Arrow Pharmaceuticals Pty Limited
Pharmaceutical Importer · Australia · Cardiovascular Focus · $15.6M Total Trade · DGFT Verified
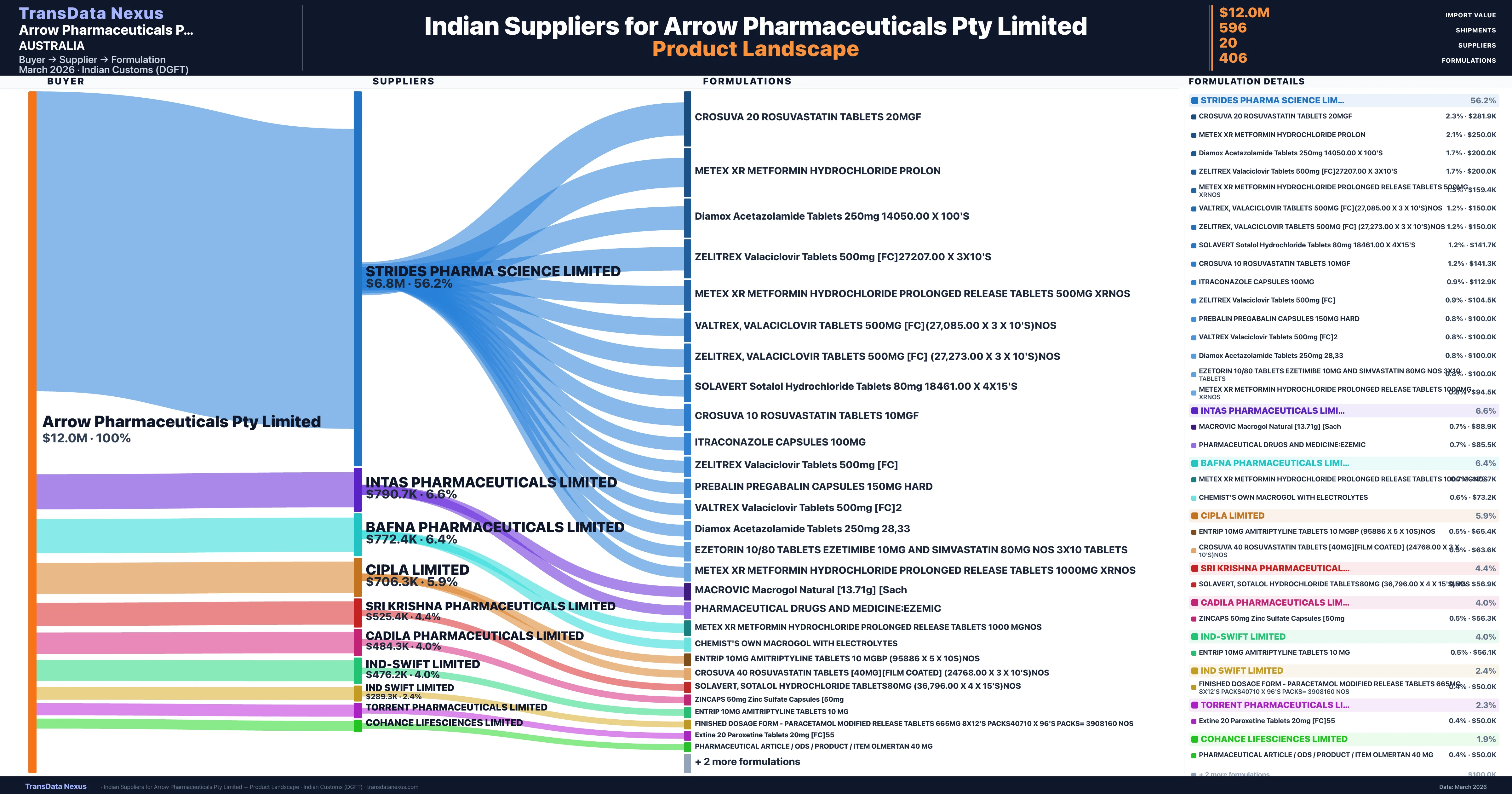
Arrow Pharmaceuticals Pty Limited is a pharmaceutical importer based in Australia with a total trade value of $15.6M across 10 products in 6 therapeutic categories. Based on 410 verified import shipments from Indian Customs (DGFT) records, Arrow Pharmaceuticals Pty Limited is the #1 buyer in 1 product including Rosuvastatin. Arrow Pharmaceuticals Pty Limited sources from 20 verified Indian suppliers, with Strides Pharma Science Limited accounting for 56.2% of imports.
Arrow Pharmaceuticals Pty Limited — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Arrow Pharmaceuticals Pty Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Strides Pharma Science Limited | $6.8M | 232 | 56.2% |
| Intas Pharmaceuticals Limited | $790.7K | 42 | 6.6% |
| Bafna Pharmaceuticals Limited | $772.4K | 28 | 6.4% |
| Cipla Limited | $706.3K | 144 | 5.9% |
| Sri Krishna Pharmaceuticals Limited | $525.4K | 22 | 4.4% |
| Cadila Pharmaceuticals Limited | $484.3K | 26 | 4.0% |
| Ind-swift Limited | $476.2K | 19 | 4.0% |
| Ind Swift Limited | $289.3K | 21 | 2.4% |
| Torrent Pharmaceuticals Limited | $271.1K | 6 | 2.3% |
| Cohance Lifesciences Limited | $225.1K | 7 | 1.9% |
| Ipca Laboratories Limited | $195.3K | 7 | 1.6% |
| Alkem Laboratories Limited | $167.9K | 10 | 1.4% |
| Macleods Pharmaceuticals Limited | $150.9K | 8 | 1.3% |
| Tenshi Kaizen Private Limited | $57.8K | 4 | 0.5% |
| Vergo Pharma Research Laboratories Private Limited | $56.4K | 13 | 0.5% |
| Msn Laboratories Private Limited | $50.0K | 1 | 0.4% |
| Fdc Limited | $30.4K | 1 | 0.3% |
| Xl Laboratories Private Limited | $16.1K | 3 | 0.1% |
| Sunglow Lifescience Private Limited | $7.9K | 1 | 0.1% |
| Mepro Pharmaceuticals Private Limited | $6.6K | 1 | 0.1% |
Arrow Pharmaceuticals Pty Limited sources from 20 verified Indian suppliers across 406 distinct formulations. The supply base is diversified across 20 suppliers, reducing single-source dependency risk.
What Formulations Does Arrow Pharmaceuticals Pty Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Crosuva 20 rosuvastatin tablets | $281.9K | 6 |
| Metex xr metformin hydrochloride | $250.0K | 5 |
| Diamox acetazolamide tablets 250MG 14050.00 x | $200.0K | 4 |
| Zelitrex valaciclovir tablets 500MG [fc]27207.00 x | $200.0K | 4 |
| Metex xr metformin hydrochloride prolonged release tablets 500MG | $159.4K | 6 |
| Valtrex, valaciclovir tablets 500MG [fc](27,085.00 x 3 x 10's | $150.0K | 3 |
| Zelitrex, valaciclovir tablets 500MG [fc] (27,273.00 x 3 x 10's | $150.0K | 3 |
| Solavert sotalol hydrochloride tablets 80MG 18461.00 x | $141.7K | 5 |
| Crosuva 10 rosuvastatin tablets | $141.3K | 4 |
| Itraconazole capsules 100MG | $112.9K | 3 |
| Zelitrex valaciclovir tablets 500MG | $104.5K | 4 |
| Prebalin pregabalin capsules 150MG | $100.0K | 2 |
| Valtrex valaciclovir tablets 500MG | $100.0K | 2 |
| Diamox acetazolamide tablets 250MG 28 | $100.0K | 2 |
| Ezetorin 10/80 tablets ezetimibe 10MG and simvastatin 80MG nos 3x10 | $100.0K | 2 |
Arrow Pharmaceuticals Pty Limited imports 406 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Arrow Pharmaceuticals Pty Limited Import?
Arrow Pharmaceuticals Pty Limited Therapeutic Categories — 6 Specializations
Arrow Pharmaceuticals Pty Limited imports across 6 therapeutic categories, with Cardiovascular (58.4%), Ayurvedic & Herbal Products (25.8%), Lipid & Metabolism (4.5%) representing the largest segments. The portfolio is concentrated — top 5 products = 86% of total imports.
Cardiovascular
4 products · 58.4% · $9.1M
Ayurvedic & Herbal Products
1 products · 25.8% · $4.0M
Lipid & Metabolism
1 products · 4.5% · $700.0K
Advanced Antibiotics
2 products · 4.3% · $667.8K
Immunosuppressants
1 products · 3.8% · $586.8K
Antimalarial & Antiparasitic
1 products · 3.2% · $498.3K
Import Portfolio — Top 10 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Rosuvastatin | Cardiovascular | $7.0M | 141 | 0.7% | 1 |
| 2 | Natural | Ayurvedic & Herbal Products | $4.0M | 114 | 5.4% | 3 |
| 3 | Pravastatin | Cardiovascular | $888.1K | 46 | 2.8% | 7 |
| 4 | Simvastatin | Cardiovascular | $752.9K | 30 | 0.6% | 11 |
| 5 | Fenofibrate | Lipid & Metabolism | $700.0K | 14 | 1.0% | 8 |
| 6 | Methotrexate | Immunosuppressants | $586.8K | 13 | 1.7% | 12 |
| 7 | Quinine | Antimalarial & Antiparasitic | $498.3K | 12 | 2.6% | 5 |
| 8 | Rivaroxaban | Cardiovascular | $400.0K | 8 | 0.8% | 13 |
| 9 | Nitrofurantoin | Advanced Antibiotics | $350.0K | 7 | 1.0% | 11 |
| 10 | Roxithromycin | Advanced Antibiotics | $317.8K | 25 | 3.6% | 7 |
Arrow Pharmaceuticals Pty Limited imports 10 pharmaceutical products across 6 categories into Australia totaling $15.6M. The company is the #1 buyer for 1 product: Rosuvastatin.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Arrow Pharmaceuticals Pty Limited.
Request DemoArrow Pharmaceuticals Pty Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Arrow Pharmaceuticals Pty Limited is an Australian pharmaceutical company specializing in the importation and distribution of generic prescription medications and over-the-counter (OTC) products. Established in 2015, the company re-entered the Australian market following Strides' acquisition of the Chemists' Own Brand and Aspen Australia Generics business. This strategic move positioned Arrow Pharmaceuticals as a key player in the pharmaceutical industry, leveraging the global strength of Strides to support Australian retail pharmacies and the communities they serve. (arrowpharma.com.au)
Headquartered in Cremorne, Victoria, Australia, Arrow Pharmaceuticals operates under the regulatory oversight of the Therapeutic Goods Administration (TGA), ensuring compliance with Australian health standards. The company's portfolio includes a growing range of products aimed at meeting the diverse needs of Australian pharmacies, with a focus on affordability and quality. (arrowpharma.com.au)
2Distribution Network
Arrow Pharmaceuticals' distribution network is strategically designed to ensure efficient delivery of pharmaceutical products across Australia. While specific warehouse locations are not publicly disclosed, the company's operations are supported by a dedicated team of industry professionals experienced in research and development, business development, professional services, sales, and marketing. This team collaborates to enhance the future of Australian retail pharmacies and the communities they serve. (arrowpharma.com.au)
3Industry Role
In Australia's pharmaceutical supply chain, Arrow Pharmaceuticals Pty Limited functions primarily as a wholesaler and distributor. By importing generic prescription medications and OTC products, the company supplies these goods to Australian pharmacies, thereby enhancing the availability of affordable and high-quality medicines. This role is crucial in supporting the healthcare needs of the Australian population. (arrowpharma.com.au)
Supplier Relationship Intelligence — Arrow Pharmaceuticals Pty Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Arrow Pharmaceuticals demonstrates a significant concentration in its sourcing strategy, with a portfolio heavily reliant on a select group of products. The top five imported products—Rosuvastatin, Natural, Pravastatin, Simvastatin, and Fenofibrate—account for 86.2% of the total import value. This concentration indicates a strategic focus on high-demand cardiovascular and lipid metabolism medications. However, such dependency on a limited product range may expose the company to risks associated with market fluctuations, regulatory changes, or supply chain disruptions affecting these specific products.
