India to Botswana Pharmaceutical Export
Bilateral Trade Intelligence · $42.1M Total Trade · 104 Exporters · 126 Buyers · DGFT Verified · Updated March 2026
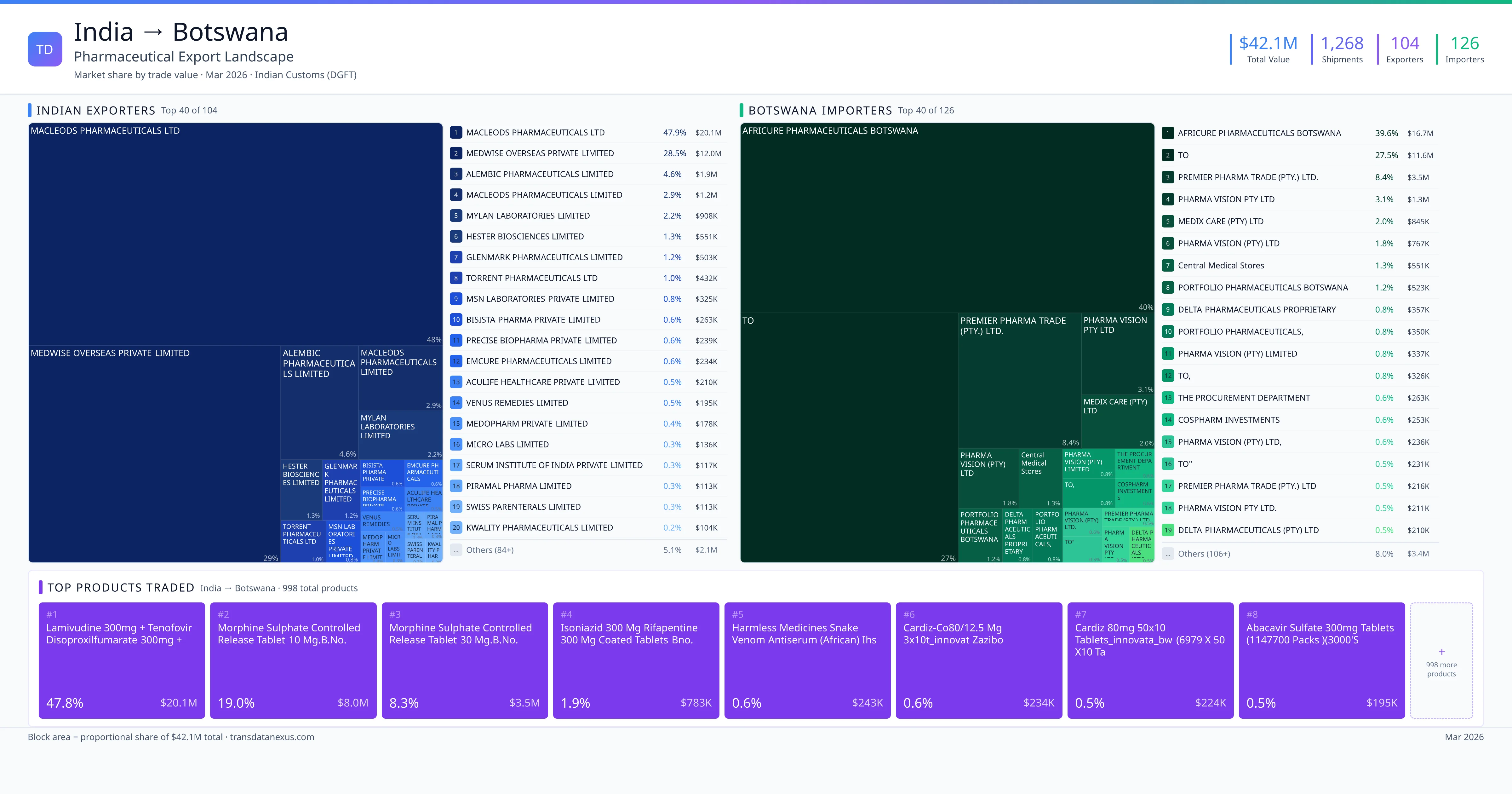
India exported $42.1M worth of pharmaceutical formulations to Botswana across 1,268 verified shipments, sourced from 104 Indian exporters supplying 126 Botswana buyers. The top exporters are MACLEODS PHARMACEUTICALS LTD ($20.1M) and MEDWISE OVERSEAS PRIVATE LIMITED ($12.0M). The leading products are Tenofovir ($17.7M) and Morphine ($8.0M). Average shipment value: $33.2K.
Top Pharmaceutical Formulations — India to Botswana
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Lamivudine 300mg + Tenofovir Disoproxilfumarate 300mg + Dolutegravir Sodium 50m… | $19.0M | 45.2% |
| 2 | Morphine Sulphate Controlled Release Tablet 10 Mg.b.no. Gt1046 Mfg Dt.09/2023 E… | $8.0M | 19.0% |
| 3 | Morphine Sulphate Controlled Release Tablet 30 Mg.b.no. Gt1047 Mfg Dt.09/2023 E… | $3.5M | 8.3% |
| 4 | Lamivudine 300mg + Tenofovir Disoproxilfumarate 300mg + Dolutegravir 50mg Table… | $1.1M | 2.6% |
| 5 | Isoniazid 300 MG Rifapentine 300 MG Coated Tablets Bno. Nie2379a/nie2381a/ Nie2… | $782.5K | 1.9% |
| 6 | Harmless Medicines Snake Venom Antiserum (african) Ihs Lyophllised (afriven) Bn… | $242.5K | 0.6% |
| 7 | Cardiz-co80/12.5 MG 3x10t_innovat Zazibo | $234.4K | 0.6% |
| 8 | Cardiz 80mg 50x10 Tablets_innovata_bw (6979 X 50 X10 Tablets = Total Qty. 3,489… | $224.4K | 0.5% |
| 9 | Abacavir Sulfate 300mg Tablets (1147700 Packs )(3000's Bsp) | $195.1K | 0.5% |
| 10 | Cefotaxime 1g Powder for Injection, 10ml | $180.7K | 0.4% |
| 11 | Ampicillin 500mg Powder for Injection, 1 | $177.4K | 0.4% |
| 12 | Cledomox 625 | $153.2K | 0.4% |
| 13 | Oralcon F Tablets 100x28's Packs 5000nos | $139.7K | 0.3% |
| 14 | Hyan - Levonorgestrel 0.03mg(100's) (100x28 Blister) | $133.0K | 0.3% |
| 15 | Emtricitabine 200mg & Tenofovir Disoproxil Fumarate 300mg Tablets Bno.14250162a… | $129.4K | 0.3% |
India exports 18+ pharmaceutical formulations to Botswana with a combined trade value of $42.1M. Key products include Tenofovir ($17.7M), Morphine ($8.0M), Isoniazid ($783K), Ampicillin ($177K), Emtricitabine ($129K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,268 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Botswana Trade Routes
These are the top pharmaceutical products exported from India to Botswana, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Botswana buyers, regulatory requirements, and logistics for that specific product corridor. Products include Tenofovir ($17.7M), Morphine ($8.0M), Isoniazid ($783K), Ampicillin ($177K), Emtricitabine ($129K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Botswana
104 Indian pharmaceutical companies export finished formulations to Botswana. Leading exporters include Macleods Pharmaceuticals Ltd, Medwise Overseas Private Limited, Alembic Pharmaceuticals Limited, Macleods Pharmaceuticals Limited. The top exporter accounts for 47.9% of total India–Botswana pharma exports. Source: Indian Customs (DGFT).
Top Botswana Buyers from India
126 companies in Botswana import pharmaceutical formulations from India. Top buyers include Africure Pharmaceuticals Botswana, To, Premier Pharma Trade (pty.) Ltd., Pharma Vision Pty Ltd. The largest buyer accounts for 39.6% of India–Botswana pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Botswana Pharmaceutical Shipments
Indian Export Ports
Botswana Import Ports
Trade Statistics
Other Destinations
Product Routes
Product Categories & Therapeutic Trends — India to Botswana
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Botswana are predominantly concentrated in antiretroviral (ARV) therapies, particularly the combination of Lamivudine, Tenofovir Disoproxil Fumarate, and Dolutegravir Sodium. This combination therapy, essential for HIV/AIDS management, accounted for approximately 42% of the total export value, amounting to $17.7 million. The dominance of ARVs aligns with Botswana's substantial efforts in combating HIV/AIDS, which remains a significant public health challenge. The country's healthcare system has been heavily invested in providing comprehensive HIV treatment and care, leading to a sustained demand for these medications.
Additionally, the export of controlled-release Morphine Sulphate tablets, totaling $11.5 million (27.3% of total exports), indicates a notable demand for pain management solutions. This demand is likely driven by the need for palliative care services, particularly for patients with advanced-stage diseases such as cancer. The presence of these products in the export data suggests that Botswana's healthcare system is addressing the need for effective pain management, reflecting a broader trend towards improving palliative care services.
2Emerging Opportunities
The projected growth of Botswana's pharmaceutical market, with revenues expected to reach $106.22 million by 2025 and a compound annual growth rate (CAGR) of 5.07% from 2025 to 2030, presents significant opportunities for Indian pharmaceutical exporters. This growth is driven by an increasing prevalence of chronic diseases, such as diabetes and hypertension, and a rising demand for specialized treatments. The government's commitment to enhancing healthcare infrastructure further supports this expansion. For Indian exporters, this trend signifies a growing market for both generic and specialized pharmaceutical products.
Botswana's efforts to establish local pharmaceutical manufacturing capabilities, as reported in February 2026, indicate a strategic move towards self-sufficiency in essential medicines. While this development may initially reduce the demand for imported pharmaceuticals, it also opens avenues for Indian companies to collaborate in technology transfer, capacity building, and supplying raw materials or specialized products not yet produced locally.
3Demand Drivers
Botswana's healthcare spending per capita was $477.83 in 2022, reflecting a 2.91% decline from 2021. Despite this decline, the overall healthcare expenditure remains substantial, indicating a continued commitment to improving health services. The rising prevalence of non-communicable diseases (NCDs), including diabetes, hypertension, and obesity, is placing additional strain on the healthcare system. The 2024 STEPS survey highlighted that nearly 40% of adults have high blood pressure, with a significant portion lacking adequate control. (afro.who.int) This epidemiological shift is driving demand for medications to manage these chronic conditions.
Furthermore, Botswana's aging population and increasing urbanization are contributing to a higher burden of NCDs. The government's focus on enhancing healthcare infrastructure and services, including the establishment of the Botswana Public Health Institute, underscores the need for a diverse range of pharmaceutical products to meet the evolving health needs of the population. (afro.who.int) These factors collectively create a robust demand for pharmaceutical imports, presenting opportunities for Indian exporters to supply a variety of products tailored to Botswana's healthcare requirements.
Trade Policy & Tariff Intelligence — India and Botswana
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Botswana's tariff structure for pharmaceutical formulations, classified under HS codes 3003 and 3004, is influenced by its membership in the Southern African Customs Union (SACU). SACU members, including Botswana, typically apply the Common Customs Tariff (CCT) established by the Southern African Development Community (SADC). This tariff framework harmonizes duty rates across member states to facilitate intra-regional trade. While specific MFN (Most-Favored-Nation) tariff rates for pharmaceutical products under HS codes 3003 and 3004 are not publicly detailed, they are generally aligned with the CCT provisions. For precise and current rates, consulting Botswana's Customs and Excise Department or the SADC Secretariat is recommended.
Regarding preferential duty rates, Botswana's participation in SACU and SADC agreements may offer reduced tariffs for intra-regional trade. However, the extent of these preferences for pharmaceutical products depends on the specific terms of each agreement. To determine applicable preferential rates, it is essential to review the latest trade agreements and consult with Botswana's Customs and Excise Department.
2Trade Agreements
As of March 2026, there is no publicly available information indicating a Free Trade Agreement (FTA) between India and Botswana. Both countries are members of the World Trade Organization (WTO), which provides a framework for trade relations, including commitments to MFN treatment and tariff bindings. While SACU and SADC agreements may influence Botswana's trade policies, their direct impact on pharmaceutical trade with India is not specified. For the most accurate and up-to-date information, it is advisable to consult official trade authorities or the SADC Secretariat.
3IP & Patent Landscape
Botswana's intellectual property (IP) framework, including patent laws, is designed to protect innovations and may impact the entry of generic pharmaceutical products. The country has provisions for compulsory licensing, allowing the government to authorize the use of a patented invention without the consent of the patent holder under specific conditions, such as public non-commercial use. This mechanism can facilitate the procurement of essential medicines at affordable prices. However, the application of compulsory licensing in Botswana is not well-documented, and its use in the pharmaceutical sector remains unclear. For detailed information on IP provisions affecting generic drug imports, consulting Botswana's Ministry of Trade and Industry or the Botswana Intellectual Property Office is recommended.
FAQ — India to Botswana Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Botswana?
India exported pharmaceuticals worth $42.1M to Botswana across 1,268 verified shipments.
Who are the top Indian pharmaceutical exporters to Botswana?
1. MACLEODS PHARMACEUTICALS LTD — $20.1M. 2. MEDWISE OVERSEAS PRIVATE LIMITED — $12.0M. 3. ALEMBIC PHARMACEUTICALS LIMITED — $1.9M. Total: 104 suppliers.
Which companies in Botswana import pharmaceuticals from India?
1. AFRICURE PHARMACEUTICALS BOTSWANA — $16.7M. 2. TO — $11.6M. 3. PREMIER PHARMA TRADE (PTY.) LTD. — $3.5M. 126 buyers total.
What pharmaceutical products does India export most to Botswana?
1. Lamivudine 300mg + Tenofovir Disoproxilfumarate 300mg + Dolutegravir Sodium 50m… ($19.0M, 45.2%); 2. Morphine Sulphate Controlled Release Tablet 10 Mg.b.no. Gt1046 Mfg Dt.09/2023 E… ($8.0M, 19.0%); 3. Morphine Sulphate Controlled Release Tablet 30 Mg.b.no. Gt1047 Mfg Dt.09/2023 E… ($3.5M, 8.3%); 4. Lamivudine 300mg + Tenofovir Disoproxilfumarate 300mg + Dolutegravir 50mg Table… ($1.1M, 2.6%); 5. Isoniazid 300 MG Rifapentine 300 MG Coated Tablets Bno. Nie2379a/nie2381a/ Nie2… ($782.5K, 1.9%)
Which ports handle pharmaceutical shipments from India to Botswana?
Export: DELHI AIR, MUNDRA SEA, COCHIN SEA, CHENNAI AIR, DELHI AIR CARGO ACC (INDEL4). Import: GABORONE, Gaborone, JOHANNESBURG - JOHAN, Durban, DURBAN.
Why does Botswana import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $42.1M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Botswana?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Botswana pharmaceutical trade?
$33.2K per consignment across 1,268 shipments.
How many Indian pharmaceutical companies export to Botswana?
104 Indian companies. Largest: MACLEODS PHARMACEUTICALS LTD with $20.1M.
How can I find verified Indian pharmaceutical suppliers for Botswana?
TransData Nexus covers 104 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Botswana Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,268 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Botswana identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,268 verified shipments from 104 Indian exporters to 126 Botswana buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
104 Exporters
126 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists