Zydus Lifesciences Limited — Lansoprazole Exporter Profile
Indian Pharmaceutical Exporter · #7 for Lansoprazole · $2.1M export value · DGFT Verified
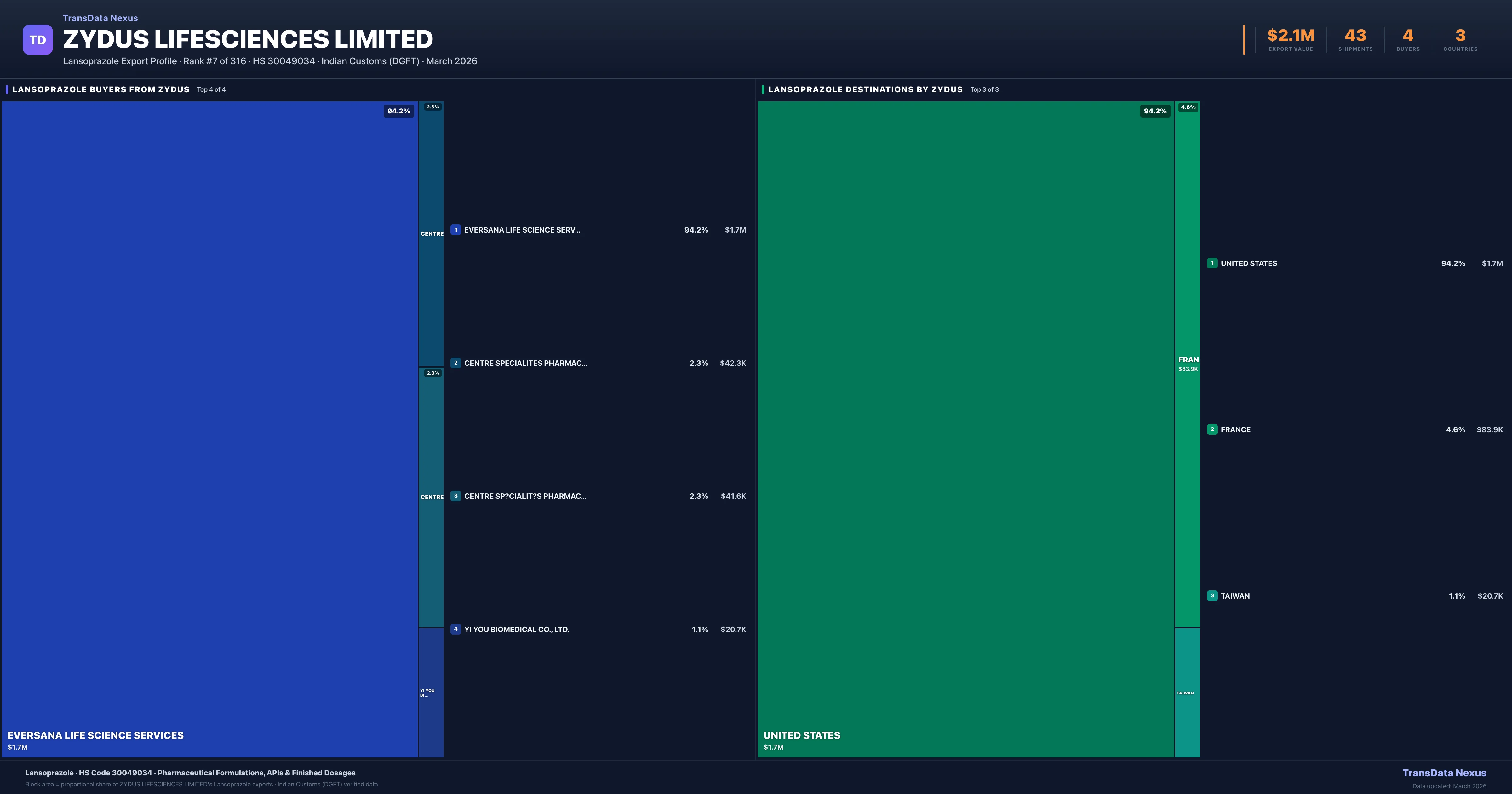
Zydus Lifesciences Limited is the #7 Indian exporter of Lansoprazole with $2.1M in export value and 43 verified shipments. Zydus Lifesciences Limited holds a 0.9% market share in Lansoprazole exports across 3 countries. The company exports 81 pharmaceutical products worth $346.3M across 25 therapeutic categories.
Zydus Lifesciences Limited — Lansoprazole Export Profile: Buyers & Destinations
Where Does Zydus Lifesciences Limited Export Lansoprazole?
| Country | Value | Shipments | Share |
|---|---|---|---|
| UNITED STATES | $1.7M | 34 | 94.2% |
| FRANCE | $83.9K | 7 | 4.6% |
| TAIWAN | $20.7K | 2 | 1.1% |
Zydus Lifesciences Limited exports Lansoprazole to 3 countries. The largest destination is UNITED STATES accounting for 94.2% of Zydus Lifesciences Limited's Lansoprazole shipments, followed by FRANCE (4.6%) and TAIWAN (1.1%). These destinations reflect Zydus Lifesciences Limited's established distribution network and regulatory approvals in key markets.