In August 2025, the FDA issued updated guidance on the labeling of dietary supplements, emphasizing the need for clear differentiation between structure/function claims and disease claims to prevent misleading consumers. This development is particularly relevant for probiotic manufacturers aiming to market their products in the U.S.
In November 2025, the European Commission published a report evaluating the safety and efficacy of probiotics in food products. The report concluded that while probiotics are generally safe for consumption, there is insufficient evidence to substantiate many health claims, reinforcing the need for compliance with existing EU regulations on health claims.
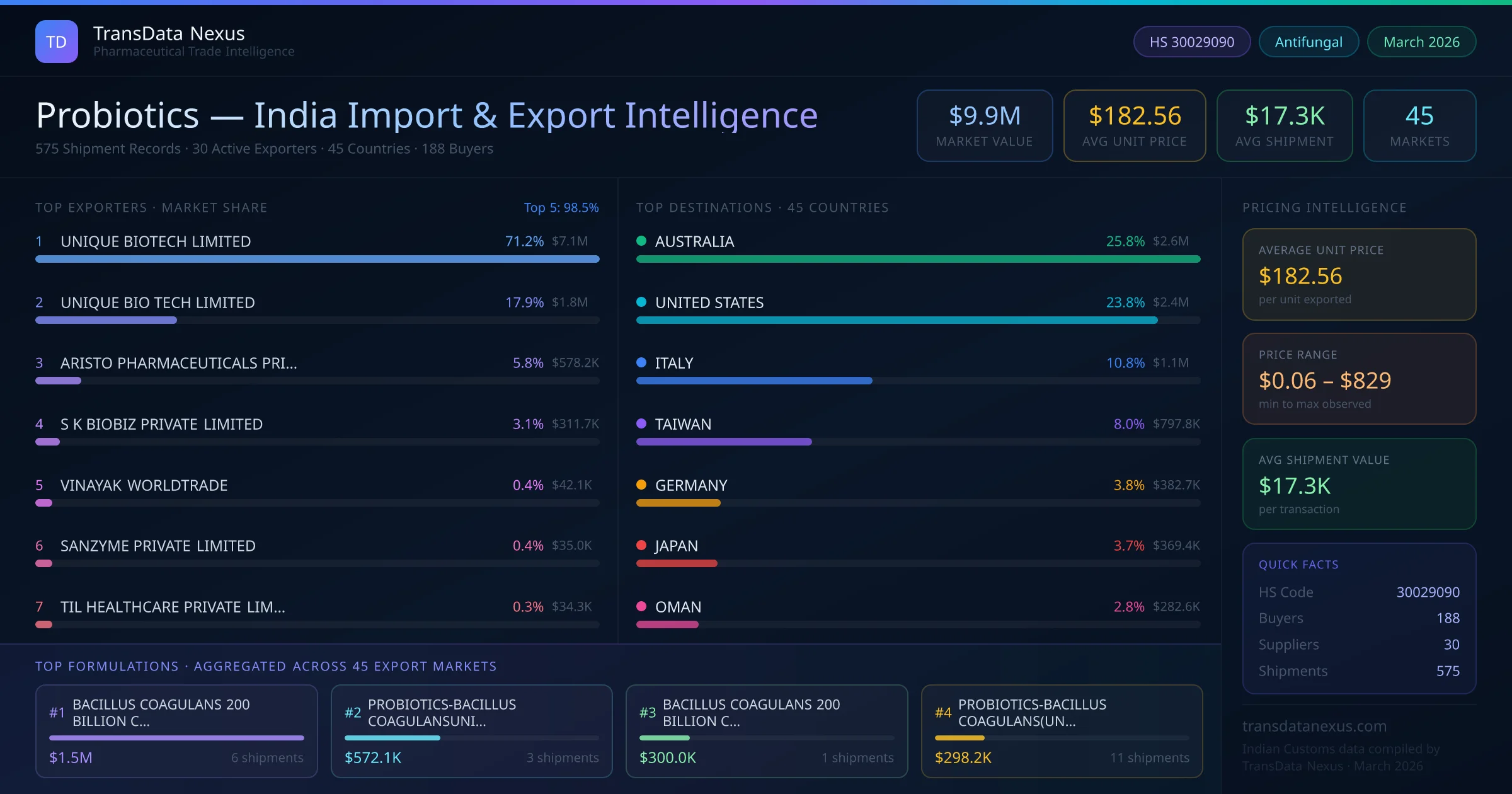
In January 2026, the FSSAI introduced new guidelines for the manufacturing and labeling of probiotics in India, aligning domestic standards more closely with international norms. These guidelines aim to enhance product quality and facilitate smoother export processes for Indian manufacturers.
In February 2026, the Australian Therapeutic Goods Administration (TGA) updated its list of permitted ingredients for listed medicines, including specific strains of probiotics. This update affects Indian exporters, as Australia accounts for 25.8% of India's probiotic exports.
In March 2026, the U.S. Department of Agriculture (USDA) reported a 5% increase in the import of dietary supplements, including probiotics, over the previous year. This trend indicates a growing market demand in the U.S., presenting opportunities for Indian exporters.