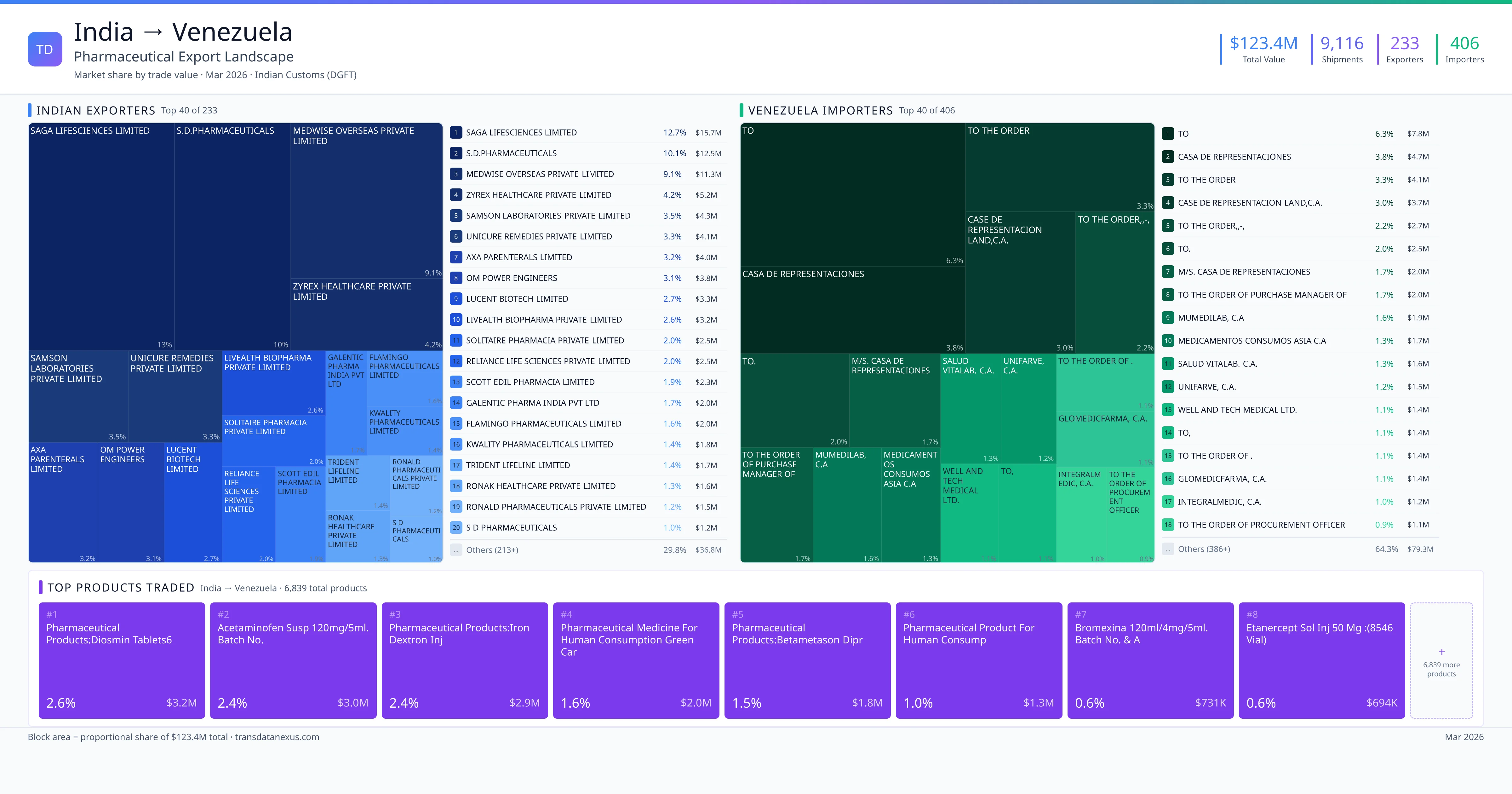
Indian pharmaceutical companies seeking to export to Venezuela must navigate a comprehensive registration process to ensure compliance with Venezuelan regulations. The National Institute of Drug and Food Control (Instituto Nacional de Control de Alimentos y Medicamentos – INCANAM), under the Ministry of Popular Power for Health (Ministerio del Poder Popular para la Salud – MPPS), requires prior approval for pharmaceutical imports. The registration dossier must include a health certificate issued by the competent authority in the country of origin, legalized by a Venezuelan consulate, and a certificate stating that the product is freely sold and consumed in the country of origin. Additionally, the importer must provide the firm's trade register, a copy of the pharmacist’s degree, evidence of good standing with the Association of Pharmacists, power of attorney from the parent company to the agency, health certificate of free sale in the country of origin, qualitative formula, and analytical information on the product.