The shipment data reveals a stable relationship with key Indian suppliers, particularly STRIDES PHARMA SCIENCE LIMITED, which accounts for 56.2% of the total import value. This suggests a strong and reliable partnership, potentially offering favorable terms and consistent product quality. Nonetheless, the heavy reliance on a single supplier for a substantial portion of imports could pose challenges if issues arise within that supplier's operations.
2Supply Chain Resilience
Arrow Pharmaceuticals' supply chain resilience is closely tied to its sourcing strategy. The company's significant reliance on STRIDES PHARMA SCIENCE LIMITED for a major portion of its imports indicates a strong partnership but also highlights potential vulnerabilities. While the data does not specify the presence of backup suppliers, the diversity of formulations imported—406 unique formulations—suggests a degree of flexibility in sourcing. However, the concentration in specific therapeutic categories, such as cardiovascular and lipid metabolism, may limit the company's ability to quickly adapt to changes in the supply chain. Additionally, the exposure to international shipping routes and the regulatory compliance of key suppliers are critical factors influencing the overall resilience of the supply chain.
3Strategic Implications
The sourcing pattern of Arrow Pharmaceuticals, characterized by a heavy reliance on a limited number of products and suppliers, has significant strategic implications. For Arrow Pharmaceuticals, this focus allows for specialization and potentially better-negotiated terms with suppliers. However, it also exposes the company to risks associated with supply chain disruptions, regulatory changes, or market shifts affecting these specific products.
For Indian exporters, understanding Arrow Pharmaceuticals' sourcing strategy presents an opportunity to become alternative suppliers, especially for products where the company seeks diversification. By offering high-quality alternatives in the therapeutic categories of interest, Indian exporters can position themselves as viable partners, potentially reducing Arrow Pharmaceuticals' dependency on a limited supplier base.
Importing Pharmaceuticals into Australia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Australia
1Regulatory Authority & Framework
In Australia, the regulation of pharmaceutical imports is governed by the Therapeutic Goods Administration (TGA), operating under the Therapeutic Goods Act 1989. This legislation establishes a uniform, national system of regulatory controls to ensure the quality, safety, efficacy, and timely availability of therapeutic goods for human use. The TGA is responsible for the pre-market evaluation and approval of products intended for supply in Australia, the licensing of pharmaceutical manufacturers, and post-market surveillance. (business.gov.au)
2Import Licensing & GMP
Import licensing requirements in Australia are stringent, with all pharmaceutical products imported into the country required to comply with the Therapeutic Goods Act 1989. This includes obtaining approvals for the importation, exportation, or supply of therapeutic goods. Manufacturers must hold a valid license, and products must be included in the Australian Register of Therapeutic Goods (ARTG) unless specifically exempted. Good Manufacturing Practice (GMP) certificates recognized by the TGA, such as EU GMP, WHO GMP, or PIC/S, are essential for ensuring product quality and safety. (business.gov.au)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet Australian standards. Stability requirements are enforced to guarantee that products remain effective throughout their shelf life. Labeling must comply with the Therapeutic Goods Order No. 91, which outlines specific requirements, including the display of the country of origin and a true description of the product. Additionally, labeling must be in English and include necessary information such as dosage instructions, active ingredients, and storage conditions. (abf.gov.au)
4Recent Regulatory Changes
Between 2024 and 2026, several policy changes have affected the importation of Indian pharmaceutical products into Australia. The TGA has implemented stricter compliance measures, including enhanced batch testing protocols and more rigorous stability requirements. Labeling regulations have been updated to ensure clearer communication of product information to consumers. These changes aim to improve the safety and efficacy of imported pharmaceuticals and may impact the operations of companies like Arrow Pharmaceuticals.
Arrow Pharmaceuticals Pty Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Arrow Pharmaceuticals' product strategy focuses on therapeutic areas with high demand, particularly cardiovascular and lipid metabolism disorders. The company's top five imported products—Rosuvastatin, Natural, Pravastatin, Simvastatin, and Fenofibrate—align with this focus, collectively accounting for 86.2% of the total import value. This strategic emphasis is driven by the prevalence of cardiovascular diseases in Australia and the need for affordable, effective treatments. By concentrating on these therapeutic areas, Arrow Pharmaceuticals aims to meet the significant market demand and support the healthcare needs of the Australian population.
2Sourcing Profile
Arrow Pharmaceuticals' sourcing strategy is centered on importing generic formulations from India, with a particular emphasis on cardiovascular and lipid metabolism medications. The company's reliance on a select group of products and suppliers indicates a strategic choice to focus on high-demand therapeutic areas. India's established pharmaceutical manufacturing capabilities, recognized GMP certifications, and cost-effectiveness make it a suitable source for these products. This approach allows Arrow Pharmaceuticals to offer affordable and high-quality medications to the Australian market.
3Market Positioning
Based on its product mix, Arrow Pharmaceuticals primarily serves the wholesale distribution segment of the Australian market. By importing and supplying generic prescription medications and OTC products, the company supports Australian pharmacies in meeting the diverse healthcare needs of their customers. This positioning enables Arrow Pharmaceuticals to play a crucial role in enhancing the availability and affordability of essential medicines across Australia.
Seller's Guide — How to Become a Supplier to Arrow Pharmaceuticals Pty Limited
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Arrow Pharmaceuticals, particularly in therapeutic areas where the company seeks diversification. By offering high-quality alternatives in cardiovascular and lipid metabolism medications, Indian exporters can position themselves as viable partners, potentially reducing Arrow Pharmaceuticals' dependency on a limited supplier base. Additionally, the evolving regulatory landscape in Australia may create openings for suppliers who can navigate compliance requirements effectively.
2Requirements & Qualifications
Indian exporters aiming to supply Arrow Pharmaceuticals and the Australian market must adhere to the TGA's import licensing requirements. This includes obtaining approvals for the importation and supply of therapeutic goods and ensuring that products are included in the ARTG unless exempted. Manufacturers must hold valid GMP certificates recognized by the TGA, such as EU GMP, WHO GMP, or PIC/S. Additionally, products must comply with Australian labeling requirements, including English language labeling and adherence to Therapeutic Goods Order No. 91. (business.gov.au)
3How to Approach
To establish a relationship with Arrow Pharmaceuticals, Indian exporters should first ensure compliance with the TGA's regulatory requirements, including obtaining necessary approvals and certifications. Engaging in direct communication with Arrow Pharmaceuticals to understand their specific product needs and quality standards is essential. Participating in relevant industry tend
Frequently Asked Questions — Arrow Pharmaceuticals Pty Limited
What products does Arrow Pharmaceuticals Pty Limited import from India?
Arrow Pharmaceuticals Pty Limited imports 10 pharmaceutical products across 6 categories. Top imports: Rosuvastatin ($7.0M), Natural ($4.0M), Pravastatin ($888.1K), Simvastatin ($752.9K), Fenofibrate ($700.0K).
Who supplies pharmaceuticals to Arrow Pharmaceuticals Pty Limited from India?
Arrow Pharmaceuticals Pty Limited sources from 20 verified Indian suppliers. The primary supplier is Strides Pharma Science Limited (56.2% of imports, $6.8M).
What is Arrow Pharmaceuticals Pty Limited's total pharmaceutical import value?
Arrow Pharmaceuticals Pty Limited's total pharmaceutical import value from India is $15.6M, based on 410 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Arrow Pharmaceuticals Pty Limited focus on?
Arrow Pharmaceuticals Pty Limited imports across 6 categories. The largest: Cardiovascular (58.4%), Ayurvedic & Herbal Products (25.8%), Lipid & Metabolism (4.5%).
Get Full Arrow Pharmaceuticals Pty Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Arrow Pharmaceuticals Pty Limited identified across shipments using consignee name normalization, aggregating 5 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Arrow Pharmaceuticals Pty Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 410 individual customs records matching Arrow Pharmaceuticals Pty Limited.
- 5.Supplier Verification: Arrow Pharmaceuticals Pty Limited sources from 20 verified Indian suppliers across 406 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
10 Products Tracked
6 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 5 company name variants from customs records. For current shipment-level data, contact TransData Nexus.