India to Italy Pharmaceutical Export
Bilateral Trade Intelligence · $66.4M Total Trade · 201 Exporters · 452 Buyers · DGFT Verified · Updated March 2026
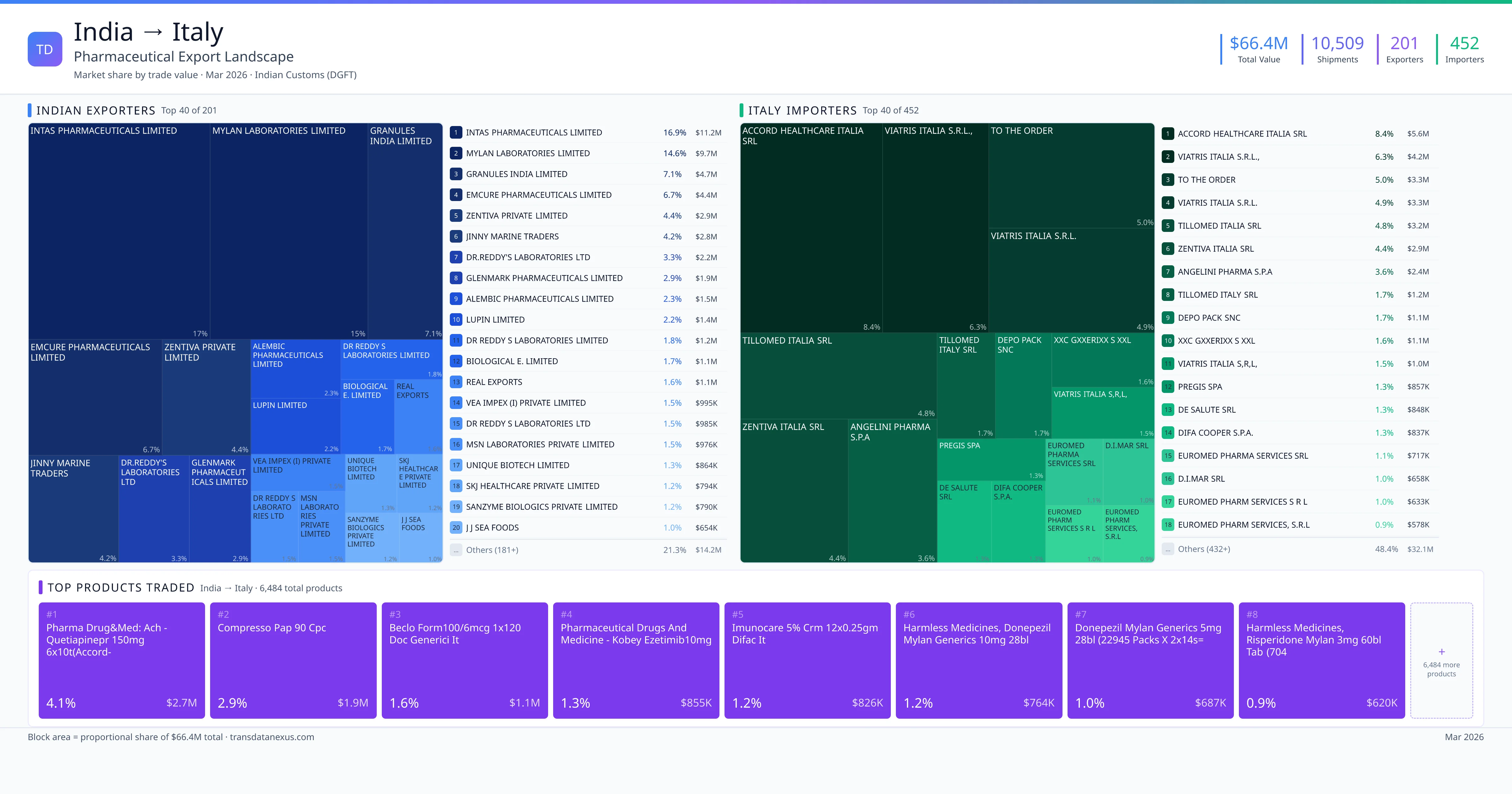
India exported $66.4M worth of pharmaceutical formulations to Italy across 10,509 verified shipments, sourced from 201 Indian exporters supplying 452 Italy buyers. The top exporters are INTAS PHARMACEUTICALS LIMITED ($11.2M) and MYLAN LABORATORIES LIMITED ($9.7M). The leading products are Ramipril ($387K) and Cabazitaxel ($376K). Average shipment value: $6.3K.
Top Pharmaceutical Formulations — India to Italy
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Pharma Drug&med: Ach - Quetiapinepr 150mg 6x10t(accord-italy Quetiapine Accord1… | $2.7M | 4.1% |
| 2 | Compresso Pap 90 Cpc | $1.9M | 2.9% |
| 3 | Beclo Form100/6mcg 1x120 Doc Generici It | $1.1M | 1.6% |
| 4 | Pharmaceutical Drugs and Medicine - Kobey Ezetimib10mg - As Per Invoice | $854.6K | 1.3% |
| 5 | Imunocare 5% Cream 12x0.25gm Difac It | $826.3K | 1.2% |
| 6 | Harmless Medicines, Donepezil Mylan Generics 10mg 28bl (15028 Packs) | $764.0K | 1.2% |
| 7 | Donepezil Mylan Generics 5mg 28bl (22945 Packs X 2x14s=642460 ) | $687.4K | 1.0% |
| 8 | Harmless Medicines, Risperidone Mylan 3mg 60bl Tablets (7044 Packs) | $619.8K | 0.9% |
| 9 | Pangran Granules | $454.5K | 0.7% |
| 10 | Azacitadine Vial 100mg X 1 It | $430.8K | 0.6% |
| 11 | Pharmaceutical Drugs and Medicineclozapina(cloza) 25mg Each Uncoated Tablet Con… | $411.0K | 0.6% |
| 12 | Pharmaceutical Products Ramipril Pc Granules 850g/1kg (428.550 Kg) Exempted Mat… | $387.4K | 0.6% |
| 13 | Cabazitaxel Dr. Reddy's 60mg/1.5ml1s It | $376.0K | 0.6% |
| 14 | Metformin Tablets 1000mg 60bl It Myg Metformina Mylan Generics 1000 Mg(15x4) | $332.8K | 0.5% |
| 15 | Daptomycin for Injection 500 MG Vial 1s Qty 23107 | $315.6K | 0.5% |
India exports 18+ pharmaceutical formulations to Italy with a combined trade value of $66.4M. Key products include Ramipril ($387K), Cabazitaxel ($376K), Metformin ($333K), Clopidogrel ($314K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 10,509 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Italy Trade Routes
These are the top pharmaceutical products exported from India to Italy, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Italy buyers, regulatory requirements, and logistics for that specific product corridor. Products include Ramipril ($387K), Cabazitaxel ($376K), Metformin ($333K), Clopidogrel ($314K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Italy
201 Indian pharmaceutical companies export finished formulations to Italy. Leading exporters include Intas Pharmaceuticals Limited, Mylan Laboratories Limited, Granules India Limited, Emcure Pharmaceuticals Limited. The top exporter accounts for 16.9% of total India–Italy pharma exports. Source: Indian Customs (DGFT).
Top Italy Buyers from India
452 companies in Italy import pharmaceutical formulations from India. Top buyers include Accord Healthcare Italia Srl, Viatris Italia S.r.l.,, To The Order, Viatris Italia S.r.l.. The largest buyer accounts for 8.4% of India–Italy pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Italy Pharmaceutical Shipments
Indian Export Ports
Italy Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Italy Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Italy pharmaceutical trade corridor has experienced significant growth over the past two decades. Between 1991 and 2011, bilateral trade expanded twelvefold, from EUR 708 million to EUR 8.5 billion. This surge was driven by India's burgeoning pharmaceutical sector, which became a major global supplier of generic medicines. In 2024, India's pharmaceutical exports to Italy were valued at $63.51 million, reflecting a steady upward trajectory.
Key milestones in this relationship include the signing of a Memorandum of Understanding (MoU) on November 29, 2017, aimed at enhancing cooperation in the health sector. This agreement focused on pooling technical, financial, and human resources to upgrade medical infrastructure, education, and research in both countries. Additionally, the India-European Free Trade Association (EFTA) Trade and Economic Partnership Agreement, signed on March 10, 2024, is set to enter into force on October 1, 2025. This agreement is expected to further strengthen trade relations, including in the pharmaceutical sector.
2India's Market Position
India's pharmaceutical exports to Italy amounted to $63.51 million in 2024, accounting for a significant portion of Italy's total pharmaceutical imports. While exact figures for Italy's total pharmaceutical imports are not specified, India's contribution underscores the strategic importance of this corridor. The Indian pharmaceutical sector's growth has been remarkable, with exports outpacing global growth rates, solidifying India's position as a key player in the global pharmaceutical market.
3Recent Developments
In January 2026, the India-European Union (EU) Free Trade Agreement (FTA) was finalized, marking a significant development in bilateral trade relations. This agreement includes substantial tariff reductions on pharmaceutical products, with tariffs on EU exports to India dropping from 11% to zero for almost all products. This reduction is expected to lower costs for Indian patients and enhance the competitiveness of Indian pharmaceutical companies in the European market. Furthermore, in February 2026, the Indian Pharmaceutical Alliance highlighted the potential of the India-EU FTA to position both regions as major suppliers of affordable and quality medicines globally.
Italy Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Italy must obtain marketing authorization through the European Medicines Agency (EMA). For generic medicines, the application should demonstrate that the product is bioequivalent to the reference medicine, which is already authorized in the European Union. This involves providing evidence of the same qualitative and quantitative composition in active substances, the same pharmaceutical form, and bioequivalence studies. (ema.europa.eu)
The Common Technical Document (CTD) format is the standard for submitting marketing authorization applications in the EU. The CTD is organized into five modules: Module 1 (Administrative and prescribing information), Module 2 (Summaries), Module 3 (Quality), Module 4 (Non-clinical study reports), and Module 5 (Clinical study reports). This standardized format facilitates the review process and ensures consistency across applications.
2GMP & Facility Requirements
Indian manufacturing facilities exporting to Italy must comply with Good Manufacturing Practice (GMP) standards as outlined by the European Union. The Italian Medicines Agency (AIFA) is responsible for coordinating and managing inspections to verify compliance with GMP for manufacturers and importers of active substances located in Italy. These inspections can also be conducted at manufacturing plants located in non-EU countries, including India, based on a relevant interest by AIFA or upon the manufacturer's request. (aifa.gov.it)
3Import Documentation
To import pharmaceutical products into Italy, Indian companies must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): This certificate attests to the product's marketing authorization and compliance with GMP in the country of origin.
- Certificate of Analysis (CoA): This document provides detailed information on the product's quality attributes, including test results for identity, purity, potency, and other relevant parameters.
- GMP Certificate: This certificate confirms that the manufacturing facility adheres to GMP standards.
These documents are essential for customs procedures and regulatory compliance when importing pharmaceutical products into Italy. (health.ec.europa.eu)
Product Categories & Therapeutic Trends — India to Italy
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Italy are predominantly concentrated in the central nervous system (CNS) and cardiovascular therapeutic areas. The leading product, Quetiapine 150 mg, accounts for 4.1% of the total export value, indicating a significant demand for antipsychotic medications in Italy. This aligns with Italy's healthcare needs, where mental health disorders are prevalent, necessitating effective antipsychotic treatments. Additionally, cardiovascular drugs, such as those containing Amlodipine, represent a substantial portion of exports, reflecting Italy's focus on managing hypertension and related cardiovascular conditions.
The prominence of these therapeutic categories is further supported by the export data, with Quetiapine 150 mg and Amlodipine-based products being among the top exported items. This trend underscores the alignment between India's pharmaceutical offerings and Italy's therapeutic requirements, highlighting the strategic importance of CNS and cardiovascular drugs in the bilateral trade.
2Emerging Opportunities
The upcoming patent expirations of several biologic drugs in Italy present significant opportunities for Indian pharmaceutical exporters. Notably, Humira (Adalimumab), a leading biologic used for various autoimmune diseases, is set to lose patent protection in 2026. This event is expected to open the market to biosimilars, creating a demand for cost-effective alternatives. Indian manufacturers, with their established expertise in producing high-quality generics and biosimilars, are well-positioned to meet this demand, offering affordable options to the Italian market.
The Italian government's active promotion of biosimilar adoption to manage pharmaceutical expenditure further enhances this opportunity. With biosimilars accounting for 49.2% of the pharmaceutical market in 2023, there is a clear trend towards embracing these alternatives. Indian exporters can leverage this shift by providing biosimilars that align with Italy's therapeutic needs and cost-containment strategies.
3Demand Drivers
Italy's aging population and the associated increase in chronic diseases are primary drivers of pharmaceutical import demand. The country's healthcare system faces challenges in managing the growing burden of conditions such as cardiovascular diseases, diabetes, and neurodegenerative disorders. This demographic shift necessitates a continuous supply of effective medications, creating a sustained demand for pharmaceutical imports.
Furthermore, Italy's healthcare spending trends indicate a focus on cost-effective solutions. The government's emphasis on generic and biosimilar drugs to reduce healthcare expenditures aligns with the availability of high-quality generics from India. This policy direction not only supports the demand for affordable medications but also presents an opportunity for Indian exporters to contribute to Italy's healthcare objectives.
Trade Policy & Tariff Intelligence — India and Italy
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Italy, as a member of the European Union (EU), applies the EU's Common Customs Tariff (CCT) to imports, including pharmaceutical products classified under Harmonized System (HS) codes 3003 and 3004. The Most Favored Nation (MFN) duty rates for these products are generally set at 0%, facilitating duty-free entry for imports from countries like India. This preferential treatment is contingent upon the products meeting the EU's rules of origin criteria, which ensure that the goods are sufficiently transformed or processed within the exporting country.
In addition to MFN rates, the EU has established preferential duty rates for countries with which it has Free Trade Agreements (FTAs). However, as of March 2026, there is no specific FTA between India and the EU that would grant preferential tariff rates beyond the standard MFN treatment. Consequently, Indian pharmaceutical exports to Italy benefit from the MFN duty rates without additional preferential reductions.
2Trade Agreements
Currently, there is no Free Trade Agreement (FTA) between India and Italy. Consequently, Indian pharmaceutical exports to Italy are subject to the EU's Common Customs Tariff (CCT), which generally imposes a 0% duty on pharmaceutical products under HS codes 3003 and 3004. This MFN treatment facilitates duty-free entry for Indian pharmaceutical products into the Italian market, provided they meet the EU's rules of origin criteria.
While India and the EU have engaged in discussions regarding trade relations, as of March 2026, no specific FTA has been established. Therefore, Indian pharmaceutical exports to Italy continue to benefit from the MFN duty rates without additional preferential reductions.
3IP & Patent Landscape
Italy's intellectual property (IP) framework, aligned with EU regulations, offers robust protection for pharmaceutical patents, encompassing both active ingredients and formulations. This protection extends to data exclusivity periods, during which generic manufacturers are restricted from using the original innovator's clinical trial data to obtain marketing authorization. The standard data exclusivity period in Italy is 8 years, with an additional 2 years of market exclusivity, allowing the originator company a total of 10 years before generics can enter the market.
For Indian pharmaceutical companies aiming to enter the Italian market with generic versions of patented drugs, it is crucial to navigate this IP landscape carefully. Strategies may include challenging patent validity, seeking compulsory licenses under specific conditions, or focusing on developing generics for drugs approaching the end of their exclusivity periods. Understanding and complying with Italy's IP regulations is essential for successful market entry and competition.
Supply Chain & Logistics — India to Italy Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Italy primarily utilize sea and air freight, each with distinct transit times and routes.
Sea Freight: The journey from Indian ports to Italian destinations typically spans 20 to 30 days, depending on specific ports and shipping conditions. For instance, shipments from Nhava Sheva (Mumbai) to Genoa have an average transit time of approximately 25 days. This route often passes through the Suez Canal, a critical artery for global trade. However, disruptions in the Suez Canal, such as the blockage in March 2021, can significantly impact transit times and logistics planning.
Air Freight: Air shipments offer a faster alternative, with transit times ranging from 4 to 8 days. Direct flights from major Indian airports like Delhi (DEL) to Milan (MXP) are available, facilitating swift delivery of pharmaceutical products. Air freight is particularly advantageous for high-value or urgently needed pharmaceutical items.
2Port Infrastructure
Key Indian export ports handling pharmaceutical shipments include Mundra Sea Port, Sahar Air Cargo (Mumbai), and Nhava Sheva Sea Port (JNPT). Mundra Sea Port, for example, accounts for 4.5% of the total pharmaceutical exports to Italy. In Italy, primary import ports are Milan Malpensa Airport and the Port of Genoa, which collectively manage a significant portion of pharmaceutical imports. Milan Malpensa Airport, in particular, serves as a major hub for air cargo, including pharmaceuticals.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical products is paramount. Shipments must adhere to Good Distribution Practice (GDP) guidelines, ensuring proper storage and handling throughout transit. This includes using validated cold chain logistics and ensuring compliance with both Indian and Italian regulations. In Italy, the Agenzia Italiana del Farmaco (AIFA) oversees the importation of pharmaceutical products, enforcing strict standards to guarantee product safety and efficacy. Additionally, customs clearance procedures in Italy require thorough documentation, including certificates of analysis and temperature control declarations, to verify compliance with regulatory standards.
Market Opportunity Assessment — Italy for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Italy's pharmaceutical market was valued at approximately $44.23 billion in 2026 and is projected to reach $62.67 billion by 2035, expanding at an annualized rate of 3.95%. The generic pharmaceuticals segment is a significant contributor, with revenues expected to grow from $17.005 billion in 2025 to $30.704 billion by 2033, reflecting a compound annual growth rate (CAGR) of 7.3% from 2026 to 2033. In 2022, generics accounted for 55.54% of the pharmaceutical market volume, indicating a strong and growing presence.
2Healthcare System
Italy's National Health Service (Servizio Sanitario Nazionale, SSN) provides universal coverage to all citizens and legal residents, ensuring access to essential healthcare services. In 2022, health expenditure amounted to 9% of GDP, with public funding accounting for 74% of the total healthcare spending. (eurohealthobservatory.who.int) The SSN is regionally organized, with 19 regions and two autonomous provinces responsible for delivering care through local health units.
3Opportunity for Indian Exporters
The Italian pharmaceutical market presents substantial opportunities for Indian exporters, particularly in the generic drugs segment. In 2022, generics accounted for 55.54% of the pharmaceutical market volume, indicating a strong and growing presence. The market is expected to grow at a CAGR of 7.3% from 2026 to 2033, with the branded generics market projected to reach $8.113 billion by 2033, growing at a CAGR of 6.5% from 2026 to 2033. Given the existing trade volume of $66.4 million in finished pharmaceutical formulations between India and Italy, there is significant potential for Indian exporters to expand their presence in the Italian market. Focusing on high-demand therapeutic areas and aligning with Italy's healthcare priorities can further enhance market penetration.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Italy
Competing origins, India's edge, challenges and threats
1Competing Origins
Italy's pharmaceutical market is supplied by a diverse array of countries, with Germany, the Netherlands, and Belgium being prominent contributors. In 2024, Italy imported pharmaceutical products valued at approximately $32.1 billion. Germany led these imports, with a value of $5.73 billion, followed by the Netherlands at $3.73 billion, and Belgium at $1.3 billion.
China also plays a role in supplying pharmaceutical products to Italy, though its share is comparatively smaller. In 2023, Italy imported laboratory, hygienic, or pharmaceutical glassware from China valued at $2.64 million.
India's pharmaceutical exports to Italy have been growing steadily. In 2024, India exported pharmaceutical products to Italy valued at $63.51 million. This positions India as a significant supplier, though its share is smaller compared to European countries.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages:
- Cost Competitiveness: India's ability to produce high-quality pharmaceuticals at lower costs makes it an attractive supplier for price-sensitive markets.
- Regulatory Compliance: India has a substantial number of US FDA-approved manufacturing facilities, indicating adherence to stringent international quality standards.
- English-Speaking Workforce: The widespread use of English facilitates smoother communication and collaboration with international partners.
- Broad Portfolio: India's pharmaceutical sector encompasses a wide range of products, including generics, active pharmaceutical ingredients (APIs), vaccines, and biosimilars, catering to diverse market needs.
3Challenges & Threats
Despite its strengths, India faces several challenges in the Italian pharmaceutical market:
- Regulatory Compliance: Navigating the complex regulatory landscape of the European Medicines Agency (EMA) and Italy's National Competent Authority requires significant resources and expertise.
- Competition from China: China's growing pharmaceutical industry poses a competitive threat, especially in cost-sensitive segments, potentially impacting India's market share.
- Quality Perception: While India maintains high manufacturing standards, any lapses can affect its reputation, making it crucial to consistently meet quality expectations.
4India's Position Over Time
India's position in the Italian pharmaceutical market has been evolving positively. The increase in export value from $11.48 million in 2022 to $23.34 million in 2023 indicates a growing presence. This upward trend reflects India's expanding footprint and the strengthening of trade relations with Italy.
FAQ — India to Italy Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Italy?
India exported pharmaceuticals worth $66.4M to Italy across 10,509 verified shipments.
Who are the top Indian pharmaceutical exporters to Italy?
1. INTAS PHARMACEUTICALS LIMITED — $11.2M. 2. MYLAN LABORATORIES LIMITED — $9.7M. 3. GRANULES INDIA LIMITED — $4.7M. Total: 201 suppliers.
Which companies in Italy import pharmaceuticals from India?
1. ACCORD HEALTHCARE ITALIA SRL — $5.6M. 2. VIATRIS ITALIA S.R.L., — $4.2M. 3. TO THE ORDER — $3.3M. 452 buyers total.
What pharmaceutical products does India export most to Italy?
1. Pharma Drug&med: Ach - Quetiapinepr 150mg 6x10t(accord-italy Quetiapine Accord1… ($2.7M, 4.1%); 2. Compresso Pap 90 Cpc ($1.9M, 2.9%); 3. Beclo Form100/6mcg 1x120 Doc Generici It ($1.1M, 1.6%); 4. Pharmaceutical Drugs and Medicine - Kobey Ezetimib10mg - As Per Invoice ($854.6K, 1.3%); 5. Imunocare 5% Cream 12x0.25gm Difac It ($826.3K, 1.2%)
Which ports handle pharmaceutical shipments from India to Italy?
Export: MUNDRA SEA, SAHAR AIR, NHAVA SHEVA SEA (INNSA1), SAHAR AIR CARGO ACC (INBOM4), DELHI AIR. Import: MILAN - MALPENSA, MILAN, Genoa, GENOA, ANCONA.
Why does Italy import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $66.4M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Italy?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Italy pharmaceutical trade?
$6.3K per consignment across 10,509 shipments.
How many Indian pharmaceutical companies export to Italy?
201 Indian companies. Largest: INTAS PHARMACEUTICALS LIMITED with $11.2M.
How can I find verified Indian pharmaceutical suppliers for Italy?
TransData Nexus covers 201 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Italy Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 10,509 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Italy identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 10,509 verified shipments from 201 Indian exporters to 452 Italy buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
201 Exporters
452 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists