CSP Viatris Sante AT Csp- Lab,
Pharmaceutical Importer · France · Cardiovascular Focus · $10.6M Total Trade · DGFT Verified
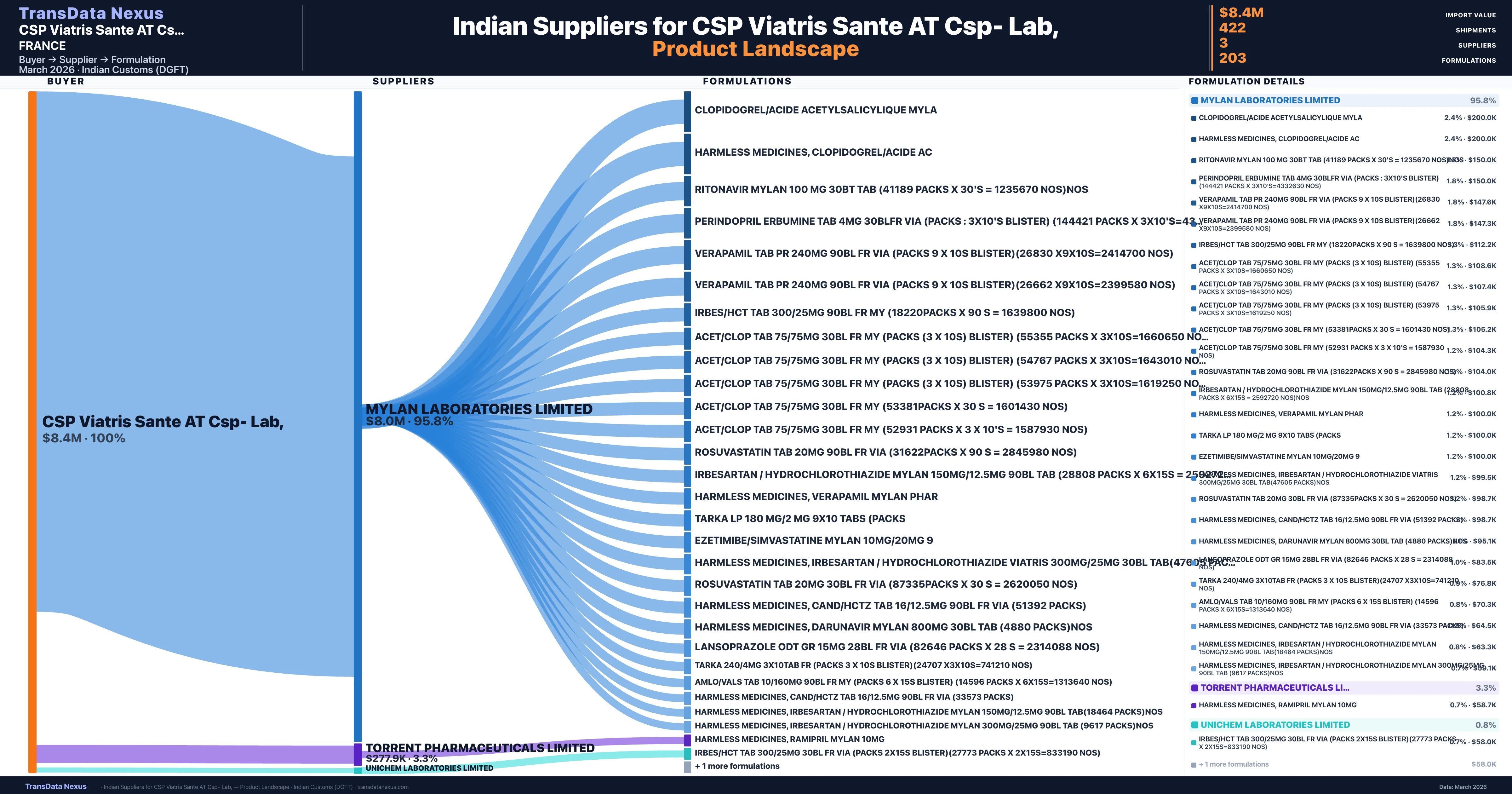
CSP Viatris Sante AT Csp- Lab, is a pharmaceutical importer based in France with a total trade value of $10.6M across 7 products in 5 therapeutic categories. Based on 212 verified import shipments from Indian Customs (DGFT) records, CSP Viatris Sante AT Csp- Lab, is the #1 buyer in 1 product including Ramipril. CSP Viatris Sante AT Csp- Lab, sources from 3 verified Indian suppliers, with Mylan Laboratories Limited accounting for 95.8% of imports.
CSP Viatris Sante AT Csp- Lab, — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to CSP Viatris Sante AT Csp- Lab,?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Mylan Laboratories Limited | $8.0M | 386 | 95.8% |
| Torrent Pharmaceuticals Limited | $277.9K | 34 | 3.3% |
| Unichem Laboratories Limited | $70.4K | 2 | 0.8% |
CSP Viatris Sante AT Csp- Lab, sources from 3 verified Indian suppliers across 203 distinct formulations. The sourcing is highly concentrated — Mylan Laboratories Limited accounts for 95.8% of total imports, indicating a strategic single-source relationship.
What Formulations Does CSP Viatris Sante AT Csp- Lab, Import?
| Formulation | Value | Ships |
|---|---|---|
| Clopidogrel/acide acetylsalicylique | $200.0K | 4 |
| Harmless medicines, clopidogrel/acide | $200.0K | 4 |
| Ritonavir mylan 100 MG 30bt TAB (41189 packs x 30's = 1235670 nos | $150.0K | 3 |
| Perindopril erbumine TAB 4MG 30blfr via (packs : 3x10's blister) (144421 packs x 3x10's=4332630 nos) | $150.0K | 3 |
| Verapamil TAB pr 240MG 90bl fr via (packs 9 x 10s blister)(26830 x9x10s=2414700 nos) | $147.6K | 3 |
| Verapamil TAB pr 240MG 90bl fr via (packs 9 x 10s blister)(26662 x9x10s=2399580 nos) | $147.3K | 3 |
| Irbes/hct TAB 300/25MG 90bl fr my (18220packs x 90 s = 1639800 nos) | $112.2K | 3 |
| Acet/clop TAB 75/75MG 30bl fr my (packs (3 x 10s) blister) (55355 packs x 3x10s=1660650 nos) | $108.6K | 3 |
| Acet/clop TAB 75/75MG 30bl fr my (packs (3 x 10s) blister) (54767 packs x 3x10s=1643010 nos) | $107.4K | 3 |
| Acet/clop TAB 75/75MG 30bl fr my (packs (3 x 10s) blister) (53975 packs x 3x10s=1619250 nos) | $105.9K | 3 |
| Acet/clop TAB 75/75MG 30bl fr my (53381packs x 30 s = 1601430 nos) | $105.2K | 3 |
| Acet/clop TAB 75/75MG 30bl fr my (52931 packs x 3 x 10's = 1587930 nos) | $104.3K | 3 |
| Rosuvastatin TAB 20MG 90bl fr via (31622packs x 90 s = 2845980 nos) | $104.0K | 3 |
| Irbesartan / hydrochlorothiazide mylan 150MG/12.5MG 90bl TAB (28808 packs x 6x15s = 2592720 nos | $100.8K | 3 |
| Harmless medicines, verapamil mylan | $100.0K | 2 |
CSP Viatris Sante AT Csp- Lab, imports 203 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does CSP Viatris Sante AT Csp- Lab, Import?
CSP Viatris Sante AT Csp- Lab, Therapeutic Categories — 5 Specializations
CSP Viatris Sante AT Csp- Lab, imports across 5 therapeutic categories, with Cardiovascular (58.5%), Diuretics (17.5%), Gastrointestinal (13.2%) representing the largest segments. The portfolio is concentrated — top 5 products = 89% of total imports.
Cardiovascular
3 products · 58.5% · $6.2M
Diuretics
1 products · 17.5% · $1.9M
Gastrointestinal
1 products · 13.2% · $1.4M
Antiviral & HIV Medications
1 products · 5.7% · $600.0K
Immunosuppressants
1 products · 5.2% · $550.0K
Import Portfolio — Top 7 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Irbesartan | Cardiovascular | $2.8M | 56 | 1.7% | 4 |
| 2 | Ramipril | Cardiovascular | $2.2M | 44 | 2.5% | 1 |
| 3 | Hydrochlorothiazide | Diuretics | $1.9M | 37 | 0.5% | 7 |
| 4 | Lansoprazole | Gastrointestinal | $1.4M | 28 | 0.5% | 8 |
| 5 | Verapamil | Cardiovascular | $1.2M | 24 | 0.2% | 3 |
| 6 | Darunavir | Antiviral & HIV Medications | $600.0K | 12 | 1.0% | 9 |
| 7 | Azathioprine | Immunosuppressants | $550.0K | 11 | 1.0% | 13 |
CSP Viatris Sante AT Csp- Lab, imports 7 pharmaceutical products across 5 categories into France totaling $10.6M. The company is the #1 buyer for 1 product: Ramipril.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for CSP Viatris Sante AT Csp- Lab,.
Request DemoCSP Viatris Sante AT Csp- Lab, — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
CSP Viatris Sante AT CSP-Lab is a pharmaceutical importer based in France, specializing in the acquisition of finished pharmaceutical formulations from international suppliers. The company is headquartered at 76-78 Avenue du Midi, 63800 Cournon d'Auvergne, France. While specific details about its parent company are not publicly available, CSP Viatris Sante AT CSP-Lab operates as an independent entity within the French pharmaceutical distribution network. Its primary role involves sourcing and importing a diverse range of pharmaceutical products to meet the needs of the French healthcare market.
2Distribution Network
CSP Viatris Sante AT CSP-Lab's distribution network encompasses strategic warehouse locations and logistics capabilities designed to ensure efficient delivery of pharmaceutical products across France. The company's operations are centered in Cournon d'Auvergne, providing a logistical advantage for nationwide distribution. While specific details about additional warehouse locations and international logistics capabilities are not publicly disclosed, CSP Viatris Sante AT CSP-Lab's established infrastructure supports its role as a key importer in the French pharmaceutical sector.
3Industry Role
In France's pharmaceutical supply chain, CSP Viatris Sante AT CSP-Lab functions primarily as a pharmaceutical importer, sourcing finished pharmaceutical formulations from international suppliers, particularly from India. The company's import activities are characterized by a focus on specific therapeutic categories, including cardiovascular, diuretics, and gastrointestinal medications. By importing these products, CSP Viatris Sante AT CSP-Lab plays a crucial role in ensuring the availability of essential medications within the French market.
Supplier Relationship Intelligence — CSP Viatris Sante AT Csp- Lab,
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
CSP Viatris Sante AT CSP-Lab demonstrates a high degree of sourcing concentration, with a significant portion of its imports originating from a limited number of suppliers. The company's total import value from India amounts to $10.6 million USD, with 95.8% of shipments sourced from Mylan Laboratories Limited, totaling $8.0 million USD. This concentration indicates a strategic choice to maintain a stable and reliable supply chain with established partners. However, such dependency may pose risks related to supply chain disruptions or changes in supplier dynamics. The stability of this relationship is crucial for CSP Viatris Sante AT CSP-Lab's operations, and any significant changes could impact the company's ability to meet market demands.
