India to New Zealand Pharmaceutical Export
Bilateral Trade Intelligence · $117.1M Total Trade · 161 Exporters · 263 Buyers · DGFT Verified · Updated March 2026
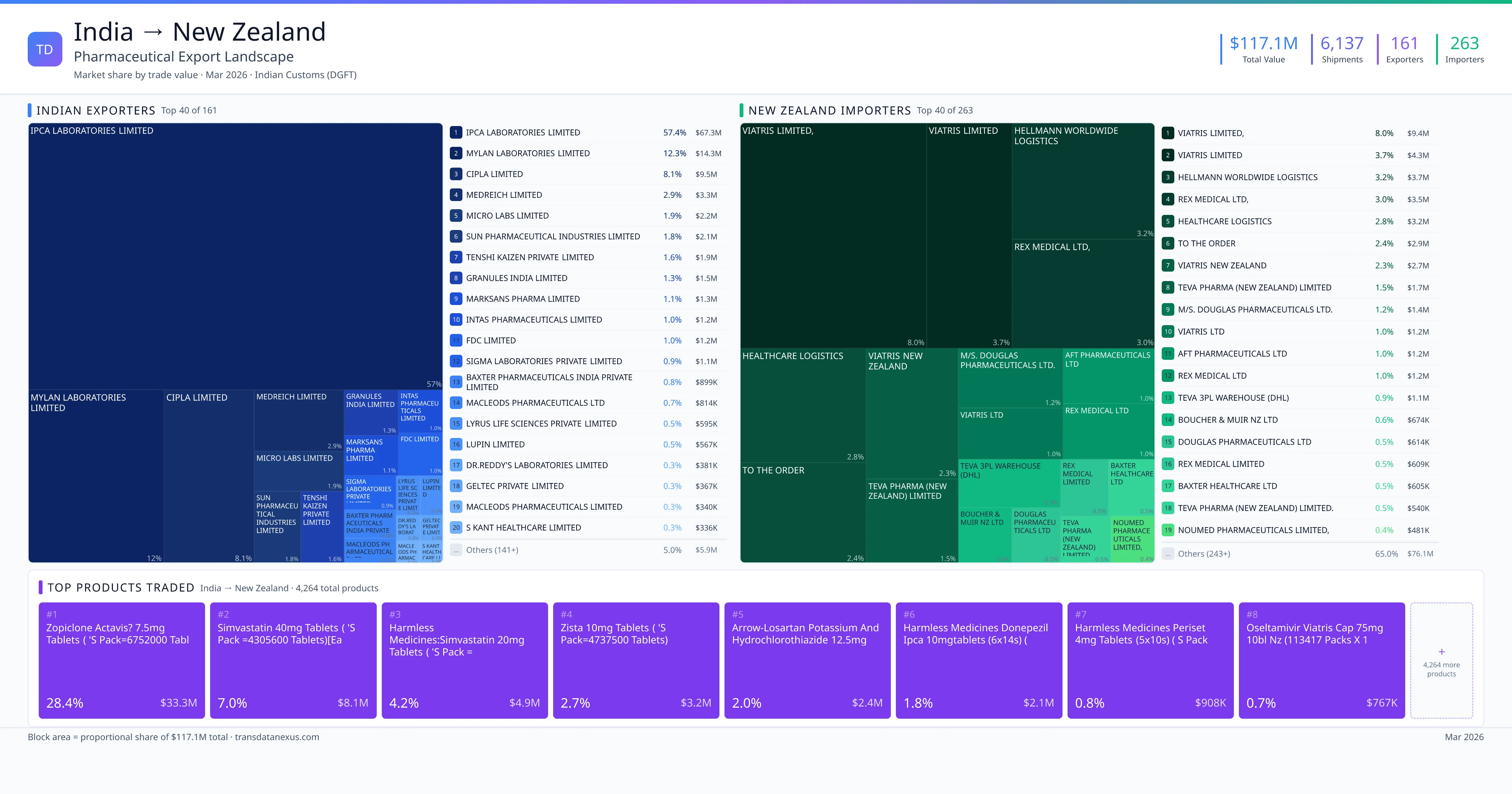
India exported $117.1M worth of pharmaceutical formulations to New Zealand across 6,137 verified shipments, sourced from 161 Indian exporters supplying 263 New Zealand buyers. The top exporters are IPCA LABORATORIES LIMITED ($67.3M) and MYLAN LABORATORIES LIMITED ($14.3M). The leading products are Simvastatin ($8.1M) and Losartan ($2.4M). Average shipment value: $19.1K.
Top Pharmaceutical Formulations — India to New Zealand
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Zopiclone Actavis? 7.5mg Tablets ( 's Pack=6752000 Tablets) | $33.3M | 28.4% |
| 2 | Simvastatin 40mg Tablets ( 's Pack =4305600 Tablets)[each Tablets Cont:simvasta… | $8.1M | 7.0% |
| 3 | Harmless Medicines:simvastatin 20mg Tablets ( 's Pack =4357440 Tablets)[each Ta | $4.9M | 4.2% |
| 4 | Zista 10mg Tablets ( 's Pack=4737500 Tablets) | $3.2M | 2.7% |
| 5 | Arrow-losartan Potassium and Hydrochlorothiazide 12.5mg Tablets(losartan Potass… | $2.4M | 2.0% |
| 6 | Harmless Medicines Donepezil Ipca 10mgtablets (6x14s) ( S Pack) Unit | $2.1M | 1.8% |
| 7 | Harmless Medicines Periset 4mg Tablets (5x10s) ( S Pack) Un | $908.0K | 0.8% |
| 8 | Oseltamivir Viatris Capsules 75mg 10bl Nz (113417 Packs X 1 X 10s= 1134170 ) | $767.2K | 0.7% |
| 9 | Harmless Medicines :- Oseltamivir Viatris Capsules 75mg 10bl Nz (112,292.000 Pa… | $757.0K | 0.6% |
| 10 | Harmless Medicines:pacimol 500mg Tablets | $660.5K | 0.6% |
| 11 | Losartan Actavis?12.5 MG Tablets(losartan Potassium Tablets 12.5mg( 's Pack=745… | $635.7K | 0.5% |
| 12 | Harmless Medicines Donepezil Ipca 5mgtablets (6x14s) ( S Pack) Unit Rat | $594.4K | 0.5% |
| 13 | Harmless Medicines, Lamivudine Viatris 150 MG 60bot (2666 Packs) | $560.1K | 0.5% |
| 14 | Miro-amoxicillin Capsules 500mg (bp) (500sx8733) - Tax Invoice No: 711205339 Dt… | $539.4K | 0.5% |
| 15 | Lorstat 10mg 500bot Tablets (96601 Packs X 500's=48300500 ) | $474.0K | 0.4% |
India exports 20+ pharmaceutical formulations to New Zealand with a combined trade value of $117.1M. Key products include Simvastatin ($8.1M), Losartan ($2.4M), Oseltamivir ($767K), Amoxicillin ($539K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 6,137 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to New Zealand Trade Routes
These are the top pharmaceutical products exported from India to New Zealand, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, New Zealand buyers, regulatory requirements, and logistics for that specific product corridor. Products include Simvastatin ($8.1M), Losartan ($2.4M), Oseltamivir ($767K), Amoxicillin ($539K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to New Zealand
161 Indian pharmaceutical companies export finished formulations to New Zealand. Leading exporters include Ipca Laboratories Limited, Mylan Laboratories Limited, Cipla Limited, Medreich Limited. The top exporter accounts for 57.4% of total India–New Zealand pharma exports. Source: Indian Customs (DGFT).
Top New Zealand Buyers from India
263 companies in New Zealand import pharmaceutical formulations from India. Top buyers include Viatris Limited,, Viatris Limited, Hellmann Worldwide Logistics, Rex Medical Ltd,. The largest buyer accounts for 8.0% of India–New Zealand pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to New Zealand Pharmaceutical Shipments
Indian Export Ports
New Zealand Import Ports
Trade Statistics
Other Destinations
Product Routes
India–New Zealand Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-New Zealand pharmaceutical trade corridor has experienced significant growth over the past decade. In 2024, India exported pharmaceutical products valued at $59.23 million to New Zealand, marking a substantial increase from previous years. This upward trajectory reflects the strengthening economic ties and the expanding demand for Indian pharmaceutical formulations in the New Zealand market.
A pivotal milestone in this bilateral trade relationship was the conclusion of the India-New Zealand Free Trade Agreement (FTA) in December 2025. The FTA is expected to eliminate tariffs on 90 pharmaceutical tariff lines, enhancing the competitiveness of Indian pharmaceutical exports. This agreement is anticipated to bolster India's market share in New Zealand's pharmaceutical imports, fostering deeper economic integration between the two nations.
2India's Market Position
India holds a significant position in New Zealand's pharmaceutical import landscape. In 2024, India was the largest supplier of pharmaceutical products to New Zealand, with exports valued at $81.43 million. This dominance underscores India's strategic importance as a key partner in meeting New Zealand's pharmaceutical needs.
The India-New Zealand FTA further solidifies this position by providing zero-duty access for Indian pharmaceutical exports. This preferential treatment is expected to enhance India's competitiveness in the New Zealand market, potentially increasing its share of New Zealand's pharmaceutical imports.
3Recent Developments
The conclusion of the India-New Zealand FTA in December 2025 marked a significant development in bilateral trade relations. The agreement is poised to eliminate tariffs on 90 pharmaceutical tariff lines, providing Indian exporters with enhanced market access and competitiveness in New Zealand.
Additionally, the repeal of the Therapeutic Products Bill in December 2024 has led to the initiation of new regulatory frameworks for medicines, medical devices, and natural health products in New Zealand. This regulatory evolution may impact the registration and approval processes for pharmaceutical products, presenting both challenges and opportunities for Indian exporters.
New Zealand Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export to New Zealand must adhere to the regulatory requirements set by the New Zealand Medicines and Medical Devices Safety Authority (Medsafe). The registration process involves submitting a comprehensive dossier that includes product information, manufacturing details, and evidence of compliance with Good Manufacturing Practice (GMP) standards. The Common Technical Document (CTD) format is typically used for this submission.
Bioequivalence studies are often required to demonstrate that the generic product performs in the same manner as the innovator drug. The timeline for product registration can vary, but it generally takes several months, depending on the complexity of the product and the completeness of the submitted documentation.
2GMP & Facility Requirements
New Zealand mandates that pharmaceutical products imported into the country be manufactured in facilities that comply with WHO-GMP standards. Indian manufacturers must ensure that their production sites meet these international standards to gain approval for export. The inspection process involves a thorough evaluation of the manufacturing facility's compliance with GMP guidelines.
The India-New Zealand FTA includes provisions for mutual recognition of major regulatory inspections, which is expected to streamline the approval process for Indian pharmaceutical products in New Zealand. This alignment aims to reduce compliance barriers and expedite market entry for Indian exporters.
3Import Documentation
To import pharmaceutical products into New Zealand, Indian exporters must provide several key documents, including:
- Certificate of Pharmaceutical Product (CPP): Certifies that the product is authorized for sale in the country of origin.
- Certificate of Analysis (CoA): Provides detailed information on the product's composition and quality.
- GMP Certificate: Confirms that the manufacturing facility adheres to GMP standards.
These documents are essential for customs clearance and regulatory approval. The import process also involves compliance with New Zealand's customs procedures, which include the payment of applicable duties and taxes, as well as adherence to any specific import restrictions or requirements.
Product Categories & Therapeutic Trends — India to New Zealand
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
The majority of India's pharmaceutical exports to New Zealand consist of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. In 2024, these products accounted for a significant portion of the export value. This trend aligns with New Zealand's healthcare needs, which are increasingly met by ready-to-use pharmaceutical products.
The therapeutic areas dominating these exports include cardiovascular drugs, central nervous system agents, and anti-infectives. This focus reflects the prevalent health concerns in New Zealand, such as heart disease, diabetes, and infectious diseases.
2Emerging Opportunities
The India-New Zealand FTA is expected to open new avenues for Indian pharmaceutical exporters, particularly in the biologics and biosimilars sectors. With the global patent expiry of several blockbuster biologic drugs, there is a growing demand for biosimilars, presenting an opportunity for Indian companies to expand their product offerings in New Zealand.
Additionally, the evolving regulatory landscape in New Zealand may create opportunities for Indian exporters to introduce innovative formulations and new therapeutic areas, provided they can navigate the updated approval processes effectively.
3Demand Drivers
New Zealand's aging population and the increasing prevalence of chronic diseases are significant drivers of pharmaceutical import demand. The country's healthcare system relies on a diverse range of imported pharmaceutical products to meet the needs of its population. The India-New Zealand FTA is expected to further stimulate demand for Indian pharmaceutical products by enhancing their competitiveness and availability in the New Zealand market.
Trade Policy & Tariff Intelligence — India and New Zealand
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Under the India-New Zealand FTA, New Zealand has committed to providing 100% duty-free market access across all tariff lines for India's current exports, including pharmaceuticals. This eliminates previous peak tariffs of up to 10% on key products, enhancing the price competitiveness of Indian pharmaceutical exports in New Zealand.
The FTA also outlines a phased elimination of tariffs on certain products, balancing liberalization with the protection of sensitive sectors. This structured approach aims to provide Indian exporters with improved market access while considering New Zealand's domestic economic priorities.
2Trade Agreements
The India-New Zealand FTA, concluded in December 2025, is a landmark agreement that is expected to significantly boost bilateral trade, including in the pharmaceutical sector. The agreement includes provisions for mutual recognition of major regulatory inspections, which is anticipated to streamline the approval process for Indian pharmaceutical products in New Zealand.
Additionally, the Comprehensive and Progressive Agreement for Trans-Pacific Partnership (CPTPP), of which New Zealand is a member, includes provisions related to pharmaceuticals. However, these provisions do not change New Zealand's Pharmaceutical Management Agency (PHARMAC) model or its ability to negotiate the best price for medicines for New Zealanders. (mfat.govt.nz)
3IP & Patent Landscape
The India-New Zealand FTA includes commitments on intellectual property, including provisions related to patents and data protection. However, the agreement does not contain obligations on data protection or market protection for new pharmaceutical products, including biologics. This means that Indian generic manufacturers may face fewer barriers to entry in the New Zealand market, potentially enhancing their competitiveness. (mfat.govt.nz)
Supply Chain & Logistics — India to New Zealand Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to New Zealand primarily utilize sea freight, with major ports such as Nhava Sheva (Jawaharlal Nehru Port) serving as key export points. The transit time for sea shipments from India to New Zealand typically ranges from 20 to 30 days, depending on the specific ports and shipping routes.
Air freight is also utilized for time-sensitive deliveries, with transit times of approximately 10 to 15 days. However, air freight costs are significantly higher than sea freight, which can impact the overall cost-effectiveness of shipments.
2Port Infrastructure
In India, major export ports for pharmaceutical products include Nhava Sheva (Jawaharlal Nehru Port), which handles a significant portion of the country's pharmaceutical exports. In New Zealand, Auckland is the primary import port for pharmaceutical products, accounting for a substantial share of the country's pharmaceutical imports.
The efficiency and capacity of these ports are crucial for ensuring timely and secure delivery of pharmaceutical shipments. Ongoing investments in port infrastructure and logistics are expected to further enhance the efficiency of the India-New Zealand pharmaceutical trade corridor.
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is paramount. Both India
FAQ — India to New Zealand Pharmaceutical Trade
What is the total value of India's pharmaceutical export to New Zealand?
India exported pharmaceuticals worth $117.1M to New Zealand across 6,137 verified shipments.
Who are the top Indian pharmaceutical exporters to New Zealand?
1. IPCA LABORATORIES LIMITED — $67.3M. 2. MYLAN LABORATORIES LIMITED — $14.3M. 3. CIPLA LIMITED — $9.5M. Total: 161 suppliers.
Which companies in New Zealand import pharmaceuticals from India?
1. VIATRIS LIMITED, — $9.4M. 2. VIATRIS LIMITED — $4.3M. 3. HELLMANN WORLDWIDE LOGISTICS — $3.7M. 263 buyers total.
What pharmaceutical products does India export most to New Zealand?
1. Zopiclone Actavis? 7.5mg Tablets ( 's Pack=6752000 Tablets) ($33.3M, 28.4%); 2. Simvastatin 40mg Tablets ( 's Pack =4305600 Tablets)[each Tablets Cont:simvasta… ($8.1M, 7.0%); 3. Harmless Medicines:simvastatin 20mg Tablets ( 's Pack =4357440 Tablets)[each Ta ($4.9M, 4.2%); 4. Zista 10mg Tablets ( 's Pack=4737500 Tablets) ($3.2M, 2.7%); 5. Arrow-losartan Potassium and Hydrochlorothiazide 12.5mg Tablets(losartan Potass… ($2.4M, 2.0%)
Which ports handle pharmaceutical shipments from India to New Zealand?
Export: NHAVA SHEVA SEA, NHAVA SHEVA SEA (INNSA1), JNPT/ NHAVA SHEVA SEA, SAHAR AIR CARGO ACC (INBOM4), JNPT. Import: Auckland, AUCKLAND, N?A, Tauranga, TAURANGA.
Why does New Zealand import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $117.1M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply New Zealand?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to New Zealand pharmaceutical trade?
$19.1K per consignment across 6,137 shipments.
How many Indian pharmaceutical companies export to New Zealand?
161 Indian companies. Largest: IPCA LABORATORIES LIMITED with $67.3M.
How can I find verified Indian pharmaceutical suppliers for New Zealand?
TransData Nexus covers 161 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to New Zealand Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 6,137 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to New Zealand identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 6,137 verified shipments from 161 Indian exporters to 263 New Zealand buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
161 Exporters
263 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists