by Health Canada under the Food and Drugs Act and its associated Regulations. This framework ensures that all health products, including human drugs, medical devices, and natural health products, meet rigorous standards for safety, effectiveness, and quality. (canada.ca)
2Import Licensing & GMP
Importers of pharmaceutical products in Canada must obtain an Establishment Licence (EL) for the activity of importation. Additionally, each drug product must have a Drug Identification Number (DIN) assigned by Health Canada prior to being marketed. Good Manufacturing Practice (GMP) compliance is mandatory, and foreign manufacturing sites must be listed on the importer's EL. Recognized GMP certifications include EU GMP, WHO GMP, and PIC/S. (canada.ca)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet Health Canada's quality standards. Stability requirements are specified to ensure product efficacy and safety throughout its shelf life. Labeling must be in both English and French, as per Canadian regulations, and must include all necessary information, such as dosage instructions, ingredients, and expiry dates. Serialization mandates may apply to facilitate traceability and prevent counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, Health Canada has implemented several policy changes affecting pharmaceutical imports. These include updates to the Access to Medicines Regime, which facilitates the importation of generic drugs to address public health needs, and revisions to GMP compliance requirements for foreign manufacturers. Importers must stay informed about these changes to ensure continued compliance. (canada.ca)
Jamp Pharma Corporation — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
JAMP Pharma's product strategy focuses on therapeutic areas with high market demand, including cardiovascular, advanced diabetes medications, and antivirals. The company's importation of specific products like Dapagliflozin, Apixaban, Acyclovir, Valacyclovir, and Bisoprolol aligns with this focus, addressing prevalent health conditions in Canada. This strategic approach ensures that JAMP Pharma meets the evolving needs of the Canadian healthcare system.
2Sourcing Profile
JAMP Pharma's sourcing strategy emphasizes the importation of generic drug formulations from India, leveraging the country's established pharmaceutical manufacturing capabilities. The company's preference for finished pharmaceutical formulations over raw active pharmaceutical ingredients (APIs) allows for a more streamlined supply chain and ensures product quality. India's compliance with recognized GMP standards, such as WHO GMP and PIC/S, aligns with JAMP Pharma's quality requirements.
3Market Positioning
JAMP Pharma Corporation serves multiple segments of the Canadian pharmaceutical market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. The company's extensive product portfolio and reliable supply chain enable it to meet the diverse needs of these segments, positioning JAMP Pharma as a versatile and dependable partner in the Canadian healthcare landscape.
Seller's Guide — How to Become a Supplier to Jamp Pharma Corporation
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to enter JAMP Pharma's supply chain, particularly in the cardiovascular, diabetes, and antiviral therapeutic areas. To capitalize on this opportunity, Indian exporters should focus on meeting Health Canada's regulatory requirements, including obtaining the necessary GMP certifications and ensuring compliance with labeling and quality standards. Establishing strong relationships with JAMP Pharma and understanding its specific product needs will be crucial for successful market entry.
2Requirements & Qualifications
Indian exporters aiming to supply JAMP Pharma Corporation must obtain GMP certifications recognized by Health Canada, such as EU GMP, WHO GMP, or PIC/S. They must also ensure that their products meet Health Canada's quality standards, including batch testing and stability requirements. Labeling must be in both English and French, as per Canadian regulations. Additionally, exporters must be prepared to obtain a Drug Identification Number (DIN) for each product prior to marketing in Canada. (canada.ca)
3How to Approach
Indian exporters should initiate contact with JAMP Pharma Corporation by presenting their product portfolio and demonstrating compliance with Health Canada's regulatory requirements. Participating in industry trade shows and conferences can facilitate networking opportunities. Understanding JAMP Pharma's specific product needs and therapeutic focus areas will enable exporters to tailor their offerings effectively. Establishing a local presence or partnering with a Canadian distributor can enhance credibility and facilitate market entry. It is essential to stay informed about Health Canada's regulatory updates and ensure ongoing compliance to maintain a successful partnership.
Frequently Asked Questions — Jamp Pharma Corporation
What products does Jamp Pharma Corporation import from India?
Jamp Pharma Corporation imports 14 pharmaceutical products across 9 categories. Top imports: Dapagliflozin ($3.0M), Apixaban ($2.3M), Acyclovir ($1.3M), Valacyclovir ($1.3M), Bisoprolol ($1.1M).
Who supplies pharmaceuticals to Jamp Pharma Corporation from India?
Jamp Pharma Corporation sources from 36 verified Indian suppliers. The primary supplier is Medreich Limited (26.5% of imports, $13.5M).
What is Jamp Pharma Corporation's total pharmaceutical import value?
Jamp Pharma Corporation's total pharmaceutical import value from India is $13.3M, based on 401 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Jamp Pharma Corporation focus on?
Jamp Pharma Corporation imports across 9 categories. The largest: Cardiovascular (31.1%), Advanced Diabetes Medications (22.9%), Antivirals (19.8%).
Get Full Jamp Pharma Corporation Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Jamp Pharma Corporation identified across shipments using consignee name normalization, aggregating 4 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Jamp Pharma Corporation's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 401 individual customs records matching Jamp Pharma Corporation.
- 5.Supplier Verification: Jamp Pharma Corporation sources from 36 verified Indian suppliers across 1,846 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
14 Products Tracked
9 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 4 company name variants from customs records. For current shipment-level data, contact TransData Nexus.