2Supply Chain Resilience
CSP Viatris Sante AT CSP-Lab's supply chain resilience is closely tied to its sourcing strategy, which heavily relies on Mylan Laboratories Limited for the majority of its imports. The company's import data indicates a focus on specific formulations, such as indapamide sustained release tablets, sourced from suppliers like Torrent Pharmaceuticals Limited. While this targeted sourcing approach allows for specialization and potential cost efficiencies, it also exposes the company to risks associated with supplier-specific disruptions. The lack of a diversified supplier base may limit CSP Viatris Sante AT CSP-Lab's ability to adapt to changes in the global pharmaceutical supply chain. Ensuring compliance with international quality standards and maintaining strong relationships with key suppliers are essential for mitigating these risks.
3Strategic Implications
CSP Viatris Sante AT CSP-Lab's sourcing pattern, characterized by a high concentration of imports from a single supplier, positions the company to benefit from established relationships and potential cost efficiencies. However, this strategy also exposes the company to risks related to supply chain disruptions or changes in supplier dynamics. For Indian exporters, understanding CSP Viatris Sante AT CSP-Lab's sourcing preferences presents an opportunity to establish partnerships, particularly in the therapeutic categories where the company imports products. Diversifying the supplier base could enhance CSP Viatris Sante AT CSP-Lab's supply chain resilience and provide Indian exporters with a stable market for their products.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the primary regulatory authority overseeing pharmaceutical imports is the French National Agency for Medicines and Health Products Safety (ANSM). The ANSM is responsible for ensuring the safety, efficacy, and quality of health products, including pharmaceuticals, medical devices, and cosmetics. Key legislation governing pharmaceutical imports includes the Public Health Code, which outlines the requirements for marketing authorization, importation, and distribution of pharmaceutical products. The marketing authorization pathway for Indian generics involves obtaining approval from the ANSM, which assesses the product's quality, safety, and efficacy before granting authorization for sale in the French market.
2Import Licensing & GMP
Import licensing requirements in France stipulate that pharmaceutical importers must hold a valid wholesale distribution authorization issued by the ANSM. This authorization ensures that importers comply with Good Distribution Practice (GDP) standards, which are essential for maintaining the quality and integrity of pharmaceutical products throughout the supply chain. Regarding Good Manufacturing Practice (GMP) certification, the ANSM recognizes certifications from the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian exporters seeking to supply CSP Viatris Sante AT CSP-Lab must ensure that their manufacturing facilities hold valid GMP certifications recognized by the ANSM to meet import requirements.
3Quality & Labeling
Pharmaceutical products imported into France must undergo batch testing to verify their quality, safety, and efficacy. Stability requirements are also enforced to ensure that products maintain their intended quality throughout their shelf life. Labeling language requirements mandate that product labels be in French, providing essential information such as dosage instructions, side effects, and storage conditions. Serialization mandates are in place to track and trace pharmaceutical products, enhancing supply chain security and preventing counterfeit products from entering the market. Indian exporters must adhere to these quality and labeling standards to ensure compliance with French regulations and facilitate the importation process.
4Recent Regulatory Changes
Between 2024 and 2026, France implemented several policy changes affecting pharmaceutical imports, including those from India. These changes involved stricter enforcement of GMP certification requirements, with an emphasis on recognizing certifications from EU GMP, WHO GMP, and PIC/S. Additionally, the ANSM introduced more rigorous batch testing protocols and stability requirements to enhance product safety and efficacy. Labeling language requirements were also updated to ensure clearer communication with consumers, and serialization mandates were expanded to improve traceability and combat counterfeit products. Indian exporters aiming to supply CSP Viatris Sante AT CSP-Lab must stay informed about these regulatory changes to ensure compliance and maintain a competitive edge in the French market.
CSP Viatris Sante AT Csp- Lab, — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
CSP Viatris Sante AT CSP-Lab's product category focus on cardiovascular, diuretics, and gastrointestinal medications aligns with prevalent health concerns in France, such as hypertension, heart disease, and digestive disorders. The company's import strategy is driven by market demand for these therapeutic areas, necessitating a consistent supply of effective and affordable treatments. By concentrating on these categories, CSP Viatris Sante AT CSP-Lab aims to address critical healthcare needs and maintain a competitive position in the French pharmaceutical market.
2Sourcing Profile
CSP Viatris Sante AT CSP-Lab's sourcing strategy emphasizes the importation of generic drugs, particularly those manufactured in India. The company's focus on specific formulations, such as indapamide sustained release tablets, indicates a preference for specialized products that meet the therapeutic needs of the French market. India's established pharmaceutical manufacturing capabilities and recognized GMP certifications make it a key supplier for CSP Viatris Sante AT CSP-Lab. This sourcing approach allows the company to offer cost-effective and high-quality generic medications to the French healthcare system.
3Market Positioning
Based on its product mix, CSP Viatris Sante AT CSP-Lab serves various segments of the French pharmaceutical market, including retail pharmacies, hospitals, and government tenders. The company's focus on essential therapeutic categories positions it to meet the needs of both outpatient and inpatient care settings. By ensuring a steady supply of critical medications, CSP Viatris Sante AT CSP-Lab plays a vital role in maintaining the overall health infrastructure in France.
Seller's Guide — How to Become a Supplier to CSP Viatris Sante AT Csp- Lab,
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with CSP Viatris Sante AT CSP-Lab, particularly in the therapeutic categories where the company imports products. The company's existing supplier base is concentrated, indicating potential for diversification and risk mitigation. Indian exporters can explore opportunities by offering specialized formulations that align with CSP Viatris Sante AT CSP-Lab's product strategy and by ensuring compliance with French regulatory standards. Establishing partnerships with CSP Viatris Sante AT CSP-Lab could provide Indian exporters with access to the French market and contribute to the company's supply chain resilience.
2Requirements & Qualifications
Indian exporters seeking to supply CSP Viatris Sante AT CSP-Lab and the French market must ensure that their manufacturing facilities hold valid GMP certifications recognized by the ANSM, such as EU GMP, WHO GMP, or PIC/S. Products must undergo batch testing to verify quality, safety, and efficacy, and comply with stability requirements. Labeling must be in French, providing essential product information. Serialization mandates must be met to enhance traceability and prevent counterfeit products. Adhering to these standards is crucial for gaining approval from French regulatory authorities and establishing a successful partnership with CSP Viatris Sante AT CSP-Lab.
Frequently Asked Questions — CSP Viatris Sante AT Csp- Lab,
What products does CSP Viatris Sante AT Csp- Lab, import from India?
CSP Viatris Sante AT Csp- Lab, imports 7 pharmaceutical products across 5 categories. Top imports: Irbesartan ($2.8M), Ramipril ($2.2M), Hydrochlorothiazide ($1.9M), Lansoprazole ($1.4M), Verapamil ($1.2M).
Who supplies pharmaceuticals to CSP Viatris Sante AT Csp- Lab, from India?
CSP Viatris Sante AT Csp- Lab, sources from 3 verified Indian suppliers. The primary supplier is Mylan Laboratories Limited (95.8% of imports, $8.0M).
What is CSP Viatris Sante AT Csp- Lab,'s total pharmaceutical import value?
CSP Viatris Sante AT Csp- Lab,'s total pharmaceutical import value from India is $10.6M, based on 212 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does CSP Viatris Sante AT Csp- Lab, focus on?
CSP Viatris Sante AT Csp- Lab, imports across 5 categories. The largest: Cardiovascular (58.5%), Diuretics (17.5%), Gastrointestinal (13.2%).
Get Full CSP Viatris Sante AT Csp- Lab, Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: CSP Viatris Sante AT Csp- Lab, identified across shipments using consignee name normalization, aggregating 5 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as CSP Viatris Sante AT Csp- Lab,'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 212 individual customs records matching CSP Viatris Sante AT Csp- Lab,.
- 5.Supplier Verification: CSP Viatris Sante AT Csp- Lab, sources from 3 verified Indian suppliers across 203 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
7 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 5 company name variants from customs records. For current shipment-level data, contact TransData Nexus.