India to Brazil Pharmaceutical Export
Bilateral Trade Intelligence · $695.5M Total Trade · 194 Exporters · 1990 Buyers · DGFT Verified · Updated March 2026
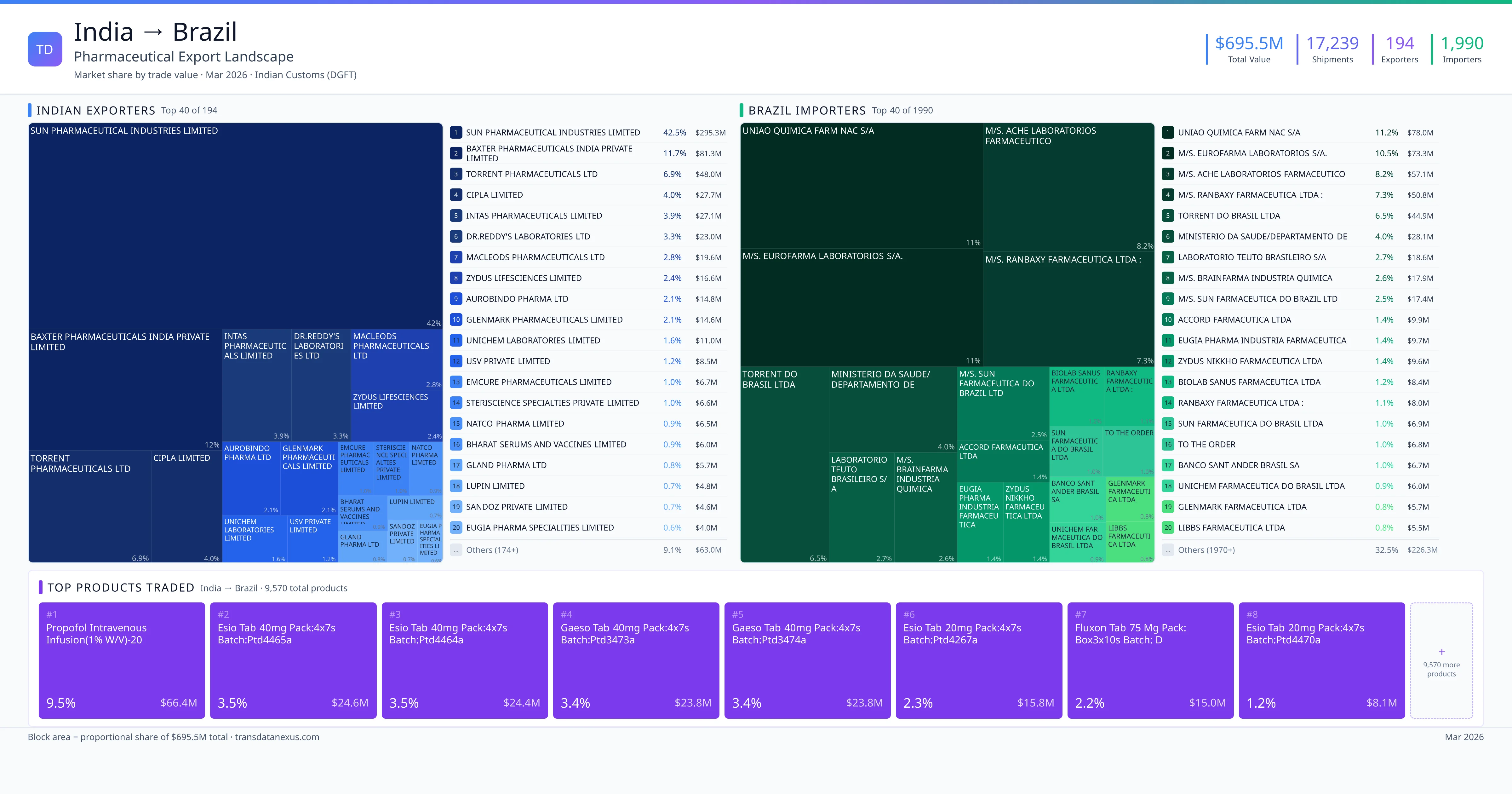
India exported $695.5M worth of pharmaceutical formulations to Brazil across 17,239 verified shipments, sourced from 194 Indian exporters supplying 1990 Brazil buyers. The top exporters are SUN PHARMACEUTICAL INDUSTRIES LIMITED ($295.3M) and BAXTER PHARMACEUTICALS INDIA PRIVATE LIMITED ($81.3M). The leading products are Isoniazid ($7.7M) and Cefuroxime ($5.1M). Average shipment value: $40.3K.
