India to Niger Pharmaceutical Export
Bilateral Trade Intelligence · $30.9M Total Trade · 62 Exporters · 83 Buyers · DGFT Verified · Updated March 2026
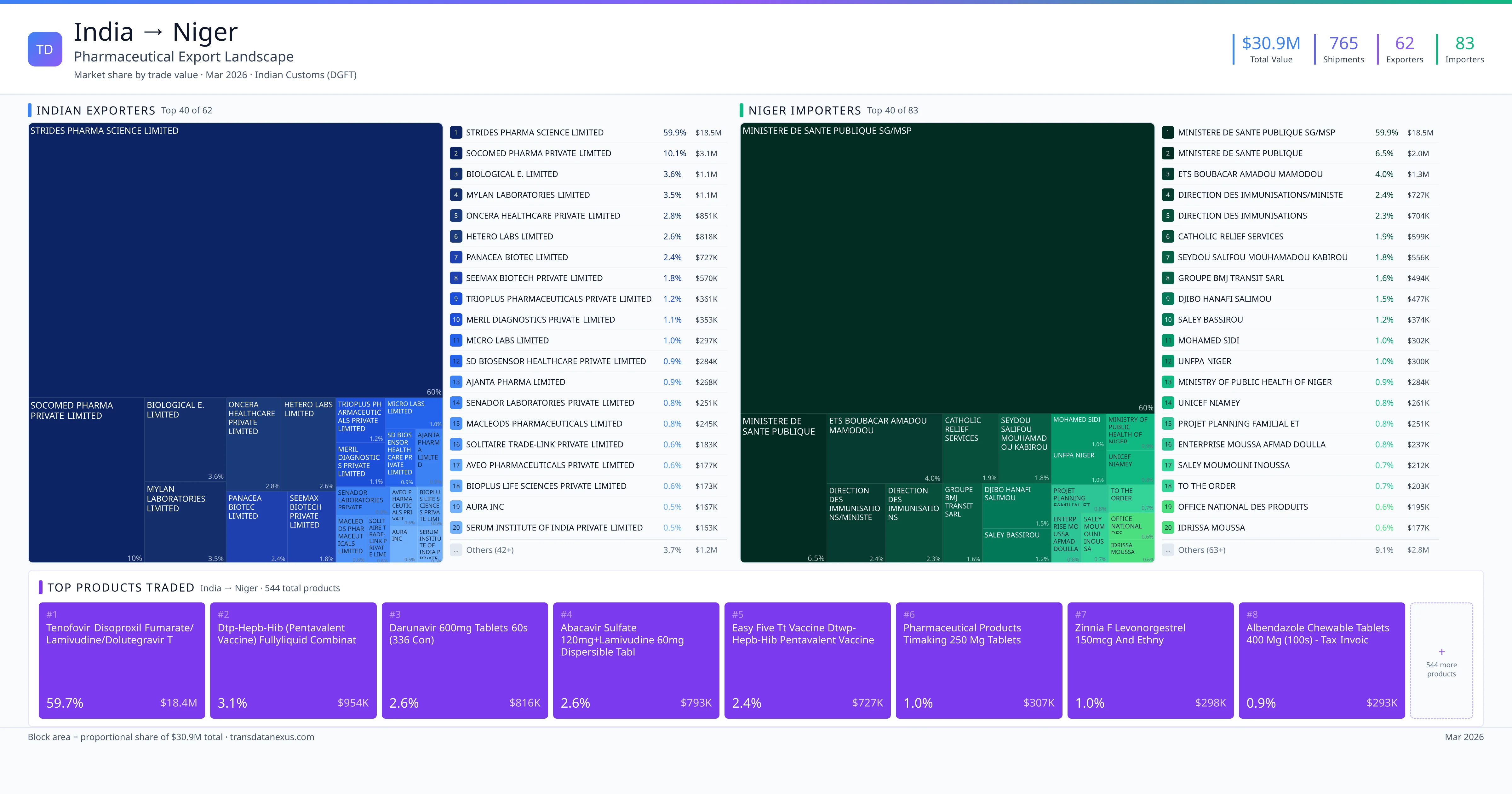
India exported $30.9M worth of pharmaceutical formulations to Niger across 765 verified shipments, sourced from 62 Indian exporters supplying 83 Niger buyers. The top exporters are STRIDES PHARMA SCIENCE LIMITED ($18.5M) and SOCOMED PHARMA PRIVATE LIMITED ($3.1M). The leading products are Tenofovir ($18.4M) and Darunavir ($816K). Average shipment value: $40.4K.
Top Pharmaceutical Formulations — India to Niger
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Tenofovir Disoproxil Fumarate/lamivudine/dolutegravir Tablets , 300mg/300mg/50m… | $18.4M | 59.7% |
| 2 | Darunavir 600mg Tablets 60s (336 Con) | $816.5K | 2.6% |
| 3 | Abacavir Sulfate 120mg+lamivudine 60mg Dispersible Tablets (60's)- No Carton (2… | $792.9K | 2.6% |
| 4 | Easy Five Tt Vaccine Dtwp-hepb-hib Pentavalent Vaccine with Vaccine Vial Monitor | $726.9K | 2.4% |
| 5 | Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine 10 Dose Vial… | $703.7K | 2.3% |
| 6 | Pharmaceutical Products Timaking 250 MG Tablets | $306.7K | 1.0% |
| 7 | Zinnia F Levonorgestrel 150mcg and Ethny | $298.3K | 1.0% |
| 8 | Albendazole Chewable Tablets 400 MG (100s) - Tax Invoice No: 701209669 Dt: 31.1… | $293.5K | 0.9% |
| 9 | Standard Q Hiv Syphilis Combo 25t | $283.7K | 0.9% |
| 10 | Pharmaceutical Products Tafrodol 120mg Capsules | $271.6K | 0.9% |
| 11 | Sutura Ll/ee+ff, 84's Niger (144000 X84x1) | $251.4K | 0.8% |
| 12 | Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine 10dose (5ml)… | $250.6K | 0.8% |
| 13 | Other: New Socomol (paracetamol 325 MG + | $238.2K | 0.8% |
| 14 | Meriscreen Malaria Pf Hrp-ii Ag - 25t Rpwmph-03nos | $199.5K | 0.6% |
| 15 | Pregnancy and Lactation Vitamin and Mineral Supplement. (100's Jar Pack) | $173.0K | 0.6% |
India exports 20+ pharmaceutical formulations to Niger with a combined trade value of $30.9M. Key products include Tenofovir ($18.4M), Darunavir ($816K), Albendazole ($293K), Diclofenac ($170K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 765 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Niger Trade Routes
These are the top pharmaceutical products exported from India to Niger, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Niger buyers, regulatory requirements, and logistics for that specific product corridor. Products include Tenofovir ($18.4M), Darunavir ($816K), Albendazole ($293K), Diclofenac ($170K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Niger
62 Indian pharmaceutical companies export finished formulations to Niger. Leading exporters include Strides Pharma Science Limited, Socomed Pharma Private Limited, Biological E. Limited, Mylan Laboratories Limited. The top exporter accounts for 59.9% of total India–Niger pharma exports. Source: Indian Customs (DGFT).
Top Niger Buyers from India
83 companies in Niger import pharmaceutical formulations from India. Top buyers include Ministere De Sante Publique Sg/msp, Ministere De Sante Publique, Ets Boubacar Amadou Mamodou, Direction Des Immunisations/ministe. The largest buyer accounts for 59.9% of India–Niger pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Niger Pharmaceutical Shipments
Indian Export Ports
Niger Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Niger Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Niger pharmaceutical trade corridor has experienced significant growth over the past decade. In 2024, India's pharmaceutical exports to Niger reached $23.15 million, marking a substantial increase from previous years. This upward trajectory underscores the strengthening trade relations between the two nations.
Key milestones include the establishment of the Mahatma Gandhi International Convention Centre in Niger in September 2018, funded by a $35.484 million grant from India. This initiative not only enhanced bilateral ties but also facilitated increased trade and cooperation in various sectors, including pharmaceuticals.
The growth trajectory is further evidenced by the rise in India's pharmaceutical exports to Niger, from $7.11 million in 2018 to $23.15 million in 2024.
2India's Market Position
India holds a dominant position in Niger's pharmaceutical import market. In 2024, India's pharmaceutical exports to Niger amounted to $23.15 million, accounting for a significant portion of Niger's total pharmaceutical imports. This substantial share highlights India's strategic importance as a key supplier of pharmaceutical products to Niger.
The dominance is further underscored by the fact that India is the largest provider of generic medicines globally, contributing to around 60% of the global vaccine supply. This global leadership enhances India's competitive edge in supplying affordable and high-quality pharmaceutical products to markets like Niger.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Niger pharmaceutical trade. The Indian government's Production Linked Incentive (PLI) scheme, launched to promote domestic manufacturing of critical pharmaceutical ingredients, has bolstered India's capacity to meet international demand.
Additionally, the Mahatma Gandhi International Convention Centre, funded by India in 2018, has enhanced bilateral relations, facilitating increased trade and cooperation in various sectors, including pharmaceuticals.
Niger Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export products to Niger must adhere to the registration requirements set by Niger's National Drug Regulatory Authority, which operates under WHO-GMP standards. The registration process involves submitting a comprehensive dossier, typically in the Common Technical Document (CTD) format, which includes detailed information on the product's quality, safety, and efficacy.
The timeline for product registration can vary, but companies should anticipate a process that may take several months, depending on the complexity of the product and the completeness of the submitted documentation. Ensuring compliance with WHO-GMP standards is crucial, as it facilitates smoother approval processes and aligns with international best practices.
2GMP & Facility Requirements
Niger requires that pharmaceutical products imported into the country be manufactured in facilities adhering to Good Manufacturing Practice (GMP) standards recognized by the World Health Organization (WHO). Indian manufacturers must ensure that their production sites comply with these standards to meet Niger's regulatory requirements.
The inspection process involves evaluations by Niger's regulatory authorities to verify compliance with GMP standards. While there is no explicit mention of mutual recognition agreements between India and Niger, adherence to WHO-GMP standards is widely accepted and facilitates smoother market access.
3Import Documentation
To import pharmaceutical products into Niger, Indian exporters must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): Certifies that the product is authorized for sale in the exporting country.
- Certificate of Analysis (CoA): Provides detailed information on the product's composition and quality.
- GMP Certificate: Confirms that the manufacturing facility complies with GMP standards.
These documents are essential for customs clearance and regulatory approval in Niger. Understanding and adhering to Niger's import procedures is vital for ensuring timely and efficient market entry.
Product Categories & Therapeutic Trends — India to Niger
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Niger are predominantly composed of antiretroviral (ARV) medications, reflecting Niger's healthcare needs. The top product exported is a combination tablet containing Tenofovir Disoproxil Fumarate, Lamivudine, and Dolutegravir, totaling $18.4 million, which constitutes 59.7% of the total export value.
This dominance aligns with Niger's efforts to combat HIV/AIDS, indicating a strategic focus on providing essential ARV therapies to address the disease burden.
2Emerging Opportunities
The global pharmaceutical industry is witnessing patent expirations, leading to increased opportunities for generic drugs. Indian exporters can capitalize on this trend by introducing biosimilars and other generic formulations to meet the growing demand in Niger. Additionally, expanding into new therapeutic areas such as oncology and diabetes care presents avenues for growth, given the rising prevalence of these conditions in the region.
3Demand Drivers
Niger's disease burden, particularly concerning HIV/AIDS, drives the demand for antiretroviral therapies. The country's aging population and increasing healthcare spending further contribute to the growing need for pharmaceutical imports. These factors create a conducive environment for Indian pharmaceutical companies to expand their presence in the Nigerien market.
Trade Policy & Tariff Intelligence — India and Niger
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Niger's tariff structure for pharmaceutical imports includes Most Favored Nation (MFN) tariff rates, preferential rates, and duty exemptions. Specific rates can vary, and it is essential for Indian exporters to consult Niger's customs regulations or engage with local trade authorities to obtain accurate and up-to-date information.
2Trade Agreements
While there is no specific mention of a Free Trade Agreement (FTA) between India and Niger, the bilateral trade relationship is supported by grants and lines of credit, such as the $35.484 million grant for the Mahatma Gandhi International Convention Centre in 2018. These initiatives enhance trade cooperation and may influence pharmaceutical trade dynamics.
3IP & Patent Landscape
Niger's intellectual property (IP) and patent landscape, including provisions for data exclusivity and compulsory licensing, can impact the entry of Indian generics. While specific details are not provided, Indian exporters should conduct thorough due diligence to understand Niger's IP regulations and ensure compliance with local laws.
Supply Chain & Logistics — India to Niger Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Niger typically transit through major ports such as Nhava Sheva (JNPT) and Kattupalli Port in India. The primary import ports in Niger include Niamey and Tema. Transit times can vary based on the shipping route and logistics arrangements, but exporters should plan for potential delays and ensure timely delivery to meet market demands.
2Port Infrastructure
Key export ports in India include Nhava Sheva (JNPT) and Kattupalli Port, which handle a significant volume of pharmaceutical exports. In Niger, Niamey serves as a major import port, facilitating the entry of pharmaceutical products into the country. Understanding the capabilities and operational status of these ports is crucial for effective logistics planning.
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is paramount. Implementing temperature-controlled logistics solutions, adhering to Good Distribution Practice (GDP) compliance, and ensuring appropriate packaging standards are essential to preserve product quality and meet regulatory requirements.
Market Opportunity Assessment — Niger for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Niger's pharmaceutical market is experiencing growth, driven by increasing healthcare needs and a rising disease burden. The demand for generic medicines, particularly antiretroviral therapies, presents significant opportunities for Indian exporters. Understanding the market size and growth projections is vital for strategic planning and investment decisions.
2Healthcare System
Niger's government healthcare programs, insurance coverage, and drug procurement systems play a role in shaping pharmaceutical demand. Engaging with local stakeholders and understanding the healthcare infrastructure can provide insights into distribution channels and potential partnerships.
3Opportunity for Indian Exporters
Indian pharmaceutical companies have opportunities to expand their presence in Niger by supplying essential medicines, including antiretrovirals, and exploring new therapeutic areas. Collaborations with local entities and participation in government healthcare initiatives can further enhance market penetration.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Niger
Competing origins, India's edge, challenges and threats
1Competing Origins
Niger imports pharmaceutical products from various countries, including China, France, and Kenya. In 2022, China was the largest supplier, accounting for $1.29 million of the $1.57 million total imports of pharmaceutical substances. This competition underscores the need for Indian exporters to differentiate their offerings.
2India's Competitive Edge
India's advantages include cost-effective manufacturing, adherence to WHO
FAQ — India to Niger Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Niger?
India exported pharmaceuticals worth $30.9M to Niger across 765 verified shipments.
Who are the top Indian pharmaceutical exporters to Niger?
1. STRIDES PHARMA SCIENCE LIMITED — $18.5M. 2. SOCOMED PHARMA PRIVATE LIMITED — $3.1M. 3. BIOLOGICAL E. LIMITED — $1.1M. Total: 62 suppliers.
Which companies in Niger import pharmaceuticals from India?
1. MINISTERE DE SANTE PUBLIQUE SG/MSP — $18.5M. 2. MINISTERE DE SANTE PUBLIQUE — $2.0M. 3. ETS BOUBACAR AMADOU MAMODOU — $1.3M. 83 buyers total.
What pharmaceutical products does India export most to Niger?
1. Tenofovir Disoproxil Fumarate/lamivudine/dolutegravir Tablets , 300mg/300mg/50m… ($18.4M, 59.7%); 2. Darunavir 600mg Tablets 60s (336 Con) ($816.5K, 2.6%); 3. Abacavir Sulfate 120mg+lamivudine 60mg Dispersible Tablets (60's)- No Carton (2… ($792.9K, 2.6%); 4. Easy Five Tt Vaccine Dtwp-hepb-hib Pentavalent Vaccine with Vaccine Vial Monitor ($726.9K, 2.4%); 5. Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine 10 Dose Vial… ($703.7K, 2.3%)
Which ports handle pharmaceutical shipments from India to Niger?
Export: KATTUPALLI PORT SEA, NHAVA SHEVA SEA (INNSA1), JNPT/ NHAVA SHEVA SEA, DELHI AIR, COCHIN SEA. Import: NIAMEY, Tema, Niamey, Cotonou, TEMA.
Why does Niger import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $30.9M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Niger?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Niger pharmaceutical trade?
$40.4K per consignment across 765 shipments.
How many Indian pharmaceutical companies export to Niger?
62 Indian companies. Largest: STRIDES PHARMA SCIENCE LIMITED with $18.5M.
How can I find verified Indian pharmaceutical suppliers for Niger?
TransData Nexus covers 62 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Niger Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 765 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Niger identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 765 verified shipments from 62 Indian exporters to 83 Niger buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
62 Exporters
83 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists