Netherlands to India Pharmaceutical Import
Bilateral Trade Intelligence · $232.7M Total Trade · 120 Foreign Suppliers · 95 Indian Buyers · DGFT Verified · Updated March 2026
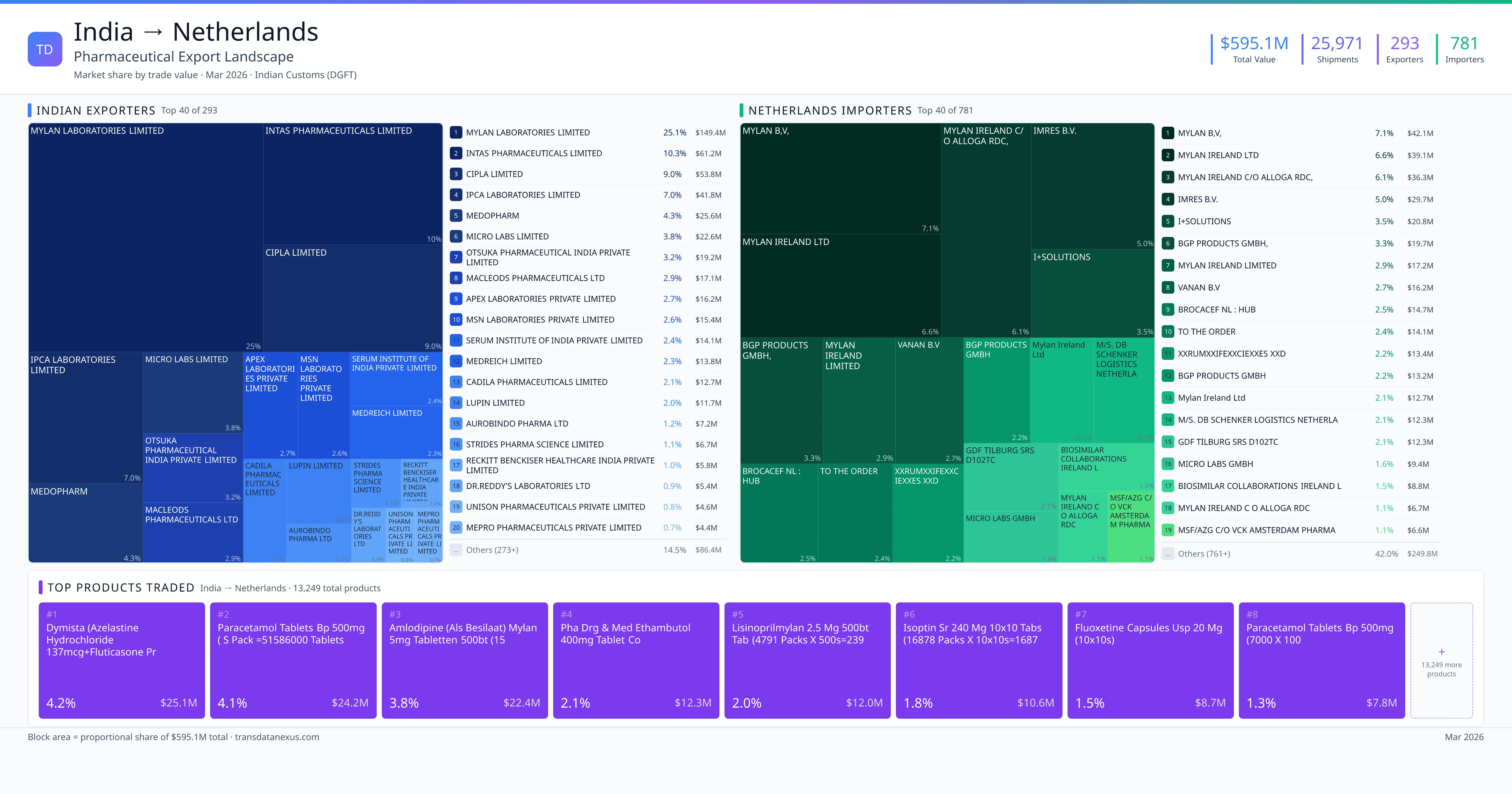
India imported $232.7M worth of pharmaceutical formulations from Netherlands across 1,546 verified shipments, from 120 foreign suppliers to 95 Indian buyers. The top suppliers are INTERVET INTERNATIONAL B.V. ($220.8M) and INTERVET INTERNATIONAL B V ($2.1M). The leading products are Vaccine ($215.4M) and Plasma ($5.9M). Average shipment value: $150.5K.
Top Pharmaceutical Formulations — Netherlands to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Vaccine | $215.4M | 92.6% |
| 2 | Plasma | $5.9M | 2.5% |
| 3 | Serum | $4.7M | 2.0% |
| 4 | Amphotericin | $2.0M | 0.9% |
| 5 | Ustekinumab | $1.5M | 0.6% |
| 6 | Protein | $509.0K | 0.2% |
| 7 | Pomalidomide | $304.3K | 0.1% |
| 8 | Cladribine | $243.9K | 0.1% |
| 9 | Iron | $234.3K | 0.1% |
| 10 | Fludarabine | $230.8K | 0.1% |
| 11 | Posaconazole | $160.3K | 0.1% |
| 12 | Cabozantinib | $134.4K | 0.1% |
| 13 | Trastuzumab | $132.1K | 0.1% |
| 14 | Pembrolizumab | $115.8K | 0.0% |
| 15 | Paclitaxel | $99.9K | 0.0% |
India imports 20+ pharmaceutical formulations from Netherlands with a combined trade value of $232.7M. Key products include Vaccine ($215.4M), Plasma ($5.9M), Serum ($4.7M), Amphotericin ($2.0M), Ustekinumab ($1.5M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,546 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Netherlands to India Trade Routes
These are the top pharmaceutical products exported from India to Netherlands, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Netherlands buyers, regulatory requirements, and logistics for that specific product corridor. Products include Vaccine ($215.4M), Plasma ($5.9M), Serum ($4.7M), Amphotericin ($2.0M), Ustekinumab ($1.5M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Netherlands Suppliers to India
120 Indian pharmaceutical companies export finished formulations to Netherlands. Leading exporters include Intervet International B.v., Intervet International B V, Janssen Pharmaceutica N.v., Merck Sharp & Dohme Asia Pacific Services Pte Ltd. The top exporter accounts for 94.9% of total India–Netherlands pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Netherlands
95 companies in Netherlands import pharmaceutical formulations from India. Top buyers include Intervet India Private Limited, Intervet India Pvt Ltd, Johnson & Johnson Private Limited, Mylan Laboratories Limited. The largest buyer accounts for 95.8% of India–Netherlands pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
FAQ — India to Netherlands Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Netherlands?
India exported pharmaceuticals worth $232.7M to Netherlands across 1,546 verified shipments.
Who are the top Indian pharmaceutical exporters to Netherlands?
1. INTERVET INTERNATIONAL B.V. — $220.8M. 2. INTERVET INTERNATIONAL B V — $2.1M. 3. JANSSEN PHARMACEUTICA N.V. — $1.2M. Total: 120 suppliers.
Which companies in Netherlands import pharmaceuticals from India?
1. INTERVET INDIA PRIVATE LIMITED — $222.8M. 2. INTERVET INDIA PVT LTD — $1.8M. 3. JOHNSON & JOHNSON PRIVATE LIMITED — $1.2M. 95 buyers total.
What pharmaceutical products does India export most to Netherlands?
1. Vaccine ($215.4M, 92.6%); 2. Plasma ($5.9M, 2.5%); 3. Serum ($4.7M, 2.0%); 4. Amphotericin ($2.0M, 0.9%); 5. Ustekinumab ($1.5M, 0.6%)
Which ports handle pharmaceutical shipments from India to Netherlands?
Export: . Import: .
Why does Netherlands import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $232.7M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Netherlands?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Netherlands pharmaceutical trade?
$150.5K per consignment across 1,546 shipments.
How many Indian pharmaceutical companies export to Netherlands?
120 Indian companies. Largest: INTERVET INTERNATIONAL B.V. with $220.8M.
How can I find verified Indian pharmaceutical suppliers for Netherlands?
TransData Nexus covers 120 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Netherlands Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,546 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Netherlands identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,546 verified shipments from 120 Indian exporters to 95 Netherlands buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
120 Exporters
95 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists