In June 2025, the NPPA announced a revision of the ceiling prices for several non-scheduled drugs, leading to a 5% increase in the maximum retail price of certain OTC products. Although "Volini" is not under price control, this adjustment may influence market dynamics and pricing strategies.
In September 2025, the CDSCO issued new guidelines for the export of pharmaceutical products, emphasizing the need for stringent quality checks and compliance with Good Manufacturing Practices (GMP). These guidelines aim to enhance the credibility of Indian pharmaceutical exports and may impact the export processes for products like "Volini."
In December 2025, the WHO updated its Model List of Essential Medicines, adding several new medications but not including "Volini." This exclusion indicates that "Volini" is not considered essential for basic healthcare systems by the WHO, which may affect its adoption in certain markets.
In February 2026, the EMA introduced a new electronic product information (ePI) implementation guide to standardize the submission of product information. While "Volini" does not have EMA approval, this development highlights the increasing emphasis on digitalization in regulatory processes.
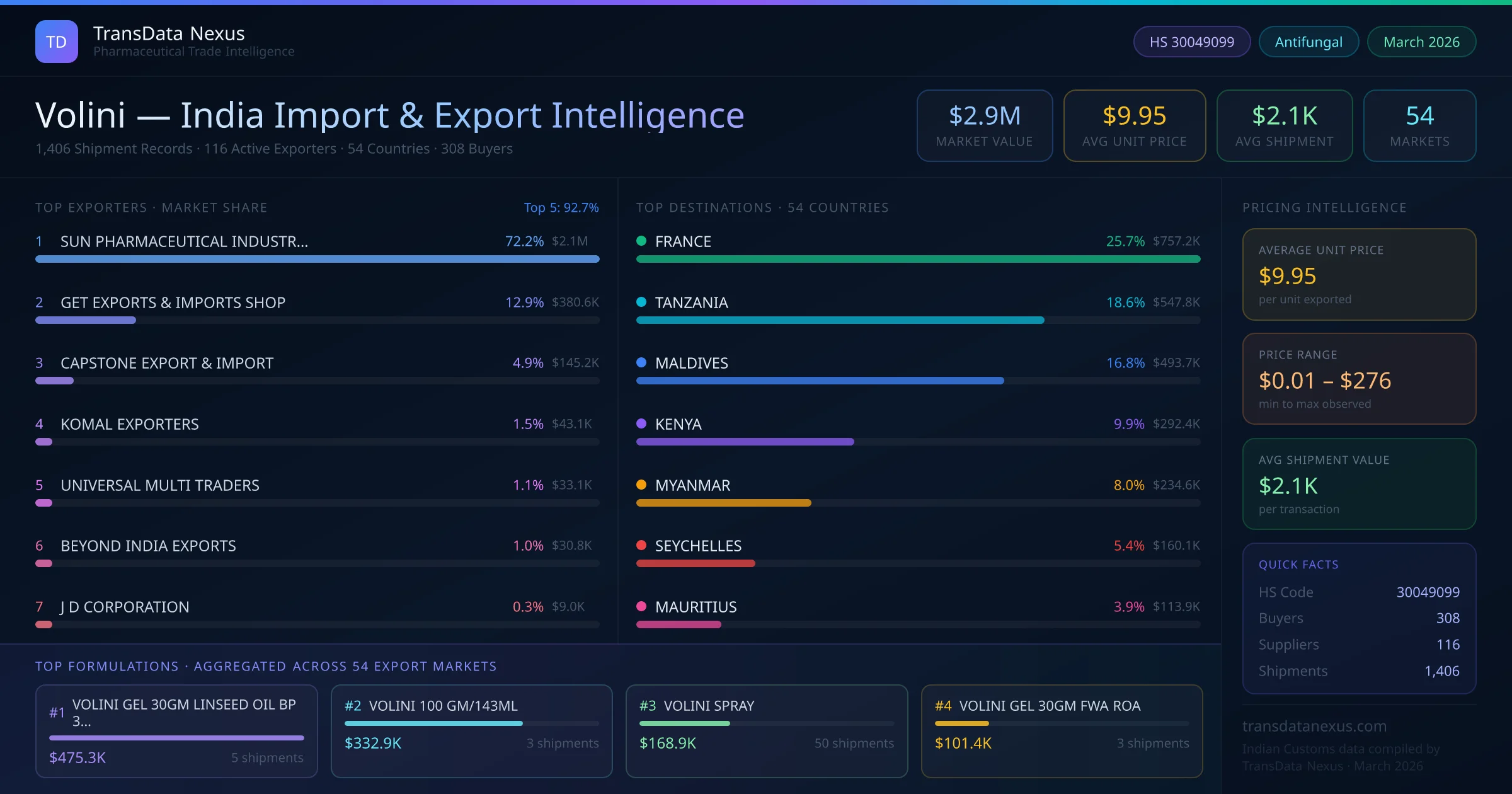
In March 2026, the DGFT reported a 10% increase in pharmaceutical exports from India compared to the previous year. This growth reflects the expanding global reach of Indian pharmaceutical products, including "Volini," which has been exported to 54 countries with a total export value of $2.9 million USD.
These developments underscore the dynamic nature of the pharmaceutical regulatory landscape and the importance of staying informed to navigate the complexities of international markets.