Top Pharmaceutical Formulations — India to Brazil
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Propofol Intravenous Infusion(1% W/v)-20 | $66.4M | 9.5% |
| 2 | Esio Tablets 40mg Pack:4x7s Batch:ptd4465a | $24.6M | 3.5% |
| 3 | Esio Tablets 40mg Pack:4x7s Batch:ptd4464a | $24.4M | 3.5% |
| 4 | Gaeso Tablets 40mg Pack:4x7s Batch:ptd3473a | $23.8M | 3.4% |
| 5 | Gaeso Tablets 40mg Pack:4x7s Batch:ptd3474a | $23.8M | 3.4% |
| 6 | Esio Tablets 20mg Pack:4x7s Batch:ptd4267a | $15.8M | 2.3% |
| 7 | Fluxon Tablets 75 MG Pack: Box3x10s Batch: D | $15.0M | 2.2% |
| 8 | Esio Tablets 20mg Pack:4x7s Batch:ptd4470a | $8.1M | 1.2% |
| 9 | Gaeso Tablets 20mg Pack:4x7s Batch:ptd3839a | $8.0M | 1.2% |
| 10 | Isoniazid 300 MG / Rifapentine 300 MG Coated Bno:nie2356b Mfg. Dt.10/23 Exp. Dt… | $7.7M | 1.1% |
| 11 | Nortriptyline Capsules 50 MG Pack:box3x10s Ba | $6.0M | 0.9% |
| 12 | Aerodini Hfa Inhaler (1 X200 Md) . | $6.0M | 0.9% |
| 13 | Cefuroxime Axe. Tablets 500mg Pack:10s S | $5.1M | 0.7% |
| 14 | Nidhi Capsules 150mg Each Capsule Contains Ni | $5.0M | 0.7% |
| 15 | Nortriptyline Capsules 50mg Pack:box3x10s Bat | $4.2M | 0.6% |
India exports 20+ pharmaceutical formulations to Brazil with a combined trade value of $695.5M. Key products include Isoniazid ($7.7M), Cefuroxime ($5.1M), Artemether ($3.9M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 17,239 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Brazil Trade Routes
These are the top pharmaceutical products exported from India to Brazil, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Brazil buyers, regulatory requirements, and logistics for that specific product corridor. Products include Isoniazid ($7.7M), Cefuroxime ($5.1M), Artemether ($3.9M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Brazil
194 Indian pharmaceutical companies export finished formulations to Brazil. Leading exporters include Sun Pharmaceutical Industries Limited, Baxter Pharmaceuticals India Private Limited, Torrent Pharmaceuticals Ltd, Cipla Limited. The top exporter accounts for 42.5% of total India–Brazil pharma exports. Source: Indian Customs (DGFT).
Top Brazil Buyers from India
1990 companies in Brazil import pharmaceutical formulations from India. Top buyers include Uniao Quimica Farm Nac S/a, M/s. Eurofarma Laboratorios S/a., M/s. Ache Laboratorios Farmaceutico, M/s. Ranbaxy Farmaceutica Ltda :. The largest buyer accounts for 11.2% of India–Brazil pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Brazil Pharmaceutical Shipments
Indian Export Ports
Brazil Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Brazil Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Brazil pharmaceutical trade corridor has experienced significant growth over the past decade. In fiscal year 2024-25, India's pharmaceutical exports to Brazil reached approximately $778 million, marking an 18.74% increase from the previous fiscal year. This upward trajectory continued into the April–January period of fiscal year 2026, with exports totaling $740.33 million, reflecting a 17.03% growth compared to the same period in fiscal year 2025.
Key milestones in this bilateral trade relationship include the establishment of the India-Brazil Healthcare Business Gateway in February 2026. Organized by the Pharmaceuticals Export Promotion Council of India (Pharmexcil) and Amoveri Farma, this initiative aimed to facilitate market access and streamline regulatory pathways for Indian pharmaceutical exports to Brazil. The meeting focused on expanding cooperation in oncology medicines, biosimilars, complex therapies, and hospital technologies, underscoring the strategic importance of this partnership.
2India's Market Position
India has solidified its position as a significant supplier of pharmaceutical products to Brazil. In fiscal year 2024-25, Brazil accounted for approximately 2.8% of India's total pharmaceutical exports, with exports exceeding $778 million. During the April–January period of fiscal year 2026, exports to Brazil reached $740.33 million, highlighting the growing importance of this market for Indian pharmaceutical manufacturers.
The strategic importance of this corridor is further emphasized by the focus on specialty medicines, biosimilars, and public healthcare supplies. Brazil's expanding healthcare demand and emphasis on affordable medicines present significant growth potential for Indian pharmaceutical exporters, particularly micro, small, and medium enterprises (MSMEs).
3Recent Developments
In February 2026, during Brazilian President Luiz Inácio Lula da Silva's official visit to India, both nations finalized agreements aimed at boosting pharmaceutical exports, cooperation in critical minerals, and support for the civil aviation sector. These agreements are expected to deepen trade and investment ties between the two countries, with pharmaceuticals emerging as a key growth driver.
Additionally, the India-Brazil Healthcare Business Gateway, held in New Delhi in February 2026, signaled the next growth phase in bilateral pharmaceutical trade. The meeting focused on expanding cooperation in oncology medicines, biosimilars, complex therapies, and hospital technologies, while facilitating structured access to the Brazilian market.
These developments underscore the strengthening of the India-Brazil pharmaceutical trade corridor, driven by strategic collaborations and a shared commitment to enhancing healthcare access and affordability.
Brazil Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Brazil must navigate the regulatory framework established by the Agência Nacional de Vigilância Sanitária (ANVISA). The registration process involves several key steps:
1. Designating a Local Representative: Foreign companies cannot directly engage with ANVISA; they must appoint a legally established partner in Brazil to act as their local representative. This entity assumes legal responsibility for the imported products within Brazilian territory. (gov.br)
2. Obtaining Operating Licenses: The local representative must secure an Operating License (LF) from the local or state health surveillance authority. This license authorizes the company's activities, confirming compliance with legal and regulatory requirements.
3. Product Registration: Once the company is authorized to operate, the next step is registering the product with ANVISA. This involves submitting a comprehensive dossier that includes detailed information about the product's composition, manufacturing process, quality control measures, and clinical data demonstrating its safety and efficacy.
Regarding dossier formats, ANVISA accepts submissions in the Common Technical Document (CTD) format, which is internationally recognized and facilitates the review process. The approval timeline can vary; however, as of January 2025, ANVISA's Resolution No. 954 introduced a simplified procedure for drug registration, post-registration, and renewal, aiming to enhance regulatory process management and sanitary control within the pharmaceutical sector.
2GMP & Facility Requirements
ANVISA requires that manufacturing facilities comply with Good Manufacturing Practices (GMP) to ensure the quality, safety, and efficacy of pharmaceutical products. As a member of the Pharmaceutical Inspection Co-operation Scheme (PIC/S), ANVISA aligns its GMP standards with internationally recognized guidelines. The primary regulation governing GMP for medicinal products in Brazil is ANVISA's Resolution RDC 301/2019, which harmonizes Brazil's requirements with PIC/S PE009 and modern ICH concepts.
For foreign manufacturers, ANVISA conducts inspections to verify GMP compliance. A valid GMP Certificate is a prerequisite for granting marketing authorization in Brazil for these products. The Brazilian legal representative of the foreign manufacturer is responsible for requesting GMP certificate renewals every two years. Depending on a risk analysis, ANVISA may decide to conduct a new on-site inspection or issue a certificate based solely on a documentary assessment and the available evidence from reliable sources. (gov.br)
3Import Documentation
When exporting pharmaceutical products to Brazil, Indian companies must ensure that all necessary import documentation is in order. This includes:
- Import License: The local representative must obtain an import license from ANVISA, authorizing the importation of the pharmaceutical products.
- Certificates of Pharmaceutical Product (CPP): A certificate issued by the competent authority in the exporting country, confirming that the product is authorized for sale and use in that country.
- Certificate of Analysis (CoA): A document issued by the manufacturer or a recognized laboratory, detailing the results of tests conducted on the product to ensure it meets specified quality standards.
- GMP Certificate: A certificate issued by the competent authority in the exporting country, confirming that the manufacturing facility complies with GMP standards.
These documents are essential for customs procedures and to demonstrate compliance with Brazilian regulations. It's important to note that the local representative in Brazil is responsible for ensuring that all import documentation is accurate and complete to facilitate the smooth importation and commercialization of the pharmaceutical products. (gov.br)
Product Categories & Therapeutic Trends — India to Brazil
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Brazil are predominantly concentrated in the therapeutic areas of anesthetics and gastrointestinal treatments. The leading product, propofol intravenous infusion (1% w/v), accounts for 9.5% of the total export value, amounting to $66.4 million. This is followed by a series of gastrointestinal medications, including Esio Tab 40mg and Gaeso Tab 40mg, each contributing approximately 3.5% of the export value. The prominence of these categories aligns with Brazil's substantial healthcare needs in anesthesia and digestive health. Anesthetics like propofol are essential for surgical procedures, reflecting the demand for advanced surgical interventions in Brazil. Gastrointestinal disorders are prevalent, necessitating effective treatments to manage conditions such as acid reflux and ulcers. The dominance of these therapeutic areas underscores the critical role of Indian pharmaceutical exports in addressing Brazil's healthcare challenges.
2Emerging Opportunities
The Brazilian pharmaceutical market presents emerging opportunities for Indian exporters, particularly in the realm of biosimilars and the management of chronic non-communicable diseases (NCDs). As Brazil's population ages, there is an increasing prevalence of NCDs such as cardiovascular diseases, diabetes, and cancers. This demographic shift is expected to drive demand for cost-effective treatments, creating a favorable environment for Indian generics and biosimilars. The Brazilian government's focus on expanding healthcare access and controlling costs further enhances the potential for Indian pharmaceutical companies to introduce affordable alternatives to expensive biologic therapies. Additionally, the projected increase in health spending in Brazil, expected to reach 12.6% of GDP by 2040, indicates a growing market for pharmaceutical products, including generics and biosimilars.
3Demand Drivers
Brazil's evolving disease burden and demographic trends are significant drivers of pharmaceutical import demand from India. The country is experiencing a rapid epidemiological transition, with chronic non-communicable diseases (NCDs) now representing the leading causes of mortality. In 2019, circulatory system diseases accounted for 27% of deaths, neoplasms for 17%, and chronic respiratory diseases for 12%. This shift is accompanied by an aging population, with projections indicating that by 2050, 21.9% of Brazilians will be 65 years or older, up from 8.9% in 2017. These demographic changes are expected to increase the demand for healthcare services and pharmaceutical products, particularly those addressing chronic conditions. Furthermore, Brazil's health expenditure is projected to rise, reaching 12.6% of GDP by 2040, reflecting a growing capacity to import and utilize pharmaceutical products. These factors collectively create a robust demand for pharmaceutical imports, presenting opportunities for Indian exporters to meet Brazil's evolving healthcare needs.
Trade Policy & Tariff Intelligence — India and Brazil
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Brazil's tariff structure for pharmaceutical formulations, classified under HS codes 3003 and 3004, is governed by the Mercosur Common External Tariff (CET). As of January 30, 2026, Brazil implemented revisions to its Most-Favored-Nation (MFN) rates, affecting various industrial and metal-related chapters. While specific MFN rates for pharmaceutical products are not detailed in the available sources, it is known that Mercosur member countries, including Brazil, maintain a CET that standardizes import duties across member states. This CET typically imposes tariffs on pharmaceutical imports, though the exact rates can vary based on product specifics and any applicable trade agreements.
Regarding preferential duty rates, Brazil's trade agreements, such as those with Egypt, Israel, Mercosur, and the Southern African Customs Union (SACU), have been updated to reflect new MFN rates and preferential treatments. These agreements may offer reduced tariffs or duty exemptions for pharmaceutical imports from partner countries. However, the precise preferential rates for pharmaceutical products under these agreements are not specified in the available information.
2Trade Agreements
As of March 2026, there is no publicly available information indicating a Free Trade Agreement (FTA) between India and Brazil. Both countries are members of the World Trade Organization (WTO) and have bilateral trade relations; however, a specific FTA between them does not exist. The absence of such an agreement means that pharmaceutical exports from India to Brazil are subject to Brazil's MFN tariff rates, without the benefit of preferential treatment that an FTA would provide.
3IP & Patent Landscape
Brazil's intellectual property (IP) framework, particularly concerning pharmaceuticals, has been a subject of international discussion. In October 2020, Brazil's National Health Regulatory Agency (ANVISA) established criteria for market authorization of medicines with synthetic and semisynthetic active principles for human use, categorized as new, innovative, generic, and similar. This regulation applies to pharmaceutical products under HS codes 3003 and 3004. While the specific details of patent provisions, data exclusivity, and compulsory licensing in Brazil are not fully detailed in the available sources, it is known that Brazil has engaged in legislative efforts to expand compulsory licensing beyond the Trade-Related Aspects of Intellectual Property Rights (TRIPS) standards. Such initiatives have raised concerns among international stakeholders about potential impacts on IP protection and investment in the pharmaceutical sector. For instance, the U.S. Chamber of Commerce has urged Brazil to align its IP standards with global best practices, citing issues like prolonged patent examination times and the lack of Regulatory Data Protection (RDP) for pharmaceuticals. These factors can influence the entry and competitiveness of Indian generic drugs in the Brazilian market.
Supply Chain & Logistics — India to Brazil Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Brazil predominantly utilize sea freight due to its cost-effectiveness for large volumes. The primary sea route involves transporting goods from major Indian ports to Brazilian ports. For instance, shipments from Nhava Sheva (Jawaharlal Nehru Port) to Santos typically take approximately 31 days.
Air freight, while faster, is less common for pharmaceutical exports from India to Brazil due to higher costs. However, it remains an option for time-sensitive deliveries. Air shipments from São Paulo (GRU) to major international hubs like Miami or Frankfurt usually take around 3–4 business days.
Recent disruptions in the Red Sea have not significantly impacted these shipping routes, as the primary sea lanes for India to Brazil trade do not traverse this area. Nonetheless, global shipping dynamics can be affected by such disruptions, potentially leading to longer transit times or increased costs.
2Port Infrastructure
In India, key export ports handling pharmaceutical shipments include Nhava Sheva (Jawaharlal Nehru Port), which accounts for 7.7% of the total shipments, and Dhannad ICD and Ahmedabad ICD, handling 2.5% and 0.1% respectively. These ports are equipped with facilities suitable for pharmaceutical exports, ensuring efficient handling and processing.
In Brazil, the primary import port for pharmaceutical products is Santos, receiving 17.1% of the total shipments. Other significant ports include Guarulhos, which handles 25.1% of the shipments, and Rio de Janeiro, which also serves as a key entry point for pharmaceutical imports. These ports are well-equipped to manage pharmaceutical imports, offering necessary infrastructure and services to facilitate smooth customs clearance and distribution.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical products throughout the supply chain is critical. Compliance with Good Distribution Practice (GDP) guidelines is mandatory, ensuring proper storage, handling, and transportation conditions. This includes using validated thermal packaging, continuous temperature monitoring, and comprehensive documentation such as Standard Operating Procedures (SOPs) and temperature logs.
In Brazil, the Agência Nacional de Vigilância Sanitária (ANVISA) regulates the importation of pharmaceutical products. All pharmaceutical imports must be registered and approved by ANVISA, and comply with Brazilian labeling standards, including Portuguese language requirements, expiration date policies, and traceability regulations.
Failure to adhere to these cold chain and compliance requirements can result in shipment delays, fines, or product rejection, underscoring the importance of meticulous planning and execution in pharmaceutical logistics.
Market Opportunity Assessment — Brazil for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Brazil's pharmaceutical market is a significant segment within Latin America's healthcare sector. In 2025, the market generated a revenue of approximately USD 36.5 million, with projections estimating it will reach USD 56.9 million by 2033, reflecting a compound annual growth rate (CAGR) of 5.7% from 2026 to 2033. This growth is driven by factors such as an aging population, increased prevalence of chronic diseases, and rising healthcare expenditures.
The generic drug segment plays a pivotal role in Brazil's pharmaceutical landscape. In 2025, the generic pharmaceuticals market was valued at approximately USD 13.7 billion, with expectations to reach USD 20.8 billion by 2033, indicating a CAGR of 5.2% from 2026 to 2033. This expansion is supported by government initiatives aimed at reducing healthcare costs and enhancing access to medications.
2Healthcare System
Brazil's healthcare system is characterized by a dual structure comprising the public Unified Health System (SUS) and a private sector. As of 2024, SUS provided services to approximately 72% of the population, while 51 million Brazilians had access to private healthcare. The government has implemented various insurance schemes to improve coverage and access to healthcare services.
The Brazilian government has also enacted policies to promote the use of generic drugs, aiming to reduce healthcare expenditures and increase medication accessibility. The Generic Drug Act of 1999 was a significant step in this direction, fostering a more competitive pharmaceutical market.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have a substantial opportunity to expand their presence in Brazil, particularly in the generic drug segment. In 2022, India's pharmaceutical exports to Brazil were valued at USD 695.5 million, encompassing 17,239 shipments from 194 Indian exporters to 1,990 Brazilian buyers. The top five Indian exporters—Sun Pharmaceutical Industries Limited, Baxter Pharmaceuticals India Private Limited, Torrent Pharmaceuticals Ltd, Cipla Limited, and Intas Pharmaceuticals Limited—have established a strong foothold in the Brazilian market.
Given the projected growth in Brazil's generic pharmaceuticals market, Indian exporters can leverage their expertise in generic drug manufacturing to meet the increasing demand. Focusing on high-demand therapeutic areas such as oncology, diabetes, and cardiovascular diseases aligns with market needs and offers avenues for growth. Additionally, the Brazilian government's favorable policies towards generic drugs present a conducive environment for Indian pharmaceutical companies to enhance their market share.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Brazil
Competing origins, India's edge, challenges and threats
1Competing Origins
Brazil's pharmaceutical market is characterized by a diverse array of suppliers, including domestic manufacturers, companies from the United States, European Union (EU) nations, China, and other emerging economies. In 2023, Brazil's pharmaceutical market was valued at approximately $35.6 billion, with imports accounting for about $7.2 billion. The United States and Germany are prominent exporters to Brazil, supplying a significant portion of high-cost medicines. China and India primarily provide raw ingredients, with China also exporting finished pharmaceutical products. Domestic companies, such as Aché Laboratórios Farmacêuticos S.A., lead the market, focusing on the production and sales of prescription drugs.
While precise market share data for each supplier is limited, the United States and Germany are recognized as major exporters to Brazil, particularly for high-cost medicines. China and India are key suppliers of raw materials, with China also exporting finished pharmaceutical products. Domestic companies, including Aché Laboratórios Farmacêuticos S.A., hold a significant share of the market, emphasizing the production and sales of prescription drugs.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages in the Brazilian market. The country's cost competitiveness is notable, with generic drugs accounting for 38% of the Brazilian market in 2024, often priced approximately 35% lower than reference drugs. India's adherence to World Health Organization Good Manufacturing Practices (WHO-GMP) ensures high-quality products. The English-speaking workforce facilitates smoother communication and regulatory compliance. Additionally, India's broad portfolio, including formulations, biologics, and biosimilars, caters to diverse therapeutic areas, enhancing its appeal to Brazilian consumers.
3Challenges & Threats
Despite its strengths, India faces challenges in the Brazilian pharmaceutical market. Regulatory complexities, particularly compliance with Agência Nacional de Vigilância Sanitária (ANVISA) standards, can be intricate. Competition from China, which supplies both raw materials and finished products, intensifies market dynamics. Additionally, perceptions regarding the quality of Indian pharmaceuticals may pose obstacles, necessitating continuous efforts to build trust and demonstrate product efficacy.
FAQ — India to Brazil Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Brazil?
India exported pharmaceuticals worth $695.5M to Brazil across 17,239 verified shipments.
Who are the top Indian pharmaceutical exporters to Brazil?
1. SUN PHARMACEUTICAL INDUSTRIES LIMITED — $295.3M. 2. BAXTER PHARMACEUTICALS INDIA PRIVATE LIMITED — $81.3M. 3. TORRENT PHARMACEUTICALS LTD — $48.0M. Total: 194 suppliers.
Which companies in Brazil import pharmaceuticals from India?
1. UNIAO QUIMICA FARM NAC S/A — $78.0M. 2. M/S. EUROFARMA LABORATORIOS S/A. — $73.3M. 3. M/S. ACHE LABORATORIOS FARMACEUTICO — $57.1M. 1990 buyers total.
What pharmaceutical products does India export most to Brazil?
1. Propofol Intravenous Infusion(1% W/v)-20 ($66.4M, 9.5%); 2. Esio Tablets 40mg Pack:4x7s Batch:ptd4465a ($24.6M, 3.5%); 3. Esio Tablets 40mg Pack:4x7s Batch:ptd4464a ($24.4M, 3.5%); 4. Gaeso Tablets 40mg Pack:4x7s Batch:ptd3473a ($23.8M, 3.4%); 5. Gaeso Tablets 40mg Pack:4x7s Batch:ptd3474a ($23.8M, 3.4%)
Which ports handle pharmaceutical shipments from India to Brazil?
Export: DHANNAD ICD, AHEMDABAD ICD, NHAVA SHEVA SEA (INNSA1), SAHAR AIR CARGO ACC (INBOM4), DELHI AIR. Import: Santos, SANTOS, Guarulhos, GUARULHOS, Rio de Janeiro.
Why does Brazil import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $695.5M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Brazil?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Brazil pharmaceutical trade?
$40.3K per consignment across 17,239 shipments.
How many Indian pharmaceutical companies export to Brazil?
194 Indian companies. Largest: SUN PHARMACEUTICAL INDUSTRIES LIMITED with $295.3M.
How can I find verified Indian pharmaceutical suppliers for Brazil?
TransData Nexus covers 194 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Brazil Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 17,239 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Brazil identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 17,239 verified shipments from 194 Indian exporters to 1990 Brazil buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
194 Exporters
1990 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists