United Arab Emirates to India Pharmaceutical Import
Bilateral Trade Intelligence · $1.3M Total Trade · 17 Foreign Suppliers · 23 Indian Buyers · DGFT Verified · Updated March 2026
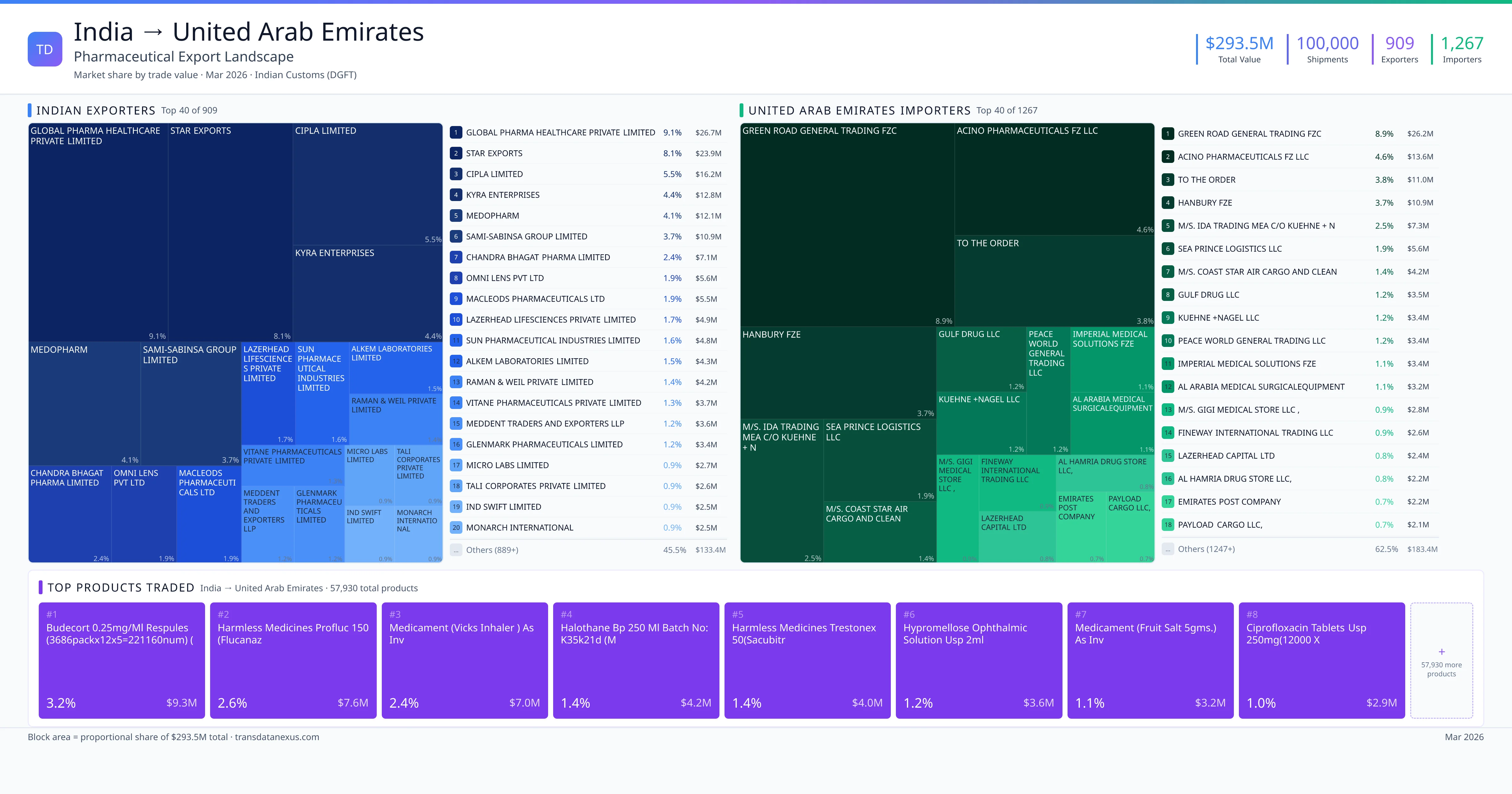
India imported $1.3M worth of pharmaceutical formulations from United Arab Emirates across 162 verified shipments, from 17 foreign suppliers to 23 Indian buyers. The top suppliers are SAVI CLINICALS FZ LLC ($778.4K) and FORWARD WAVE EQUIPMENT TRADING ($318.9K). The leading products are Semaglutide ($320K) and Ibrutinib ($197K). Average shipment value: $8.0K.
Top Pharmaceutical Formulations — United Arab Emirates to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Semaglutide | $320.2K | 24.9% |
| 2 | Ibrutinib | $197.0K | 15.3% |
| 3 | Pantoprazole | $182.1K | 14.1% |
| 4 | Osimertinib | $137.7K | 10.7% |
| 5 | Carfilzomib | $59.4K | 4.6% |
| 6 | Potassium | $49.0K | 3.8% |
| 7 | Amoxicillin | $47.7K | 3.7% |
| 8 | Ceftriaxone | $41.4K | 3.2% |
| 9 | Pembrolizumab | $35.8K | 2.8% |
| 10 | Sofosbuvir | $29.9K | 2.3% |
| 11 | Velpatasvir | $29.9K | 2.3% |
| 12 | Ivermectin | $19.4K | 1.5% |
| 13 | Dolutegravir | $18.2K | 1.4% |
| 14 | Pemetrexed | $17.8K | 1.4% |
| 15 | Sodium | $17.7K | 1.4% |
India imports 20+ pharmaceutical formulations from United Arab Emirates with a combined trade value of $1.3M. Key products include Semaglutide ($320K), Ibrutinib ($197K), Pantoprazole ($182K), Osimertinib ($138K), Carfilzomib ($59K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 162 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — United Arab Emirates to India Trade Routes
These are the top pharmaceutical products exported from India to United Arab Emirates, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, United Arab Emirates buyers, regulatory requirements, and logistics for that specific product corridor. Products include Semaglutide ($320K), Ibrutinib ($197K), Pantoprazole ($182K), Osimertinib ($138K), Carfilzomib ($59K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top United Arab Emirates Suppliers to India
17 Indian pharmaceutical companies export finished formulations to United Arab Emirates. Leading exporters include Savi Clinicals Fz Llc, Forward Wave Equipment Trading, Savi Clinicals Fz Llc, Aadi International Fzco. The top exporter accounts for 60.4% of total India–United Arab Emirates pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from United Arab Emirates
23 companies in United Arab Emirates import pharmaceutical formulations from India. Top buyers include Msn Laboratories Private Limited, Chandra Bhagat Pharma Limited, Hetero Labs Limited, Biological E. Limited. The largest buyer accounts for 50.2% of India–United Arab Emirates pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–United Arab Emirates Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between the United Arab Emirates (UAE) and India has experienced notable growth from 2022 to 2026. In 2022, the total import value was $1.3 million USD, with 162 shipments from 17 UAE suppliers to 23 Indian buyers. By 2026, this value increased to $5.53 million USD, indicating a significant expansion in trade volume. This growth reflects the strengthening of bilateral trade relations and the UAE's growing role as a key supplier of pharmaceutical products to India.
2United Arab Emirates's Role in India's Pharma Imports
The UAE's contribution to India's pharmaceutical imports remains modest but is on an upward trajectory. In 2024, the UAE exported pharmaceutical products worth $5.53 million USD to India, accounting for a small fraction of India's total pharmaceutical imports. India imports a diverse range of pharmaceutical products from the UAE, including finished formulations such as tablets, capsules, syrups, and injections. This diversity indicates the UAE's capability to supply a broad spectrum of pharmaceutical products to meet India's market demands.
3Recent Developments
Between 2024 and 2026, there have been significant developments in the pharmaceutical trade between the UAE and India. The Central Drugs Standard Control Organisation (CDSCO) has streamlined the registration process for foreign pharmaceutical products, facilitating easier market entry for UAE suppliers. Additionally, trade agreements between the two nations have been enhanced, leading to preferential duty rates and improved market access. These developments have contributed to the growth in pharmaceutical imports from the UAE to India.
IMPORT_REGULATORY
United Arab Emirates Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
UAE pharmaceutical companies seeking to export products to India must navigate the CDSCO registration process. This involves obtaining an Import License (Form 10) and a Registration Certificate for each product. The process includes submitting an application with detailed product information, manufacturing details, and quality control measures. Clinical trial data may be required, depending on the product's classification. The timeline for registration varies but typically ranges from several months to a year, depending on the complexity of the product and the completeness of the application.
2GMP & Manufacturing Standards
India requires that pharmaceutical products imported from the UAE adhere to Good Manufacturing Practice (GMP) standards. Manufacturing sites must be WHO-GMP certified, ensuring compliance with international quality standards. The CDSCO conducts inspections to verify adherence to these standards, assessing facilities, processes, and quality control measures. This rigorous evaluation ensures that imported pharmaceutical products are safe and effective for the Indian market.
3Import Documentation
To import pharmaceutical products into India, UAE companies must provide several key documents:
- Import License (Form 10): Authorizes the importation of pharmaceutical products into India.
- Registration Certificate: Confirms that the product is approved for sale in India.
- No Objection Certificate (NOC): Issued by the UAE regulatory authority, indicating no objection to the export of the product.
- Test License: Permits the testing of imported pharmaceutical products in India.
- Customs Documentation: Includes invoices, packing lists, and shipping documents required for customs clearance.
These documents ensure compliance with Indian regulations and facilitate the smooth importation of pharmaceutical products.
IMPORT_TRENDS
Product Categories & Therapeutic Trends — India to United Arab Emirates
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
India's pharmaceutical imports from the UAE are dominated by specific therapeutic areas and product types. Notably, the top imported products include Semaglutide, Ibrutinib, and Pantoprazole, which are used in the treatment of diabetes, cancer, and gastrointestinal disorders, respectively. This indicates a significant demand for specialized and high-value pharmaceutical products from the UAE, reflecting India's focus on advanced therapies to address complex health conditions.
2Innovation & Specialty Imports
The UAE exports several innovative and specialty pharmaceutical products to India that are not manufactured domestically. These include novel biologics and targeted therapies such as Osimertinib and Carfilzomib, which are used in the treatment of specific types of cancer. The importation of these advanced therapies underscores India's reliance on international suppliers for cutting-edge treatments that are essential for managing complex diseases.
3Import Demand Drivers
India's importation of pharmaceutical products from the UAE is driven by several factors:
- Patent Protection: The UAE offers patented drugs that are not yet available in India, fulfilling unmet medical needs.
- Technology Gaps: The UAE's advanced manufacturing capabilities provide products with superior quality and efficacy.
- Quality Requirements: The UAE's adherence to international quality standards ensures that imported products meet India's stringent regulatory requirements.
- Disease Burden: The prevalence of certain diseases in India necessitates the importation of specialized treatments not produced domestically.
These factors collectively contribute to the sustained demand for pharmaceutical imports from the UAE.
IMPORT_POLICY
Trade Policy & Tariff Intelligence — India and United Arab Emirates
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes the following components:
- Most Favored Nation (MFN) Tariff Rates: These are the standard import duty rates applied to pharmaceutical products from all WTO member countries, including the UAE.
- Basic Customs Duty (BCD): A percentage of the product's value, levied on imported goods.
- Integrated Goods and Services Tax (IGST): Applied to the total value of the imported goods, including BCD.
- Health Cess: An additional levy imposed on certain pharmaceutical products to fund health initiatives.
These tariffs are subject to change based on government policies and trade agreements.
2Trade Agreements & Preferences
India and the UAE have engaged in bilateral trade agreements aimed at enhancing economic cooperation. While there is no comprehensive Free Trade Agreement (FTA) between the two countries, preferential duty rates have been established for certain products, including pharmaceuticals. These agreements facilitate smoother trade flows and provide mutual benefits, encouraging increased pharmaceutical imports from the UAE to India.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, allows for the granting of compulsory licenses under specific conditions, such as public health needs. The National Pharmaceutical Pricing Authority (NPPA) regulates the prices of essential drugs, including imported pharmaceuticals, to ensure affordability. These policies aim to balance the interests of patent holders and the accessibility of medicines to the Indian population.
IMPORT_LOGISTICS
Supply Chain & Logistics — India to United Arab Emirates Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical products from the UAE to India are primarily transported via sea and air routes. Major sea routes include shipments from the Port of Jebel Ali in Dubai to Indian ports such as Mumbai and Chennai. Air shipments typically arrive at international airports like Indira Gandhi International Airport in Delhi and Chhatrapati Shivaji Maharaj International Airport in Mumbai. Transit times vary, with sea shipments taking approximately 7-10 days and air shipments around 3-5 days, depending on the specific ports and logistics arrangements.
2Port Infrastructure
The Port of Jebel Ali in Dubai is a key export hub for the UAE's pharmaceutical products, offering advanced facilities and connectivity. In India, major import ports handling pharmaceutical shipments include Jawaharlal Nehru Port Trust (JNPT) in Mumbai and Chennai Port. These ports are equipped with specialized facilities for handling pharmaceutical goods, ensuring efficient processing and distribution within India.
3Indian Customs & Cold Chain
Upon arrival at Indian ports, pharmaceutical imports undergo customs clearance, which includes verification of documentation and compliance with CDSCO regulations. Imported pharmaceutical products are subject to testing by the CDSCO to ensure they meet Indian quality standards. Maintaining the cold chain is critical for temperature-sensitive products; therefore, logistics providers must adhere to Good Distribution Practice (GDP) guidelines to ensure product integrity throughout the transit process.
IMPORT_OPPORTUNITY
Market Opportunity Assessment — United Arab Emirates for Indian Pharma
Market size, healthcare system, growth outlook
1India's Import Dependency
Despite being a major pharmaceutical manufacturer, India imports specific products from the UAE due to factors such as patent protection, technology gaps, and the need for specialized treatments not produced domestically. The UAE's advanced manufacturing capabilities and adherence to international quality standards make it a valuable source for these specialized pharmaceutical products.
2United Arab Emirates Companies in India
Several UAE-based pharmaceutical companies have established a presence in India, either through direct exports or by forming partnerships with Indian firms. These collaborations aim to leverage local manufacturing capabilities, enhance distribution networks, and navigate regulatory requirements effectively. Such strategies enable UAE companies to expand their market reach and cater to the diverse needs of the Indian pharmaceutical market.
3Outlook & Self-Reliance
India's Atmanirbhar Bharat initiative and the Production-Linked Incentive (PLI) scheme are designed to boost domestic pharmaceutical manufacturing. While these
FAQ — India to United Arab Emirates Pharmaceutical Trade
What is the total value of India's pharmaceutical export to United Arab Emirates?
India exported pharmaceuticals worth $1.3M to United Arab Emirates across 162 verified shipments.
Who are the top Indian pharmaceutical exporters to United Arab Emirates?
1. SAVI CLINICALS FZ LLC — $778.4K. 2. FORWARD WAVE EQUIPMENT TRADING — $318.9K. 3. SAVI Clinicals FZ LLC — $107.0K. Total: 17 suppliers.
Which companies in United Arab Emirates import pharmaceuticals from India?
1. MSN LABORATORIES PRIVATE LIMITED — $646.4K. 2. CHANDRA BHAGAT PHARMA LIMITED — $318.9K. 3. HETERO LABS LIMITED — $101.7K. 23 buyers total.
What pharmaceutical products does India export most to United Arab Emirates?
1. Semaglutide ($320.2K, 24.9%); 2. Ibrutinib ($197.0K, 15.3%); 3. Pantoprazole ($182.1K, 14.1%); 4. Osimertinib ($137.7K, 10.7%); 5. Carfilzomib ($59.4K, 4.6%)
Which ports handle pharmaceutical shipments from India to United Arab Emirates?
Export: . Import: .
Why does United Arab Emirates import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $1.3M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply United Arab Emirates?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to United Arab Emirates pharmaceutical trade?
$8.0K per consignment across 162 shipments.
How many Indian pharmaceutical companies export to United Arab Emirates?
17 Indian companies. Largest: SAVI CLINICALS FZ LLC with $778.4K.
How can I find verified Indian pharmaceutical suppliers for United Arab Emirates?
TransData Nexus covers 17 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to United Arab Emirates Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 162 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to United Arab Emirates identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 162 verified shipments from 17 Indian exporters to 23 United Arab Emirates buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
17 Exporters
23 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists