Who Exports Irbesartan from India — 123 Suppliers Behind a $106.3M Market
India's irbesartan export market is supplied by 123 active exporters who collectively shipped $106.3M across 2,669 shipments. MACLEODS PHARMACEUTICALS LTD leads with a 33.7% market share, followed by MYLAN LABORATORIES LIMITED and CAPLIN POINT LABORATORIES LIMITED. The top 5 suppliers together control 76.1% of total export value, reflecting a concentrated market structure.
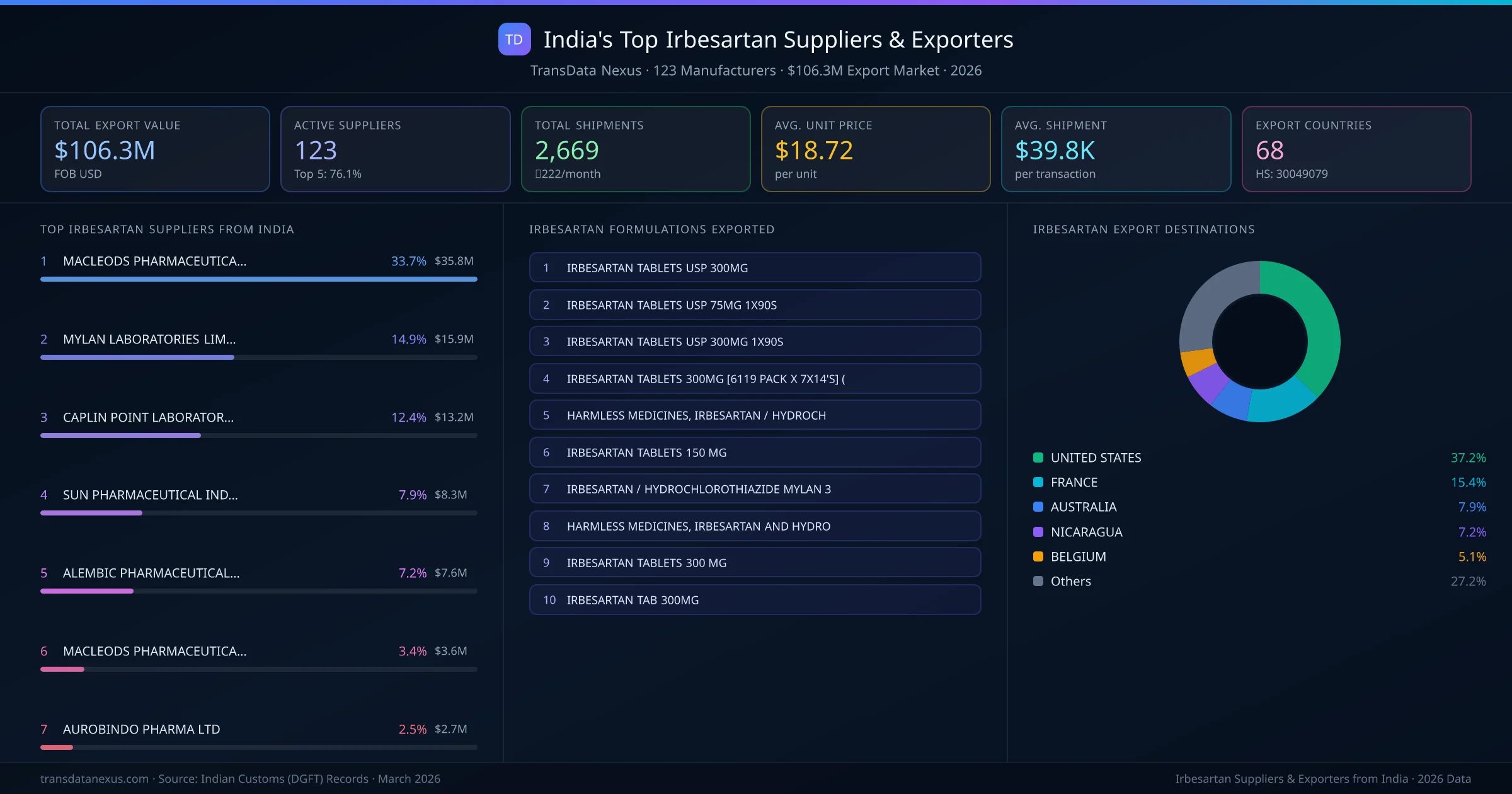
Top Irbesartan Exporters from India — Ranked by Export Value
MACLEODS PHARMACEUTICALS LTD is the leading irbesartan exporter from India, holding a 33.7% share of the $106.3M market across 2,669 shipments from 123 exporters. The top 5 suppliers — MACLEODS PHARMACEUTICALS LTD, MYLAN LABORATORIES LIMITED, CAPLIN POINT LABORATORIES LIMITED, SUN PHARMACEUTICAL INDUSTRIES LIMITED, ALEMBIC PHARMACEUTICALS LIMITED — collectively control 76.1% of total export value, indicating a highly concentrated market. Individual shares are: MACLEODS PHARMACEUTICALS LTD (33.7%), MYLAN LABORATORIES LIMITED (14.9%), CAPLIN POINT LABORATORIES LIMITED (12.4%), SUN PHARMACEUTICAL INDUSTRIES LIMITED (7.9%), ALEMBIC PHARMACEUTICALS LIMITED (7.2%).
Top Irbesartan Exporters from India
Ranked by export value · 123 active suppliers · Indian Customs (DGFT) data
| # | Supplier & Formulations | Value | Ctry. | Share |
|---|---|---|---|---|
| 1 | MACLEODS PHARMACEUTICALS LTD IRBESARTAN TABLETS USP 75MG 1X90SIRBESARTAN TABLETS USP 300MG 1X90SIRBESARTAN TABLETS BP 300MG (103000X10X | $35.8M | 12 | 33.7% |
| 2 | MYLAN LABORATORIES LIMITED HARMLESS MEDICINES, IRBESARTAN / HYDROCHIRBESARTAN / HYDROCHLOROTHIAZIDE MYLAN 3HARMLESS MEDICINES, IRBESARTAN AND HYDRO | $15.9M | 5 | 14.9% |
| 3 | CAPLIN POINT LABORATORIES LIMITED IRBESARTAN TABLETS USP 300MGIRBESARTAN TABLETS BP 300MG (103000X10XIRBESARTAN TABLETS USP 150MG | $13.2M | 9 | 12.4% |
| 4 | SUN PHARMACEUTICAL INDUSTRIES LIMITED IRBESARTAN TABLETS 300MG [6119 PACK X 7X14'S] (IRBESARTAN TABLETS 150 MGIRBESARTAN TABLETS 300 MG | $8.3M | 13 | 7.9% |
| 5 | ALEMBIC PHARMACEUTICALS LIMITED IRBESARTAN TABLETS USP 75MG 1X90SIRBESARTAN TABLETS USP 300MG 1X90SIRBESARTAN TABLETS 150 MG | $7.6M | 2 | 7.2% |
| 6 | MACLEODS PHARMACEUTICALS LIMITED IRBESARTAN TABLETS USP 75MG 1X90SIRBESARTAN TABLETS USP 300MG 1X90SIRBESARTAN TABLETS BP 300MG (103000X10X | $3.6M | 7 | 3.4% |
| 7 | AUROBINDO PHARMA LTD HARMLESS MEDICINES, IRBESARTAN / HYDROCHIRBESARTAN / HYDROCHLOROTHIAZIDE MYLAN 3IRBESARTAN TABLETS 150MG (IRBESARTAN ARR | $2.7M | 4 | 2.5% |
| 8 | ZYDUS LIFESCIENCES LIMITED HARMLESS MEDICINES, IRBESARTAN / HYDROCHIRBESARTAN / HYDROCHLOROTHIAZIDE MYLAN 3IRBESARTAN TABLETS 150MG (IRBESARTAN ARR | $2.4M | 6 | 2.3% |
| 9 | INTAS PHARMACEUTICALS LIMITED PHARM DRUG&MED VIRBEZ 300PHARM DRUG&MED:VIRBEZ 300PHARM DRUG&MED:VIRBEZ 300NOS | $1.8M | 5 | 1.7% |
| 10 | MEDICO REMEDIES LIMITED IRBESARTAN TABLETS USP 300MGIRBESARTAN TABLETS BP 300MG (103000X10XIRBESARTAN TABLETS BP 300MG (85000X10X1 | $1.4M | 2 | 1.3% |
| 11 | JUBILANT GENERICS LIMITED IRBESARTAN TABLETS USP 75MG 1X90SIRBESARTAN TABLETS USP 300MG 1X90SIRBESARTAN TABLETS 300MG [6119 PACK X 7X14'S] ( | $1.3M | 7 | 1.2% |
| 12 | MICRO LABS LIMITED IRBESARTAN TABLETS 300MG [6119 PACK X 7X14'S] (IRBESARTAN TABLETS 150 MGIRBESARTAN TABLETS 300 MG | $1.3M | 6 | 1.2% |
| 13 | AUROBINDO PHARMA LIMITED HARMLESS MEDICINES, IRBESARTAN / HYDROCHIRBESARTAN / HYDROCHLOROTHIAZIDE MYLAN 3IRBESARTAN TABLETS 150MG (IRBESARTAN ARR | $1.2M | 3 | 1.1% |
| 14 | MEPRO PHARMACEUTICALS PRIVATE LIMITED | $1.0M | 1 | 1.0% |
| 15 | HETERO LABS LIMITED IRBESARTAN TABLETS USP 300MGIRBESARTAN TABLETS USP 75MG 1X90SIRBESARTAN TABLETS USP 300MG 1X90S | $1.0M | 8 | 1.0% |
Related Analysis
Supplier Certification & Compliance Matrix
FDA, WHO-GMP, and EU GMP status for top Irbesartan exporters
| Supplier | US FDA | WHO-GMP | EU GMP | ANDAs | Notes |
|---|---|---|---|---|---|
| Alembic Pharmaceuticals Limited | Approved | Yes | Yes | 15 (Jan–Sept 2023) | Received multiple FDA approvals between 2024 and 2025. |
| Zydus Lifesciences Limited | Approved | Yes | Yes | 59 (Jan–Sept 2023) | Ranked 100th in Fortune India 500 list in 2020. |
| Intas Pharmaceuticals Limited | Approved | Yes | Yes | Not specified | FDA cited data falsification at Sanand plant in Dec 2022. |
| Micro Labs Limited | Approved | Yes | Yes | 11 (Jan–Sept 2023) | Operates 14 GMP-compliant plants in India. |
| Macleods Pharmaceuticals Limited | Approved | Yes | Yes | 10 (Jan–Sept 2023) | Approved under India's PLI Scheme as of Sept 2022. |
| Aurobindo Pharma Limited | Approved | Yes | Yes | 41 (Jan–Sept 2023) | Ranked 2nd in India's PLI Scheme as of Sept 2022. |
| Sun Pharmaceutical Industries Limited | Approved | Yes | Yes | 10 (Jan–Sept 2023) | Ranked 1st in India's PLI Scheme as of Sept 2022. |
| Mylan Laboratories Limited | Approved | Yes | Yes | Not specified | Ranked 5th in India's PLI Scheme as of Sept 2022. |
| Hetero Labs Limited | Approved | Yes | Yes | 10 (Jan–Sept 2023) | Received 10 ANDA approvals from FDA in 2023. |
TransData Nexus reviewed the regulatory standing of 9 leading Irbesartan exporters from India. 9 hold US FDA facility approvals, 9 maintain WHO-GMP certification, and 9 are EU GMP compliant. For regulated markets (US, EU, Australia), prioritise suppliers with active FDA or EU GMP approvals. For semi-regulated markets (Africa, ASEAN, Latin America), WHO-GMP certification is the minimum recommended standard.
Certification status compiled from publicly available regulatory databases including FDA Orange Book, WHO Prequalification database, and EMA GMP registry. Buyers should independently verify compliance status with the relevant regulatory authority before placing orders.
TransData Nexus Research · Mar 2026
Manufacturing Hub Analysis — Vendor Proximity
India's pharmaceutical clusters relevant to Irbesartan sourcing
1Hyderabad — Bulk Drug Capital
Hyderabad, often referred to as "India's pharmaceutical capital," has been a pivotal center for bulk drug manufacturing since the establishment of Indian Drugs and Pharmaceuticals Limited (IDPL) in 1961. The city contributes significantly to India's bulk drug production, manufacturing one-third of the nation's bulk drugs and 16% of biotechnology products as of 2010. This robust infrastructure supports the production of active pharmaceutical ingredients (APIs) essential for medications like Irbesartan.
2Ahmedabad-Vadodara — Formulations Hub
The Ahmedabad-Vadodara corridor in Gujarat is renowned for its pharmaceutical formulations. Gujarat accounts for 33% of India's drug manufacturing and 28% of drug exports, with Ahmedabad and Vadodara being central to this output. The region hosts major companies such as Alembic Pharmaceuticals, which has been operational since 1907, and Sun Pharmaceutical Industries, with a significant plant in Halol, Vadodara. These companies are instrumental in producing finished dosage forms, including Irbesartan tablets, catering to both domestic and international markets.
3Mumbai-Thane-Raigad — Export Gateway
The Mumbai-Thane-Raigad belt serves as a crucial export gateway for pharmaceuticals. Mumbai, being India's financial capital, offers extensive port facilities and logistical advantages, facilitating the export of pharmaceutical products. Companies in this region, such as Sun Pharmaceutical Industries, leverage these advantages to distribute products like Irbesartan to global markets efficiently.
4Baddi-Nalagarh — Tax Incentive Zone
The Baddi-Nalagarh region in Himachal Pradesh has emerged as a significant pharmaceutical hub, primarily due to favorable tax incentives. This zone attracts numerous pharmaceutical companies focusing on cost-effective manufacturing of various drugs, including antihypertensive medications like Irbesartan.
5Sourcing Recommendations
- Diversify Supplier Base: While major suppliers like MACLEODS PHARMACEUTICALS LTD and MYLAN LABORATORIES LIMITED dominate the market, consider engaging with emerging manufacturers in regions like Baddi-Nalagarh to mitigate supply chain risks.
- Leverage Export Hubs: Utilize the logistical advantages of the Mumbai-Thane-Raigad region to streamline the export process and reduce lead times for international shipments.
- Monitor Regulatory Compliance: Ensure that all suppliers adhere to international quality standards and regulatory requirements, particularly when sourcing from tax incentive zones where compliance levels may vary.
- Assess Cost-Benefit Ratios: Evaluate the cost-effectiveness of sourcing from different clusters, balancing factors such as production costs, tax benefits, and logistical expenses to optimize the supply chain for Irbesartan.
Recent M&A, Collaborations & Capacity Expansions
Industry developments among top Irbesartan exporters from India
Caplin Point Laboratories Limited — Caplin acquires 10 approved ANDAs for injectables and ophthalmic products
Caplin Point Laboratories Limited, through its subsidiaries Caplin Steriles Limited and Caplin One Labs Limited, acquired 10 approved Abbreviated New Drug Applications (ANDAs) for injectable and ophthalmic products from a leading multinational generic product manufacturer. The addressable market for these products was $473.2 million for the 12-month period ending August 2025. - IMPACT: This acquisition is expected to enhance Caplin's product portfolio, potentially increasing its market share in the U.S. and other key markets, which may indirectly influence its Irbesartan export capabilities.
Impact: This acquisition is expected to enhance Caplin's product portfolio, potentially increasing its market share in the U.S. and other key markets, which may indirectly influence its Irbesartan export capabilities.
Alembic Pharmaceuticals Limited — Alembic acquires Utility Therapeutics Ltd for $12 million
Alembic Pharmaceuticals' U.S. subsidiary acquired Utility Therapeutics Ltd for $12 million, adding the FDA-approved urinary tract infection drug Pivya (pivmecillinam 185 mg) to its portfolio. - IMPACT: This acquisition expands Alembic's product offerings in the U.S. market, potentially strengthening its overall export capabilities, including those related to Irbesartan.
Impact: This acquisition expands Alembic's product offerings in the U.S. market, potentially strengthening its overall export capabilities, including those related to Irbesartan.
Sun Pharmaceutical Industries Limited — Sun Pharma acquires Checkpoint Therapeutics for $355 million
Sun Pharma acquired Checkpoint Therapeutics, a company specializing in immunotherapy and targeted oncology treatments, for $355 million. - IMPACT: This acquisition enhances Sun Pharma's oncology portfolio, potentially increasing its global market presence and indirectly affecting its Irbesartan export strategies.
Impact: This acquisition enhances Sun Pharma's oncology portfolio, potentially increasing its global market presence and indirectly affecting its Irbesartan export strategies.
Common Questions — Irbesartan Suppliers from India
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which irbesartan supplier from India is the most reliable for bulk orders?
Based on shipment frequency and export consistency, MACLEODS PHARMACEUTICALS LTD leads with 178 recorded shipments worth $35.8M. MYLAN LABORATORIES LIMITED (217 shipments) and CAPLIN POINT LABORATORIES LIMITED (106 shipments) are also established high-volume exporters.
Q How many irbesartan manufacturers are there in India?
India has 123 active irbesartan exporters with a combined export market of $106.3M across 2,669 shipments to 68 countries. The top 5 suppliers hold 76.1% of total export value.
Q What certifications should I verify?
Verify: WHO-GMP certification (most markets), US FDA approval (United States), EU GMP certificate (EU/EEA), and Free Sale Certificate from CDSCO. Always request a Certificate of Analysis (CoA) and Certificate of Origin (CoO).
Q What is the typical price range for irbesartan from India?
Average FOB unit price: $18.72 per unit, ranging from $0.00 to $4772.17. Average shipment value: $39.8K.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Ranking: 123 verified Indian exporters of Irbesartan ranked by capped export value from DGFT shipping bill records.
- 2.Export Value Analysis: Total export value aggregated from 2,669 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Market Concentration: Supplier market share and geographic reach analyzed across 68 destination countries.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2,669 Verified Shipments
123 exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists