In April 2025, the WHO unveiled a global repository for National Essential Medicines Lists (nEMLs), aiming to harmonize and improve access to essential medicines worldwide. This initiative is expected to streamline procurement processes and enhance the availability of medications like Darunavir in low- and middle-income countries.
In February 2025, the WHO published the 24th edition of the Model List of Essential Medicines, reaffirming the inclusion of Darunavir. This underscores the ongoing global recognition of Darunavir's importance in HIV treatment protocols.
In May 2025, the 25th WHO Expert Committee on Selection and Use of Essential Medicines convened to review and update the Model List of Essential Medicines. The committee's recommendations continue to influence national policies and procurement strategies, impacting the global supply chain for medications like Darunavir.
In March 2025, the FDA approved a new generic version of Darunavir tablets, further increasing competition in the U.S. market and potentially affecting pricing dynamics for Indian exporters.
In January 2026, the WHO released a fact sheet on essential medicines, highlighting the ongoing challenges in ensuring universal access to essential medications, including antiretrovirals like Darunavir. The report emphasizes the need for sustained efforts to improve availability and affordability globally.
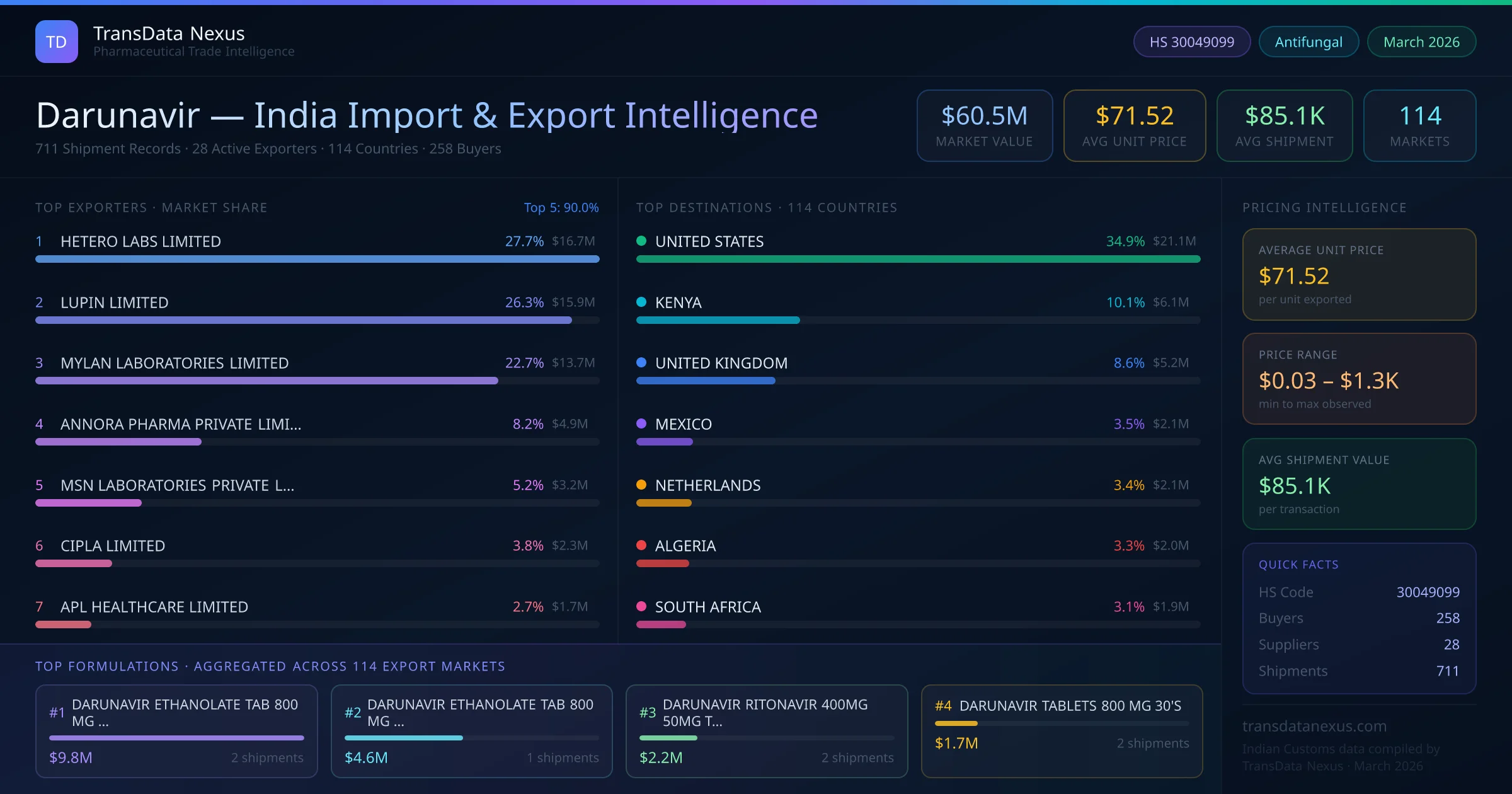
These developments reflect a dynamic regulatory and market environment for Darunavir, with implications for Indian exporters in terms of compliance, competition, and market access.