How India Exports Miconazole to the World
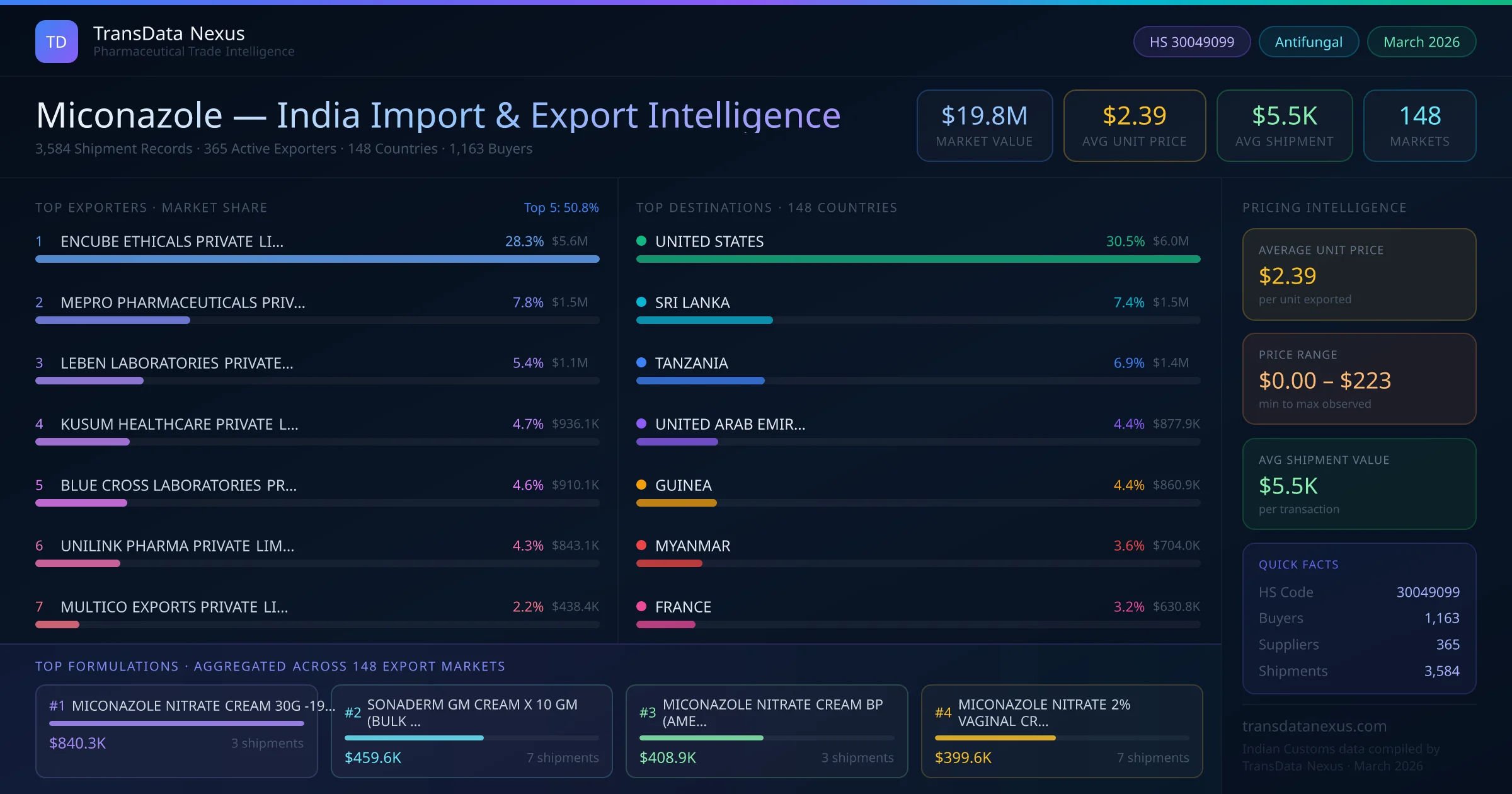
Between 2022 and 2026, India exported $19.8M worth of miconazole across 3,584 verified shipments to 148 countries — covering 76% of world markets in the Antifungals segment. The largest destination is UNITED STATES (30.5%). ENCUBE ETHICALS PRIVATE LIMITED leads with a 28.3% share. All figures are drawn from Indian Customs (DGFT) shipping bill records spanning four years of trade activity.
Top Miconazole Exporters from India
365 active exporters · Ranked by export value
| # | Supplier Name | Export Value (USD) | Market Share |
|---|---|---|---|
| 1 | ENCUBE ETHICALS PRIVATE LIMITED | $5.6M | 28.3% |
| 2 | MEPRO PHARMACEUTICALS PRIVATE LIMITED | $1.5M | 7.8% |
| 3 | LEBEN LABORATORIES PRIVATE LIMITED | $1.1M | 5.4% |
| 4 | KUSUM HEALTHCARE PRIVATE LIMITED | $936.1K | 4.7% |
| 5 | BLUE CROSS LABORATORIES PRIVATE LIMITED | $910.1K | 4.6% |
| 6 | UNILINK PHARMA PRIVATE LIMITED | $843.1K | 4.3% |
| 7 | MULTICO EXPORTS PRIVATE LIMITED | $438.4K | 2.2% |
| 8 | AGIO PHARMACEUTICALS LIMITED | $389.8K | 2.0% |
| 9 | GALENTIC PHARMA INDIA PVT LTD | $372.4K | 1.9% |
| 10 | CIPLA LIMITED | $362.5K | 1.8% |
Based on customs records from 2022 through early 2026, India's miconazole export market is led by ENCUBE ETHICALS PRIVATE LIMITED, which holds a 28.3% share of all miconazole exports — the largest of any single manufacturer over this period. The top 5 suppliers together account for 50.8% of total export value, reflecting a moderately competitive supplier landscape among the 365 active exporters. Each supplier handles an average of 10 shipments, indicating selective, specialised distribution patterns.
Top Countries Importing Miconazole from India
148 destination markets · Ranked by import value
| # | Country | Import Value (USD) | Market Share |
|---|---|---|---|
| 1 | UNITED STATES | $6.0M | 30.5% |
| 2 | SRI LANKA | $1.5M | 7.4% |
| 3 | TANZANIA | $1.4M | 6.9% |
| 4 | UNITED ARAB EMIRATES | $877.9K | 4.4% |
| 5 | GUINEA | $860.9K | 4.4% |
| 6 | MYANMAR | $704.0K | 3.6% |
| 7 | FRANCE | $630.8K | 3.2% |
| 8 | UKRAINE | $519.7K | 2.6% |
| 9 | UZBEKISTAN | $432.3K | 2.2% |
| 10 | GHANA | $413.7K | 2.1% |
UNITED STATES is India's largest miconazole export destination, absorbing 30.5% of total exports worth $6.0M. The top 5 importing countries — UNITED STATES, SRI LANKA, TANZANIA, UNITED ARAB EMIRATES, GUINEA — together account for 53.6% of India's total miconazole export value. The remaining 143 destination countries collectively receive the other 46.4%, indicating a well-diversified global distribution network spanning all major continents.
Who Supplies Miconazole to India?
7 origin countries · Total import value: $377.0K
India imports miconazole from 7 countries with a combined import value of $377.0K. The largest supplier is FRANCE ($200.3K, 4 shipments), followed by UNITED KINGDOM and BELGIUM. All values are from Indian Customs (DGFT) import records.
| # | Origin Country | Import Value (USD) | Share |
|---|---|---|---|
| 1 | FRANCE | $200.3K | 53.1% |
| 2 | UNITED KINGDOM | $132.9K | 35.2% |
| 3 | BELGIUM | $29.2K | 7.7% |
| 4 | BRAZIL | $11.9K | 3.1% |
| 5 | LATVIA | $1.6K | 0.4% |
| 6 | PORTUGAL | $1.2K | 0.3% |
| 7 | GERMANY | $44 | 0.0% |
FRANCE is the largest supplier of miconazole to India, accounting for 53.1% of total import value. The top 5 origin countries — FRANCE, UNITED KINGDOM, BELGIUM, BRAZIL, LATVIA — together supply 99.7% of India's miconazole imports. Click any country to see detailed supplier and buyer data for that import corridor.
Quick Facts
Related Antifungals
All products in Antifungals category • Anti-fungal medications
Related Analysis
Key Players
Regulatory Landscape — Miconazole
Product-specific regulatory status across FDA, EMA, WHO, and CDSCO · As of March 2026
1FDA & US Market Regulatory Status
In the United States, miconazole is approved for various formulations, including creams, powders, and suppositories. The FDA's Orange Book lists multiple Abbreviated New Drug Applications (ANDAs) for miconazole, indicating a competitive generic market. Notably, the FDA has not issued any import alerts specifically targeting miconazole products, suggesting compliance with regulatory standards. Given that 30.5% of India's miconazole exports are destined for the U.S., it is imperative for Indian exporters to adhere strictly to FDA regulations to maintain market access.
2EU & UK Regulatory Framework
In the European Union, miconazole-containing products require marketing authorization from the European Medicines Agency (EMA) or national competent authorities. The EMA has conducted periodic safety update report single assessments (PSUSAs) for miconazole, with the most recent in August 2025, resulting in variations to product information to enhance safety and efficacy. Compliance with EU Good Manufacturing Practice (GMP) standards is mandatory for manufacturers exporting to the EU and UK markets.
3WHO Essential Medicines & Global Standards
Miconazole is included in the World Health Organization's Model List of Essential Medicines, underscoring its importance in treating fungal infections. It is also listed in major pharmacopoeias, including the United States Pharmacopeia (USP), British Pharmacopoeia (BP), European Pharmacopoeia (EP), and Indian Pharmacopoeia (IP), ensuring standardized quality and efficacy across different regions.
4India Regulatory Classification
In India, miconazole is classified under Schedule H of the Drugs and Cosmetics Act, indicating that it is a prescription-only medication. The National Pharmaceutical Pricing Authority (NPPA) has not set a ceiling price for miconazole, allowing market-driven pricing. For exports, the Directorate General of Foreign Trade (DGFT) mandates a No Objection Certificate (NOC) to ensure compliance with national and international regulations.
5Patent & Exclusivity Status
The primary patents for miconazole have expired, leading to a robust generic market with multiple manufacturers producing various formulations. This competitive landscape has resulted in widespread availability and affordability of miconazole products globally.
6Recent Industry Developments
In August 2025, the EMA completed a PSUSA for miconazole, leading to updates in product information to enhance safety profiles. In October 2025, the FDA implemented import certification requirements for certain products, emphasizing the need for compliance among exporters. Additionally, in January 2026, the NPPA conducted a review of antifungal drug prices, including miconazole, to ensure affordability and accessibility in the domestic market.
These developments highlight the dynamic regulatory environment surrounding miconazole, necessitating continuous vigilance and compliance from manufacturers and exporters to maintain market access and ensure patient safety.
Global Price Benchmark — Miconazole
Retail & reference prices across 9 markets vs. India FOB export price of $2.39/unit
| Market | Price (USD/unit) |
|---|---|
| United States | $10–$20 |
| United Kingdom | Approximately $6.50–$13 |
| Germany | Approximately $9–$17 |
| Australia | Approximately $8–$13 |
| Brazil | Approximately $4–$7 |
| Nigeria | Approximately $3–$5 |
| Kenya | Approximately $4–$7 |
| WHO/UNFPA Procurement | $2–$4 |
| India Domestic (NPPA)ORIGIN | Approximately $0.65–$2 |
India Cost Advantage
India's pharmaceutical industry offers a significant cost advantage in the production of Active Pharmaceutical Ingredients (APIs) like Miconazole. This efficiency is driven by well-established manufacturing clusters in Hyderabad, Ahmedabad, and Mumbai, which benefit from economies of scale and a skilled workforce. Additionally, the Pharmaceuticals Export Promotion Council of India (Pharmexcil) provides robust support to manufacturers, facilitating competitive pricing and quality assurance in both domestic and international markets.
Supply Chain Risk Assessment — Miconazole
API sourcing, concentration risk, storage requirements, and current alerts
1API Sourcing & Raw Material Dependency
India's pharmaceutical industry heavily relies on China for Key Starting Materials (KSMs) and Active Pharmaceutical Ingredients (APIs). Approximately 60–70% of these essential components are imported from China, making the supply chain vulnerable to disruptions. This dependency poses a significant risk, as any geopolitical tensions or trade restrictions between India and China could severely impact the production of Miconazole and other pharmaceuticals.
Recent analyses have highlighted that 58% of KSMs used for U.S.-approved APIs are solely sourced from a single country, underscoring the fragility of the supply chain. This concentration increases the risk of supply interruptions due to geopolitical events, natural disasters, or other unforeseen circumstances affecting the supplier country.
2Supplier Concentration & Single-Source Risk
The export market for Miconazole from India is notably concentrated. The top five exporters—ENCUBE ETHICALS PRIVATE LIMITED (28.3% share), MEPRO PHARMACEUTICALS PRIVATE LIMITED (7.8%), LEBEN LABORATORIES PRIVATE LIMITED (5.4%), KUSUM HEALTHCARE PRIVATE LIMITED (4.7%), and BLUE CROSS LABORATORIES PRIVATE LIMITED (4.6%)—collectively account for 50.8% of total exports. This high concentration indicates a significant single-source risk; any operational or financial issues within these companies could disrupt the supply chain.
To mitigate such risks, the Indian government has implemented the Production Linked Incentive (PLI) scheme, aiming to boost domestic manufacturing of APIs and reduce dependency on imports. However, the effectiveness of this initiative in diversifying the supplier base for Miconazole remains to be fully realized.
3Geopolitical & Shipping Disruptions
The Strait of Hormuz, a critical maritime chokepoint through which approximately 20% of the world's daily oil supply transits, has experienced significant disruptions since February 28, 2026, due to escalating military conflicts involving Iran, the United States, and Israel. These tensions have led to attacks on merchant vessels and a sharp decline in maritime traffic, with tanker traffic dropping by approximately 70% and over 150 ships anchoring outside the strait to avoid risks. Such disruptions have caused oil prices to surge, with Brent crude surpassing $100 per barrel on March 8, 2026, for the first time in four years.
These geopolitical tensions and shipping disruptions have had cascading effects on global supply chains, including the pharmaceutical sector. The increased shipping costs and delays can impact the timely export of Miconazole from India to key markets, potentially leading to shortages and increased prices.
4Risk Mitigation Recommendations
- Diversify API and KSM Sources: Encourage the development of alternative suppliers for APIs and KSMs, both domestically and in other countries, to reduce dependency on a single source.
- Strengthen Domestic Manufacturing: Accelerate the implementation and scaling of the PLI scheme to enhance domestic production capabilities for critical pharmaceutical components.
- Enhance Supply Chain Visibility: Implement advanced tracking and monitoring systems to identify potential disruptions early and develop contingency plans.
- Develop Alternative Shipping Routes: Explore and invest in alternative maritime and overland routes to mitigate the impact of disruptions in critical chokepoints like the Strait of Hormuz.
- Engage in International Collaboration: Work with international bodies and trade partners to ensure the security and stability of global shipping lanes and supply chains.
RISK_LEVEL: HIGH
Access Complete Miconazole Trade Intelligence
Shipment-level records, verified supplier contacts, buyer histories, and pricing analytics for all 3,584 transactions across 148 markets.
Frequently Asked Questions — Miconazole Exports from India
Data-backed answers sourced from Indian Customs shipping bill records
Who are the top miconazole exporters from India?
The leading miconazole exporters from India are ENCUBE ETHICALS PRIVATE LIMITED, MEPRO PHARMACEUTICALS PRIVATE LIMITED, LEBEN LABORATORIES PRIVATE LIMITED, and 12 others. ENCUBE ETHICALS PRIVATE LIMITED leads with 28.3% market share ($5.6M). The top 5 suppliers together control 50.8% of total export value.
What is the total export value of miconazole from India?
The total export value of miconazole from India is $19.8M, recorded across 3,584 shipments from 365 active exporters to 148 countries. The average shipment value is $5.5K.
Which countries import miconazole from India?
India exports miconazole to 148 countries. The top importing countries are UNITED STATES (30.5%), SRI LANKA (7.4%), TANZANIA (6.9%), UNITED ARAB EMIRATES (4.4%), GUINEA (4.4%), which together account for 53.6% of total export value.
What is the HS code for miconazole exports from India?
The primary HS code for miconazole exports from India is 30049099. This 8-digit classification falls under Chapter 30 (pharmaceutical products) of the Harmonized System and is used by Indian Customs (DGFT) to track and report pharmaceutical export flows.
What is the average price of miconazole exports from India?
The average unit price for miconazole exports from India is $2.39 per unit, with prices ranging from $0.00 to $222.85 depending on formulation and order volume.
Which ports handle miconazole exports from India?
The primary export ports for miconazole from India are SAHAR AIR (18.2%), SAHAR AIR CARGO ACC (INBOM4) (13.3%), NHAVA SHEVA SEA (INNSA1) (11.4%), JNPT/ NHAVA SHEVA SEA (7.3%). These ports handle pharmaceutical exports under temperature-controlled and GDP (Good Distribution Practice) compliant conditions.
Why is India a leading exporter of miconazole?
India is a leading miconazole exporter due to its large base of 365 manufacturers — many WHO-GMP and US FDA approved — combined with significantly lower production costs, well-developed API supply chains, and strong government support through Pharmexcil. India's miconazole exports reach 148 countries (76% of world markets), making it a dominant global supplier of antifungals compounds.
What certifications do Indian miconazole exporters need?
Indian miconazole exporters typically require WHO-GMP certification for regulated markets, US FDA approval for the United States, and EU GMP certification for European markets. Additional requirements include Schedule M compliance under Indian drug laws, Free Sale Certificates from CDSCO, and country-specific approvals for markets in Africa, Asia, and Latin America.
How many buyers import miconazole from India?
1,163 buyers import miconazole from India across 148 countries. The repeat buyer rate is 48.2%, indicating strong ongoing trade relationships.
What is the market share of the top miconazole exporter from India?
ENCUBE ETHICALS PRIVATE LIMITED is the leading miconazole exporter from India with a market share of 28.3% and export value of $5.6M across 203 shipments. The top 5 suppliers together hold 50.8% of the market.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data on this page is sourced from Indian Customs (DGFT) shipping bill records. Verify regulatory status with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Product Identification: Miconazole shipments identified from HS code matching and DGFT product description fields across 3,584 shipping bill records.
- 2.Supplier/Buyer Matching: 365 Indian exporters and 1,163 global buyers matched using company name normalization.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort supplier or buyer rankings.
- 4.Market Share Calculation: Export value distributed across 148 destination countries. Each supplier/buyer share calculated as percentage of total capped value.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3,584 Verified Shipments
365 exporters to 148 countries
Expert-Reviewed
By pharmaceutical trade specialists