Cameskin/cordaid
Pharmaceutical Importer · Congo Dr · Tuberculosis Medications Focus · $1.3M Total Trade · DGFT Verified
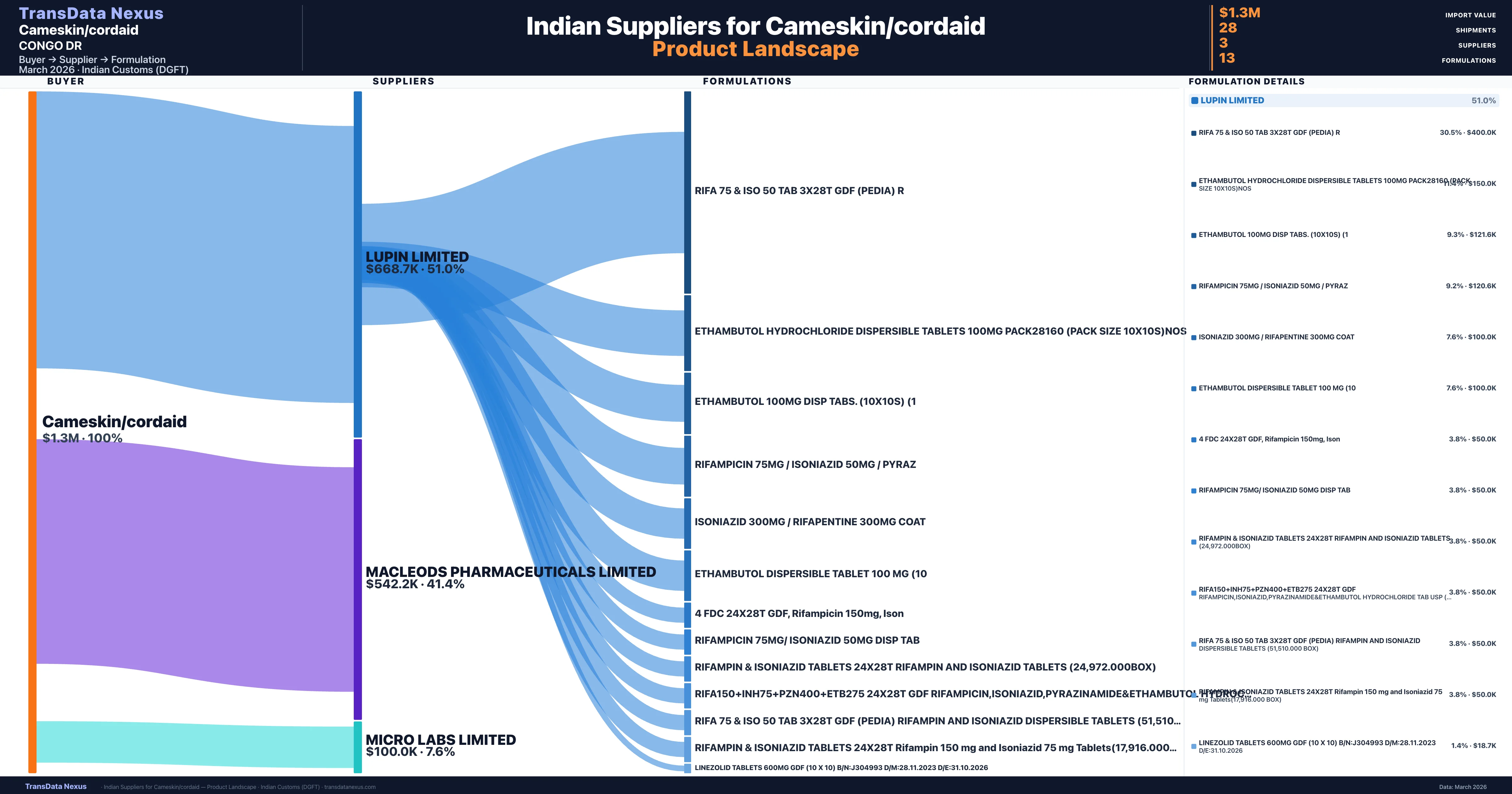
Cameskin/cordaid is a pharmaceutical importer based in Congo Dr with a total trade value of $1.3M across 3 products in 2 therapeutic categories. Based on 25 verified import shipments from Indian Customs (DGFT) records, Cameskin/cordaid is the #1 buyer in 3 products including Isoniazid, Ethambutol, Rifampicin. Cameskin/cordaid sources from 3 verified Indian suppliers, with Lupin Limited accounting for 51.0% of imports.
Cameskin/cordaid — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Cameskin/cordaid?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Lupin Limited | $668.7K | 14 | 51.0% |
| Macleods Pharmaceuticals Limited | $542.2K | 12 | 41.4% |
| Micro Labs Limited | $100.0K | 2 | 7.6% |
Cameskin/cordaid sources from 3 verified Indian suppliers across 13 distinct formulations. Total import value: $1.3M across 28 shipments.
What Formulations Does Cameskin/cordaid Import?
| Formulation | Value | Ships |
|---|---|---|
| Rifa 75 & iso 50 TAB 3x28t gdf (pedia) | $400.0K | 8 |
| Ethambutol hydrochloride dispersible tablets 100MG pack28160 (Pack size 10x10s | $150.0K | 3 |
| Ethambutol 100MG disp tabs. (10x10s) | $121.6K | 3 |
| Rifampicin 75MG / isoniazid 50MG / | $120.6K | 3 |
| Isoniazid 300MG / rifapentine 300MG | $100.0K | 2 |
| Ethambutol dispersible tablet 100 MG | $100.0K | 2 |
| 4 fdc 24x28t gdf, rifampicin 150MG | $50.0K | 1 |
| Rifampicin 75MG/ isoniazid 50MG disp TAB | $50.0K | 1 |
| Rifampin & isoniazid tablets 24x28t rifampin and isoniazid tablets (24,972.000box) | $50.0K | 1 |
| Rifa150+inh75+pzn400+etb275 24x28t gdf rifampicin,isoniazid,pyrazinamideðambutol hydrochloride TAB USP (12263.000box) | $50.0K | 1 |
| Rifa 75 & iso 50 TAB 3x28t gdf (pedia) rifampin and isoniazid dispersible tablets (51,510.000 Box) | $50.0K | 1 |
| Rifampin & isoniazid tablets 24x28t rifampin 150 MG and isoniazid 75 MG tablets(17,916.000 Box) | $50.0K | 1 |
| Linezolid tablets 600MG gdf (10 x 10) b/n:j304993 d/m:28.11.2023 | $18.7K | 1 |
Cameskin/cordaid imports 13 distinct pharmaceutical formulations. Showing top 13 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Cameskin/cordaid Import?
Top Products by Import Value
Cameskin/cordaid Therapeutic Categories — 2 Specializations
Cameskin/cordaid imports across 2 therapeutic categories, with Tuberculosis Medications (76.0%), Advanced Antibiotics (24.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Tuberculosis Medications
2 products · 76.0% · $950.0K
Advanced Antibiotics
1 products · 24.0% · $300.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Isoniazid | Tuberculosis Medications | $500.0K | 10 | 0.0% | 1 |
| 2 | Ethambutol | Tuberculosis Medications | $450.0K | 9 | 0.0% | 1 |
| 3 | Rifampicin | Advanced Antibiotics | $300.0K | 6 | 0.0% | 1 |
Cameskin/cordaid imports 3 pharmaceutical products across 2 categories into Congo Dr totaling $1.3M. The company is the #1 buyer for 3 products: Isoniazid, Ethambutol, Rifampicin.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Cameskin/cordaid.
Request DemoCameskin/cordaid — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Cameskin/Cordaid is a pharmaceutical importer and buyer based in the Democratic Republic of Congo (DRC). The company operates as a wholesaler, sourcing finished pharmaceutical formulations from international suppliers to meet the domestic healthcare needs of the DRC. While specific details about its parent company are not publicly available, Cordaid is known for its humanitarian work in the DRC, focusing on health, education, and emergency response. Cameskin/Cordaid plays a crucial role in the DRC's pharmaceutical distribution network by ensuring the availability of essential medications, particularly for the treatment of tuberculosis, a significant health concern in the region.
2Distribution Network
Cameskin/Cordaid's distribution network within the DRC is not extensively documented in public sources. However, given the company's focus on importing and wholesaling pharmaceutical products, it is likely that they collaborate with local distributors, pharmacies, and healthcare facilities across the country to ensure the widespread availability of their products. The DRC's vast geography and infrastructure challenges necessitate a robust logistics system to effectively distribute medical supplies. Cameskin/Cordaid's partnerships with international suppliers, such as Lupin Limited and Macleods Pharmaceuticals Limited, suggest a well-established supply chain capable of reaching various regions within the DRC.
3Industry Role
Cameskin/Cordaid serves as a primary wholesaler in the DRC's pharmaceutical supply chain. By importing finished pharmaceutical formulations, they bridge the gap between international manufacturers and local healthcare providers. Their focus on essential medications, particularly those used in the treatment of tuberculosis, underscores their commitment to addressing critical health issues in the DRC. Through their import activities, Cameskin/Cordaid contributes to the availability and accessibility of vital medicines, playing a pivotal role in the country's healthcare system.
Supplier Relationship Intelligence — Cameskin/cordaid
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Cameskin/Cordaid's sourcing strategy exhibits a high degree of concentration, with 100% of their pharmaceutical imports originating from three Indian suppliers: Lupin Limited, Macleods Pharmaceuticals Limited, and Micro Labs Limited. This concentrated sourcing approach indicates a strategic choice to maintain strong, reliable relationships with these suppliers, ensuring consistent product quality and supply. However, such dependency also poses risks, including potential supply chain disruptions due to geopolitical issues, regulatory changes, or operational challenges within the supplier companies. The shipment data reveals a stable relationship, with multiple shipments over several years, suggesting effective collaboration and mutual trust between Cameskin/Cordaid and their Indian suppliers.
2Supply Chain Resilience
Cameskin/Cordaid's supply chain resilience appears robust, given their partnerships with reputable Indian pharmaceutical manufacturers. The diversity of formulations imported—13 unique formulations—indicates a strategic approach to sourcing a comprehensive range of tuberculosis medications. While the concentration on three suppliers may limit exposure to alternative sources, the established relationships and consistent shipment patterns suggest a stable supply chain. Nonetheless, the company should remain vigilant to potential risks, such as regulatory changes in the DRC or India, and consider developing contingency plans to mitigate any disruptions.
3Strategic Implications
Cameskin/Cordaid's concentrated sourcing strategy positions them to leverage strong supplier relationships, ensuring consistent product quality and supply. This approach can lead to favorable pricing and priority access to products. For Indian exporters, the company's focus on tuberculosis medications presents an opportunity to strengthen partnerships and expand their market presence in the DRC. However, potential risks associated with supply chain disruptions due to geopolitical or regulatory changes should be considered. Diversifying the supplier base could enhance resilience and mitigate risks, but it may require additional resources and due diligence.
Importing Pharmaceuticals into Congo Dr — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Congo Dr
1Regulatory Authority & Framework
The Congolese Pharmaceutical Regulatory Authority (ACOREP) is responsible for regulating pharmaceutical imports and exports in the DRC. ACOREP oversees the authorization and control of drug imports, ensuring compliance with national health standards. The regulatory framework governing pharmaceutical imports includes obtaining an import license from an authorized commercial bank and undergoing a pre-shipment inspection by Bureau Veritas for shipments valued over $2,500. Additionally, a Certificate of Pharmaceutical Product (CPP) is required for quality control, with initial screenings conducted locally and more comprehensive analyses performed at WHO-prequalified laboratories abroad.
2Import Licensing & GMP
Importers in the DRC must secure an import license from an authorized commercial bank before the physical shipment of goods. A pre-shipment inspection by Bureau Veritas is mandatory for all imports valued at $2,500 FOB or greater. While Good Manufacturing Practice (GMP) certification is officially required, enforcement is often inconsistent; a Certificate of Pharmaceutical Product (CPP) is a critical document. The entire process, from clinical trial application to market entry, is managed by ACOREP, which aims to provide a decision on clinical trial applications within 30 days of submission, following ethics committee approval.
3Quality & Labeling
Imported pharmaceutical products must undergo quality control measures, including pre-shipment inspections and screenings. Batch testing and stability requirements are enforced to ensure product efficacy and safety. Labeling must comply with local regulations, including the use of French on all documentation and labeling. Serialization mandates may be in place to enhance traceability and prevent counterfeit products. Non-compliance with these requirements can lead to significant sanctions, including product recalls and fines.
4Recent Regulatory Changes
In June 2024, the DRC's Ministry of Health updated its list of banned pharmaceutical imports through Ministerial Decree N°1250/cab/min/SPHP/010/CJ/OBK/2024. This decree prohibits the importation of certain pharmaceutical products without prior authorization, with non-compliant goods subject to destruction or re-exportation. Importers must ensure that their products are not on the banned list and comply with all regulatory requirements to avoid penalties.
Cameskin/cordaid — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Cameskin/Cordaid's focus on importing tuberculosis medications, including Isoniazid, Ethambutol, and Rifampicin, aligns with the DRC's significant health needs. The high import values and volume of these products indicate a substantial market demand for effective tuberculosis treatments. This strategic focus allows Cameskin/Cordaid to address a critical health issue while establishing a strong position in the pharmaceutical market.
2Sourcing Profile
Cameskin/Cordaid's sourcing strategy emphasizes the procurement of generic drug formulations, particularly those used in the treatment of tuberculosis. The company's preference for finished pharmaceutical formulations over raw active pharmaceutical ingredients (APIs) suggests a focus on ready-to-use products that meet local regulatory standards. India's well-established pharmaceutical manufacturing sector, known for producing high-quality generics, aligns with Cameskin/Cordaid's procurement needs, ensuring a reliable supply of essential medications.
3Market Positioning
Based on its product mix, Cameskin/Cordaid primarily serves the wholesale distribution segment of the DRC's pharmaceutical market. By importing and wholesaling essential tuberculosis medications, they supply local distributors, pharmacies, and healthcare facilities, ensuring the availability of critical treatments across the country. Their strategic focus on high-demand therapeutic areas positions them as a key player in addressing public health challenges within the DRC.
Seller's Guide — How to Become a Supplier to Cameskin/cordaid
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Cameskin/Cordaid, given the company's concentrated sourcing strategy and the substantial demand for tuberculosis medications in the DRC. Potential gaps in Cameskin/Cordaid's current sourcing include the need for additional suppliers to mitigate risks associated with supply chain disruptions. New suppliers can offer competitive pricing, product diversification, and enhanced supply chain resilience, making them attractive partners for Cameskin/Cordaid.
2Requirements & Qualifications
Indian exporters seeking to supply Cameskin/Cordaid and the DRC market must adhere to several requirements:
- Import License: Obtain an import license from an authorized commercial bank in the DRC.
- Pre-Shipment Inspection: Ensure that shipments valued over $2,500 undergo inspection by Bureau Veritas.
- Certificate of Pharmaceutical Product (CPP): Provide a CPP to demonstrate compliance with international quality standards.
- Regulatory Compliance: Ensure that products are not on the DRC's banned import list and comply with all local regulations, including labeling requirements in French.
Adhering to these standards is crucial for successful market entry and sustained business operations in the DRC.
3How to Approach
To establish a partnership with Cameskin/Cordaid, Indian exporters should:
- Build Relationships: Engage in direct communication to understand Cameskin/Cordaid's specific needs and requirements.
- Participate in Tenders: Monitor and respond to relevant tenders issued by Cameskin/Cordaid, demonstrating the value and quality of their products.
- Regulatory Filing Strategy:
Frequently Asked Questions — Cameskin/cordaid
What products does Cameskin/cordaid import from India?
Cameskin/cordaid imports 3 pharmaceutical products across 2 categories. Top imports: Isoniazid ($500.0K), Ethambutol ($450.0K), Rifampicin ($300.0K).
Who supplies pharmaceuticals to Cameskin/cordaid from India?
Cameskin/cordaid sources from 3 verified Indian suppliers. The primary supplier is Lupin Limited (51.0% of imports, $668.7K).
What is Cameskin/cordaid's total pharmaceutical import value?
Cameskin/cordaid's total pharmaceutical import value from India is $1.3M, based on 25 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Cameskin/cordaid focus on?
Cameskin/cordaid imports across 2 categories. The largest: Tuberculosis Medications (76.0%), Advanced Antibiotics (24.0%).
Get Full Cameskin/cordaid Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Cameskin/cordaid identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Cameskin/cordaid's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 25 individual customs records matching Cameskin/cordaid.
- 5.Supplier Verification: Cameskin/cordaid sources from 3 verified Indian suppliers across 13 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.